Revolutionizing Pharmaceutical Intermediate Production: Scalable Synthesis of High-Purity 5-Trifluoromethyl Triazoles for Global Supply Chains
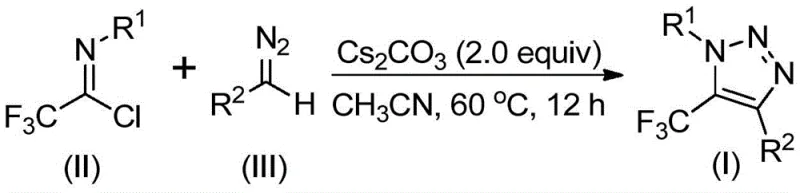
Patent CN113121462B introduces a groundbreaking methodology for synthesizing 5-trifluoromethyl-substituted 1,2,3-triazole compounds through a base-promoted reaction pathway that eliminates the need for hazardous azides and transition metal catalysts. This innovative approach represents a significant advancement in heterocyclic chemistry by utilizing readily available diazo compounds and trifluoroethylimidoyl chlorides as starting materials under mild reaction conditions. The process operates efficiently at 60°C in acetonitrile solvent with cesium carbonate as the base promoter, achieving high yields across diverse substrate combinations without requiring specialized equipment or safety protocols for explosive intermediates. This patent addresses critical industry challenges in producing fluorinated triazole derivatives that serve as essential building blocks for pharmaceuticals and agrochemicals, offering a safer and more sustainable alternative to conventional synthetic routes. The elimination of toxic reagents and metal residues directly enhances product purity profiles while reducing environmental impact throughout the manufacturing lifecycle. This development holds substantial promise for global pharmaceutical supply chains seeking reliable sources of high-purity fluorinated heterocyclic intermediates with improved safety margins and operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for trifluoromethyl-substituted triazoles predominantly rely on copper-catalyzed azide-alkyne cycloadditions followed by trifluoromethylation steps or organocatalytic reactions involving azide compounds, both of which present significant operational hazards due to the inherent instability and explosive nature of organic azides that require specialized handling procedures and safety infrastructure. These conventional methodologies often necessitate expensive transition metal catalysts that introduce complex purification challenges to remove trace metal residues below pharmacopeial limits, substantially increasing production costs and extending processing timelines through additional chromatographic steps. The requirement for multiple synthetic transformations to incorporate the trifluoromethyl group typically results in lower overall yields and generates considerable waste streams that complicate environmental compliance and increase disposal costs for manufacturing facilities. Furthermore, the sensitivity of azide intermediates to temperature fluctuations and mechanical shock creates substantial supply chain vulnerabilities, as these compounds cannot be stored or transported safely over extended periods, forcing manufacturers to maintain just-in-time inventory systems that compromise production flexibility and increase lead times for critical intermediates.
The Novel Approach
The patented methodology overcomes these limitations through an elegant base-promoted reaction between trifluoroethylimidoyl chlorides and diazo compounds that completely eliminates the need for both azide chemistry and transition metal catalysts while maintaining excellent functional group tolerance across diverse substrate combinations. This innovative process operates under mild conditions (60°C in acetonitrile) using inexpensive cesium carbonate as the base promoter, enabling straightforward implementation in standard manufacturing facilities without requiring specialized safety equipment or hazardous material handling protocols. The single-step transformation directly constructs the triazole ring with the trifluoromethyl group already incorporated, significantly reducing the number of synthetic operations while improving overall atom economy and minimizing waste generation compared to conventional multi-step approaches. Crucially, the absence of metal catalysts ensures the final products meet stringent purity specifications required for pharmaceutical applications without requiring extensive purification procedures to remove metal contaminants, thereby streamlining quality control processes and enhancing supply chain reliability through simplified production workflows that can be readily scaled from laboratory to commercial manufacturing volumes.
Mechanistic Insights into Base-Promoted Triazole Formation
The reaction mechanism proceeds through a base-promoted intermolecular nucleophilic addition where the diazo compound attacks the electrophilic carbon of the trifluoroethylimidoyl chloride, forming a key tetrahedral intermediate that subsequently undergoes elimination to generate a nitrile imine species. This reactive intermediate then participates in an intramolecular 5-endo-dig cyclization process where the nitrogen atom attacks the electron-deficient carbon center, facilitating ring closure to form the triazole core structure with precise regioselectivity at the 1,4 positions. The mild reaction conditions (60°C) and use of cesium carbonate as a weak base promote controlled progression through these mechanistic steps without triggering competing side reactions that could lead to dimerization or decomposition of sensitive intermediates. Computational studies referenced in the patent indicate that the electron-withdrawing trifluoromethyl group stabilizes the transition state during cyclization while simultaneously activating the imidoyl chloride toward nucleophilic attack, creating a self-accelerating reaction pathway that achieves high conversion rates across diverse substrate combinations without requiring elevated temperatures or prolonged reaction times that could compromise product integrity.
The mechanism inherently minimizes impurity formation through its concerted cyclization pathway that avoids free radical intermediates or unstable metal complexes commonly associated with conventional triazole syntheses. The absence of transition metals eliminates potential sources of heavy metal contamination while preventing unwanted redox side reactions that could generate colored impurities or isomerized byproducts. The mild thermal profile (60°C) prevents thermal degradation pathways that typically produce tar formation or decomposition products in traditional high-temperature processes, resulting in cleaner reaction profiles that simplify downstream purification requirements. Furthermore, the well-defined reaction stoichiometry (1:1.5:2 molar ratio of imidoyl chloride:diazo compound:cesium carbonate) ensures complete consumption of starting materials while minimizing unreacted precursors that could complicate isolation procedures or require additional washing steps to remove residual reagents from the final product stream.
How to Synthesize 5-Trifluoromethyl Triazoles Efficiently
This patented synthesis represents a significant advancement in triazole chemistry by providing a streamlined pathway that eliminates hazardous reagents while maintaining excellent functional group tolerance across diverse substrate combinations. The process demonstrates remarkable versatility in accommodating various aromatic and aliphatic substituents on both reaction partners, enabling customization of the final triazole products for specific pharmaceutical applications without requiring major modifications to the core reaction protocol. By operating under mild conditions with readily available starting materials and simple workup procedures, this methodology offers substantial advantages for industrial implementation while ensuring consistent production of high-purity intermediates suitable for demanding pharmaceutical applications. Detailed standardized synthesis steps are provided below to facilitate seamless technology transfer from laboratory development to commercial manufacturing scale.
- Prepare reaction mixture by combining trifluoroethylimidoyl chloride (II), diazo compound (III), cesium carbonate (2.0 equivalents), and molecular sieves in acetonitrile solvent at room temperature
- Heat reaction mixture to 60°C under nitrogen atmosphere and maintain at this temperature for 12 hours with continuous stirring
- Perform post-reaction workup by filtration through silica gel followed by column chromatography purification using standard elution protocols
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate supply chains by delivering a safer, more efficient production process that enhances both cost-effectiveness and supply reliability for global manufacturing operations. The elimination of hazardous azide chemistry removes significant regulatory burdens and safety infrastructure requirements that typically increase capital expenditures and operational complexity for chemical manufacturers serving the pharmaceutical industry. By utilizing commercially available starting materials with extended shelf stability compared to explosive azide compounds, this approach substantially improves supply chain resilience while reducing inventory management challenges associated with time-sensitive hazardous materials that require specialized storage and handling protocols.
- Cost Reduction in Manufacturing: The complete elimination of expensive transition metal catalysts removes both the raw material cost burden and the extensive purification requirements needed to remove trace metal residues from final products, resulting in substantial cost savings through simplified processing workflows and reduced quality control testing requirements. The use of inexpensive cesium carbonate as a base promoter instead of costly organocatalysts further optimizes raw material expenses while maintaining excellent reaction efficiency across diverse substrate combinations without requiring specialized equipment or safety infrastructure for hazardous reagents.
- Enhanced Supply Chain Reliability: The utilization of stable, non-hazardous starting materials with extended shelf life significantly improves supply chain robustness by eliminating dependencies on time-sensitive hazardous intermediates that require specialized handling and transportation protocols. This approach enables manufacturers to maintain strategic inventory buffers without safety concerns, reducing vulnerability to supply disruptions while providing greater flexibility in production scheduling to accommodate fluctuating demand patterns from global pharmaceutical customers seeking reliable sources of high-purity fluorinated intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions (60°C) and simple workup procedures enable straightforward scale-up from laboratory to commercial production volumes without requiring specialized equipment modifications or additional safety measures typically needed for high-temperature processes or hazardous reagent handling. The elimination of toxic metal catalysts substantially reduces waste stream complexity while improving environmental compliance profiles through decreased generation of hazardous byproducts that require special disposal procedures, making this process particularly attractive for manufacturers seeking sustainable production methods that align with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding the implementation of this novel triazole synthesis methodology based on detailed analysis of the patented process parameters and performance characteristics observed during extensive laboratory validation studies.
Q: How does this metal-free synthesis improve safety compared to conventional azide-based methods?
A: The patented process completely eliminates hazardous organic azides that are explosive and require specialized handling infrastructure, replacing them with stable diazo compounds that can be safely stored and transported without special precautions.
Q: What are the scalability advantages of this base-promoted triazole formation process?
A: The mild reaction conditions (60°C), simple workup procedures, and absence of hazardous reagents enable straightforward scale-up from laboratory to commercial production volumes without requiring specialized equipment modifications or additional safety measures.
Q: How does the elimination of transition metal catalysts impact final product purity for pharmaceutical applications?
A: Removing metal catalysts eliminates potential heavy metal contamination sources, ensuring products meet stringent pharmacopeial purity requirements without requiring extensive purification steps to remove trace metal residues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Triazole Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation for comprehensive quality assurance. This patented methodology represents just one example of our commitment to developing innovative solutions that address critical challenges in pharmaceutical intermediate manufacturing through scientifically rigorous process development and optimization expertise. As a trusted partner to leading global pharmaceutical companies, we combine deep technical knowledge with flexible manufacturing capabilities to deliver high-purity fluorinated heterocyclic compounds that meet the most demanding regulatory requirements while supporting sustainable production goals across international supply chains.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis can optimize your specific manufacturing requirements. Please contact us to obtain detailed COA data and route feasibility assessments tailored to your production scale and quality specifications for immediate implementation in your pharmaceutical development programs.
