Advanced Synthetic Routes for Capsaicin Homologues: Commercial Scalability and Purity
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways that balance high purity with environmental sustainability. Patent CN101717346B presents a significant advancement in the artificial synthesis of capsaicin homologues, specifically targeting the production of capsaicin, dihydrocapsaicin, and nordihydrocapsaicin. Unlike traditional extraction methods which yield complex mixtures with variable compositions, this synthetic approach offers precise control over molecular structure and impurity profiles. The core innovation lies in the construction of the long-chain fatty acid moiety without relying on toxic phosphine reagents, marking a substantial shift towards greener chemistry in the manufacturing of high-value bioactive compounds. This technical breakthrough provides a reliable foundation for producing consistent batches of active ingredients essential for topical analgesics and specialized coating applications.
Historically, the industrial demand for capsaicinoids has been met through solvent extraction from natural peppers, a process fraught with variability in pungency and composition. However, the artificial synthesis method disclosed in this patent addresses the critical limitations of natural sourcing by providing a defined chemical identity. The ability to synthesize specific homologues allows manufacturers to tailor the physicochemical properties of the final product, ensuring batch-to-batch consistency that is crucial for regulatory compliance in pharmaceutical formulations. By establishing a fully synthetic route, producers can decouple supply from agricultural fluctuations, offering a stable source of raw materials for downstream formulation teams who require stringent quality specifications for their final products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional synthetic routes for constructing the branched long-chain acids required for capsaicinoids often rely heavily on Wittig olefination reactions. These traditional methods typically utilize triphenylphosphine as a key reagent to form the carbon-carbon double bond necessary for the unsaturated acid precursors. While chemically effective on a laboratory scale, the use of triphenylphosphine generates stoichiometric amounts of triphenylphosphine oxide as a byproduct. This byproduct is notoriously difficult to separate from the desired product, often requiring extensive chromatography or recrystallization steps that reduce overall yield and increase solvent consumption. Furthermore, triphenylphosphine is toxic and poses significant handling hazards in large-scale operations, creating additional burdens for waste management and environmental compliance teams who must treat phosphorus-containing effluents before discharge.
The Novel Approach
The novel approach detailed in patent CN101717346B circumvents these issues by employing a sequence involving Grignard addition followed by an orthoester Claisen rearrangement. This strategy constructs the carbon skeleton using readily available alkyl bromides and acrolein, followed by a thermal rearrangement that establishes the necessary branching and unsaturation without phosphine reagents. The elimination of toxic phosphine compounds simplifies the workup procedure significantly, as there is no stubborn phosphine oxide residue to remove. This streamlined process not only enhances the environmental profile of the manufacturing operation but also reduces the number of purification steps required, leading to a more efficient use of resources and a cleaner final product profile that meets the rigorous standards expected by a reliable pharmaceutical intermediates supplier.
Mechanistic Insights into Grignard Addition and Orthoester Rearrangement
The synthesis begins with the formation of a Grignard reagent from an appropriate alkyl bromide, such as 2-bromopropane or 1-bromo-3-methylbutane, reacting with magnesium metal in anhydrous ether. This organometallic species then undergoes nucleophilic addition to acrolein, yielding an unsaturated alcohol intermediate. The critical step follows, where this alcohol reacts with triethyl orthoacetate in the presence of a catalytic amount of propionic acid. Upon heating, this mixture undergoes a [3,3]-sigmatropic rearrangement, effectively extending the carbon chain and introducing the ester functionality with high regioselectivity. This mechanistic pathway ensures that the branching methyl group is positioned precisely at the required location on the fatty acid chain, which is essential for the biological activity of the resulting capsaicinoid homologue.
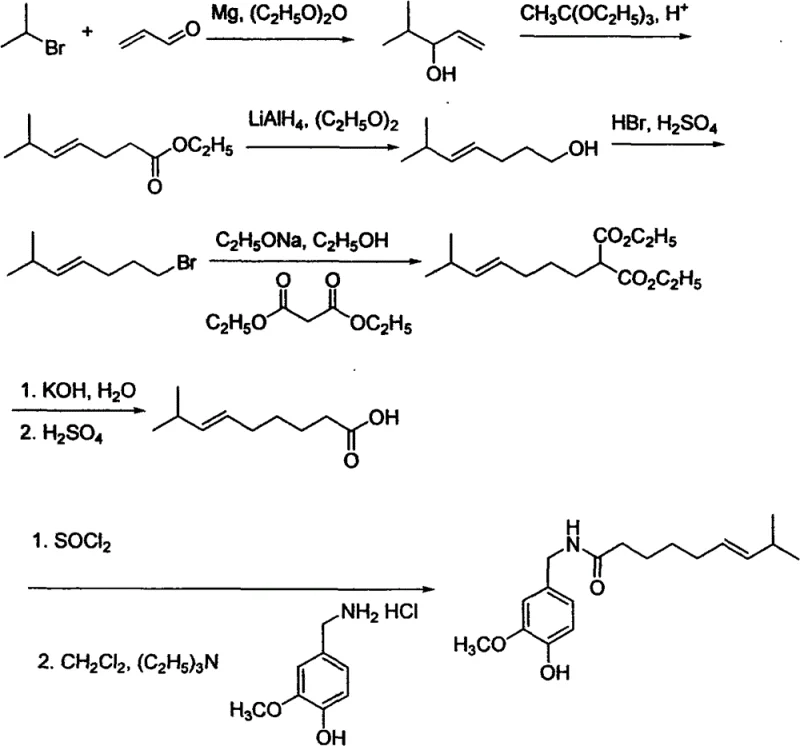
Following the rearrangement, the unsaturated ester is subjected to reduction using lithium aluminum hydride to generate the corresponding allylic alcohol. This alcohol is then converted to an alkyl bromide using hydrobromic acid, setting the stage for chain extension via malonate alkylation. The diethyl malonate anion attacks the alkyl bromide, adding two carbons to the chain and introducing the carboxylic acid precursor functionality. Subsequent hydrolysis and decarboxylation yield the target long-chain unsaturated acid. For dihydrocapsaicin, a hydrogenation step using Raney nickel saturates the double bond. Finally, the acid is activated with thionyl chloride and coupled with vanillylamine hydrochloride to form the amide bond. This multi-step sequence demonstrates a high degree of chemical control, ensuring high-purity capsaicin homologues with minimal structural impurities.
How to Synthesize Capsaicin Homologues Efficiently
The implementation of this synthetic route requires careful control of reaction conditions, particularly during the Grignard formation and the orthoester rearrangement steps. Moisture exclusion is critical during the organometallic phases to prevent reagent decomposition, while temperature control during the rearrangement dictates the ratio of isomers formed. The patent outlines specific molar ratios and distillation parameters that optimize the yield of the desired intermediates. For process chemists looking to adopt this methodology, adherence to the specified stoichiometry and purification protocols is essential to replicate the high purity levels reported. The detailed standardized synthesis steps see the guide below provide a roadmap for translating this laboratory-scale success into a robust manufacturing protocol.
- Preparation of unsaturated alcohol intermediates via Grignard reaction of alkyl bromides with acrolein followed by orthoester Claisen rearrangement.
- Conversion of unsaturated esters to long-chain acids through reduction, bromination, malonate alkylation, and subsequent hydrolysis.
- Final amidation of the synthesized long-chain acid chlorides with vanillylamine hydrochloride to yield pure capsaicin homologues.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift away from triphenylphosphine-based chemistry offers substantial cost savings in fine chemical manufacturing. The removal of expensive phosphine reagents and the associated waste treatment costs directly impacts the bottom line, making the synthetic product more competitive against natural extracts. Additionally, the starting materials such as alkyl bromides and acrolein are commodity chemicals with stable supply chains, reducing the risk of raw material shortages that often plague botanical extraction processes. This stability allows for better long-term planning and contract negotiation, ensuring that production schedules are not disrupted by agricultural variances or seasonal availability of pepper crops.
- Cost Reduction in Manufacturing: The elimination of triphenylphosphine removes the need for costly disposal of phosphorus-containing waste, which is a significant expense in chemical processing. Furthermore, the simplified purification process reduces solvent consumption and energy usage associated with multiple recrystallization or chromatography steps. By streamlining the synthesis to fewer effective steps with higher atom economy, the overall production cost per kilogram is significantly reduced. This efficiency allows suppliers to offer more competitive pricing structures without compromising on the quality or purity specifications required for pharmaceutical grade materials.
- Enhanced Supply Chain Reliability: Relying on petrochemical-derived starting materials rather than agricultural products insulates the supply chain from weather-related disruptions and price volatility inherent in the spice market. The synthetic route ensures a consistent quality of raw materials, which translates to predictable lead times for high-purity active ingredients. Manufacturers can guarantee continuous supply to their clients, mitigating the risk of stockouts that could halt downstream formulation lines. This reliability is a key differentiator for partners seeking a dependable source of critical intermediates for their commercial products.
- Scalability and Environmental Compliance: The process is explicitly designed for large-scale industrial production, utilizing standard unit operations such as distillation and liquid-liquid extraction that are easily scalable from pilot plant to commercial tonnage. The absence of toxic phosphine byproducts simplifies environmental permitting and reduces the regulatory burden on the manufacturing site. This alignment with green chemistry principles not only future-proofs the production facility against tightening environmental regulations but also enhances the corporate sustainability profile of the brands utilizing these synthetically derived ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of capsaicin homologues. These answers are derived directly from the technical specifications and experimental data provided in the patent literature. Understanding these details helps stakeholders make informed decisions about integrating these synthetic intermediates into their product development pipelines. The clarity on purity, scalability, and environmental impact is vital for cross-functional teams evaluating new supply partners.
Q: How does this synthesis method improve environmental safety compared to traditional routes?
A: This method eliminates the use of triphenylphosphine, a toxic reagent common in Wittig reactions, thereby avoiding the generation of triphenylphosphine oxide waste which is difficult to remove and environmentally hazardous.
Q: What is the purity level achievable for dihydrocapsaicin using this process?
A: According to patent data, the process yields dihydrocapsaicin with a purity exceeding 98.5%, verified by HPLC retention time comparison with standard references and consistent NMR spectral data.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method, particularly for dihydrocapsaicin, is designed for large-scale industrial production due to the use of commercially available raw materials and simplified purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Capsaicin Homologues Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of transitioning from extraction-dependent models to robust synthetic solutions for high-value natural product analogues. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be realized in your supply chain. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of capsaicin or dihydrocapsaicin meets the exacting standards required for pharmaceutical and specialty chemical applications. Our commitment to quality assurance means that you receive materials that are consistent, documented, and ready for immediate integration into your formulations.
We invite you to discuss how our synthetic capabilities can optimize your raw material costs and secure your supply chain against market fluctuations. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact us to request specific COA data and route feasibility assessments for your next project. By partnering with us, you gain access to advanced chemical technologies that drive efficiency and reliability in the production of complex organic molecules.
