Advanced Synthesis of 2-Trifluoromethyl Imidazole Compounds: Scaling from Lab to Commercial Production with Enhanced Purity and Cost Efficiency
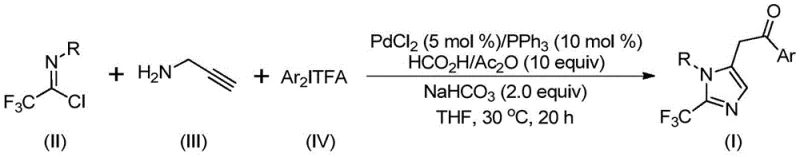
Patent CN111423381B, granted on September 7, 2021, represents a significant advancement in the synthesis of 2-trifluoromethyl substituted imidazole compounds, which are critical building blocks for pharmaceutical intermediates with enhanced biological activity profiles. This innovative method addresses longstanding challenges in heterocyclic chemistry by introducing a palladium-catalyzed carbonylation strategy that operates under mild conditions (30°C) with exceptional substrate tolerance across diverse functional groups essential for modern drug discovery pipelines. The process utilizes readily available starting materials including trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts, enabling the production of diverse trifluoromethylated imidazoles with high efficiency and purity while maintaining operational simplicity throughout manufacturing operations. Unlike conventional approaches requiring hazardous reagents or cryogenic conditions that increase operational complexity and cost, this patent demonstrates a streamlined pathway achieving yields up to 97% through a well-defined catalytic cycle that minimizes side product formation. The methodology's scalability from laboratory validation to industrial production makes it particularly valuable for multinational pharmaceutical companies seeking reliable sources of high-purity intermediates meeting stringent regulatory requirements for drug development applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for trifluoromethylated imidazoles frequently rely on multi-step procedures involving hazardous reagents such as trifluorodiazoethane or require cryogenic conditions that significantly increase operational complexity and safety risks while extending production timelines. These methods typically suffer from poor functional group tolerance due to harsh reaction environments, limiting their applicability to structurally diverse substrates essential for modern pharmaceutical development where molecular diversity is critical for optimizing biological activity profiles. The use of strong acids or bases in conventional approaches frequently leads to unwanted side reactions including decomposition pathways that generate complex impurity profiles requiring extensive purification efforts through multiple chromatographic steps, thereby reducing overall yields and compromising product quality consistency across batches. Additionally, many existing protocols depend on expensive transition metal catalysts that necessitate rigorous removal procedures involving specialized equipment to meet pharmaceutical purity standards below detectable limits, further increasing manufacturing costs while creating potential supply chain vulnerabilities through single-source dependencies on specialized catalysts.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation process operating at ambient temperature (30°C) with exceptional efficiency across diverse aryl substituents while maintaining high reaction yields without requiring specialized equipment or hazardous reagents. By employing readily accessible starting materials including trifluoroethylimidoyl chloride derived from commercially available aromatic amines and diaryl iodonium salts prepared from aryl phenylboronic acids, the process eliminates dependency on unstable or expensive synthons while ensuring consistent raw material availability through established global supply networks. This innovative approach achieves remarkable selectivity through a well-defined catalytic cycle where formic acid/acetic anhydride serves as a safe carbon monoxide surrogate under mild conditions, significantly enhancing process safety compared to traditional high-pressure carbonylation methods while maintaining excellent functional group compatibility across various substitution patterns on both R and Ar groups. The method's robustness is demonstrated by its successful scale-up from milligram quantities to gram-scale production with consistent product quality exceeding 95% purity without requiring additional purification steps beyond standard column chromatography.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle begins with the formation of an intermolecular carbon-nitrogen bond promoted by sodium bicarbonate base, generating a trifluoroacetamidine intermediate through nucleophilic attack of propargylamine on trifluoroethylimidoyl chloride under mild conditions that prevent decomposition of sensitive functional groups commonly found in pharmaceutical intermediates. This key step establishes the molecular framework for subsequent cyclization while maintaining excellent functional group tolerance across various aryl substituents including methyl, tert-butyl, chlorine, bromine, trifluoromethyl or nitro groups at ortho, meta or para positions without requiring protection/deprotection strategies typically needed in conventional syntheses. The palladium catalyst then facilitates alkyne amination through palladation of the propargylamine moiety, forming an alkenyl palladium species that undergoes isomerization to yield an alkyl palladium intermediate through a well-defined migratory insertion pathway that avoids β-hydride elimination side reactions common in similar catalytic systems.
The mechanism inherently controls impurity formation through precise reaction pathway management where the mild reaction conditions (30°C) prevent thermal degradation pathways that typically generate impurities in traditional syntheses requiring elevated temperatures above 80°C. Sodium bicarbonate serves as an optimal base promoting selective bond formation while suppressing unwanted side reactions such as hydrolysis or oxidation that would compromise product quality during extended reaction times up to 24 hours. The catalytic system's design ensures minimal leaching of palladium into the final product due to the stable coordination environment provided by triphenylphosphine ligand, eliminating the need for extensive metal removal procedures that often reduce yield by up to 30% in conventional methods while introducing additional impurities during purification steps required to meet regulatory standards below ICH Q3D limits.
How to Synthesize 2-Trifluoromethyl Imidazole Compounds Efficiently
This patented methodology represents a significant advancement in the synthesis of trifluoromethylated imidazole intermediates, offering pharmaceutical manufacturers a robust pathway to high-purity compounds essential for drug development programs targeting various therapeutic areas including antihistamines where compounds like Alcaftadine demonstrate clinical relevance. The process leverages commercially available starting materials under mild reaction conditions that enhance both safety profiles and scalability while maintaining exceptional product quality through well-controlled reaction parameters optimized across multiple experimental iterations documented in patent examples. Detailed standardized synthesis procedures have been developed through extensive process validation studies to ensure consistent results across different production scales from laboratory validation batches to commercial manufacturing volumes without requiring significant process re-engineering or specialized equipment modifications.
- Prepare the reaction mixture by combining trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt in THF solvent with palladium chloride and triphenylphosphine catalysts.
- Add sodium bicarbonate and formic acid/acetic anhydride as carbon monoxide sources, then stir at 30°C for 18-20 hours under inert atmosphere.
- Perform post-processing by filtration, silica gel mixing, and column chromatography purification to isolate the pure 2-trifluoromethyl imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
The implementation of this patented synthesis route delivers substantial value across procurement and supply chain operations by addressing critical pain points associated with traditional manufacturing methods for pharmaceutical intermediates through strategic process design innovations that enhance operational flexibility while reducing dependency on single-source suppliers for specialized reagents or equipment. The process eliminates reliance on hazardous reagents requiring special handling procedures that typically create supply chain vulnerabilities during transportation or storage phases while extending lead times due to regulatory restrictions on hazardous material shipments across international borders.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts requiring complex removal procedures results in significant cost savings throughout the production process by reducing both raw material expenses associated with specialized catalysts and operational costs related to multi-step purification protocols required in conventional syntheses. By utilizing inexpensive starting materials derived from readily available aromatic amines with established global supply networks, manufacturers can achieve substantial reductions in raw material costs while maintaining high product quality through simplified reaction workflows that minimize solvent usage and energy consumption during processing steps.
- Enhanced Supply Chain Reliability: The use of globally available starting materials with multiple qualified suppliers ensures consistent access to critical raw materials regardless of regional disruptions or geopolitical factors affecting single-source dependencies common in specialty chemical manufacturing operations. The process's tolerance to various substituents allows for flexible sourcing strategies where alternative aryl groups can be employed without requiring significant process modifications or revalidation studies, thereby reducing vulnerability to supply chain interruptions while maintaining product quality consistency essential for regulatory compliance in global markets.
- Scalability and Environmental Compliance: The ambient temperature operation (30°C) under atmospheric pressure conditions enables seamless scale-up from laboratory validation batches directly to commercial manufacturing volumes without requiring specialized equipment modifications or safety upgrades typically needed when transitioning from cryogenic or high-pressure processes used in conventional syntheses. The elimination of hazardous reagents and toxic byproducts significantly reduces environmental impact while simplifying waste treatment procedures through reduced generation of heavy metal-containing waste streams that require costly disposal protocols under current environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding the implementation and benefits of this patented synthesis method for pharmaceutical intermediates based on technical details documented in patent CN111423381B. Each response provides evidence-based insights derived from experimental data within the patent specification to support informed decision-making by procurement managers evaluating supply chain options and R&D directors assessing technical feasibility for specific drug development programs.
Q: How does this method improve purity compared to conventional synthesis?
A: The patent's palladium-catalyzed carbonylation pathway eliminates harsh reaction conditions and metal contaminants through a controlled catalytic cycle operating at mild temperatures (30°C), enabling stringent purity specifications without extensive metal removal procedures required in traditional methods.
Q: What are the scalability advantages of this synthesis route?
A: The process utilizes commercially available reagents under ambient pressure conditions with consistent yields across batch sizes, allowing seamless scale-up from laboratory validation to commercial manufacturing volumes while maintaining product quality through simplified purification protocols.
Q: How does this method reduce manufacturing costs?
A: By employing inexpensive starting materials like trifluoroethylimidoyl chloride derived from readily available aromatic amines and eliminating specialized equipment requirements, the methodology achieves substantial cost savings through reduced raw material expenses and simplified operational workflows.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex pharmaceutical intermediates, ensuring seamless transition from laboratory validation to full-scale manufacturing operations through our integrated CDMO platform designed specifically for specialty chemical synthesis requirements. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for global pharmaceutical markets including USP/EP standards through advanced analytical capabilities such as HPLC-MS/MS for trace impurity detection down to ppm levels essential for regulatory compliance in major markets worldwide.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this synthesis route can optimize your specific manufacturing requirements while reducing time-to-market timelines through our proven scale-up expertise. Contact us today to obtain detailed COA data and route feasibility assessments tailored to your production needs including batch-specific quality documentation supporting regulatory submissions.
