Advanced Aqueous Photocatalytic Synthesis of Quinazolinones for Commercial Scale-up
Introduction to Green Photocatalytic Innovation
The pharmaceutical and fine chemical industries are constantly seeking sustainable methodologies that align with green chemistry principles without compromising yield or purity. Patent CN111777564B introduces a groundbreaking approach for the synthesis of quinazolinone compounds, a privileged scaffold ubiquitous in drug discovery and agrochemical applications. This technology leverages visible light photocatalysis in an aqueous phase, marking a significant departure from traditional thermal methods that rely on hazardous oxidants. By utilizing potassium iodide (KI) as a cost-effective catalyst and water as the sole solvent, this process addresses critical pain points regarding environmental safety and operational complexity. For R&D directors and procurement specialists, this represents a viable pathway to producing high-purity pharmaceutical intermediates with a drastically reduced ecological footprint. The method operates under mild conditions, typically at room temperature, which preserves sensitive functional groups and minimizes energy consumption. This report analyzes the technical merits and commercial viability of this aqueous photocatalytic strategy, highlighting its potential to redefine supply chain standards for heterocyclic compound manufacturing.
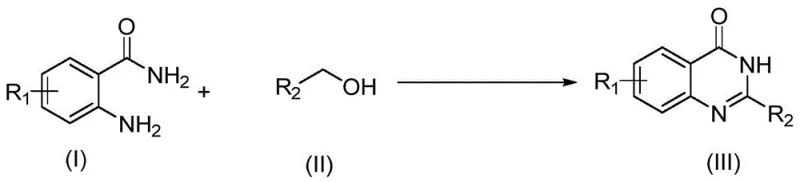
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied heavily on the condensation of anthranilamides with aldehydes or ketones, often necessitating the use of stoichiometric amounts of strong oxidizing agents such as potassium permanganate, manganese dioxide, or DDQ. These traditional protocols frequently require elevated temperatures and toxic organic solvents like DMF or DMSO to drive the reaction to completion. Such harsh conditions pose significant challenges for industrial scale-up, including high energy costs, difficult product isolation, and the generation of substantial hazardous waste streams. Furthermore, the use of heavy metal oxidants can lead to residual metal contamination in the final API intermediate, necessitating expensive and time-consuming purification steps to meet stringent regulatory limits. The reliance on pre-functionalized aldehydes also adds a synthetic step, increasing the overall cost of goods and extending the production timeline. These factors collectively hinder the efficiency and sustainability of conventional quinazolinone manufacturing processes.
The Novel Approach
In stark contrast, the methodology disclosed in CN111777564B utilizes readily available alcohols as direct coupling partners, bypassing the need for pre-oxidation to aldehydes. The reaction proceeds through a tandem oxidation-cyclization sequence facilitated by visible light irradiation, typically using a 40W LED source. This photochemical activation allows the reaction to proceed efficiently at ambient temperatures ranging from 20°C to 30°C, eliminating the thermal stress on substrates. The use of water as the reaction medium not only enhances safety by removing flammable organic solvents but also simplifies the downstream processing, as many organic products precipitate out or can be easily extracted. This novel approach exemplifies atom economy and step efficiency, transforming a multi-step, waste-intensive process into a streamlined, one-pot transformation. The compatibility with a wide range of substrates, including those with electron-withdrawing and electron-donating groups, underscores the robustness of this new synthetic platform for diverse chemical libraries.
Mechanistic Insights into KI-Catalyzed Photocyclization
The core of this innovation lies in the unique role of potassium iodide under visible light irradiation. Unlike traditional transition metal photocatalysts that operate via complex ligand-to-metal charge transfer mechanisms, the iodide ion acts as a simple yet effective single-electron transfer agent. Upon absorption of visible light photons, the iodide species generates reactive radical intermediates capable of abstracting hydrogen atoms from the alpha-position of the alcohol substrate. This initiates the oxidation of the alcohol to the corresponding aldehyde in situ, which then immediately undergoes nucleophilic attack by the amino group of the anthranilamide. The subsequent oxidative cyclization completes the formation of the quinazolinone ring system. This mechanism avoids the formation of stable metal-substrate complexes that often lead to catalyst deactivation or difficult removal. The mild nature of the radical generation ensures that sensitive functional groups, such as nitro or halo substituents, remain intact throughout the reaction, thereby preserving the structural integrity required for downstream biological activity.
Impurity control is inherently superior in this aqueous photocatalytic system due to the high selectivity of the light-driven process. In thermal oxidations, over-oxidation to carboxylic acids or polymerization of aldehyde intermediates are common side reactions that degrade yield and complicate purification. However, the controlled flux of photons in the LED reactor regulates the rate of radical formation, preventing runaway oxidation. Additionally, the aqueous environment helps to solubilize inorganic byproducts while keeping the organic quinazolinone product distinct, facilitating cleaner separation. The absence of transition metals means there is no risk of metal-catalyzed decomposition pathways that often plague long reaction times. This results in a cleaner crude reaction profile, reducing the burden on purification teams and ensuring that the final material meets the rigorous purity specifications demanded by global pharmaceutical regulators.
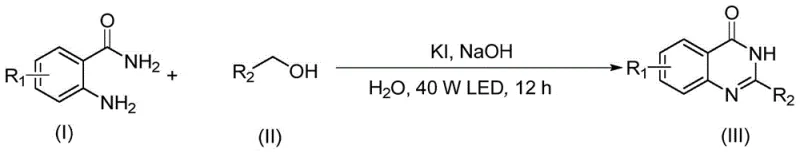
How to Synthesize Quinazolinones Efficiently
Implementing this photocatalytic protocol requires minimal specialized equipment, making it highly accessible for both laboratory research and pilot plant operations. The standard procedure involves charging a reaction vessel with 2-aminobenzamide, the selected alcohol substrate, potassium iodide catalyst, and sodium hydroxide base in water. The mixture is then subjected to irradiation from a standard white LED lamp positioned at a fixed distance to ensure uniform light penetration. Reaction progress is conveniently monitored via TLC, with typical completion times around 12 hours at room temperature. This simplicity allows for easy parallelization in process development studies. For detailed operational parameters, stoichiometry, and workup procedures tailored to specific substrates, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining 2-aminobenzamide and the chosen alcohol substrate in water with KI catalyst and NaOH base.
- Irradiate the reaction mixture with a 40W white LED light source at room temperature (20-30°C) for approximately 12 hours.
- Upon completion, purify the crude product via column chromatography to isolate the high-purity quinazolinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers transformative benefits for cost reduction in pharmaceutical intermediates manufacturing. The substitution of expensive noble metal catalysts with commodity-grade potassium iodide represents a direct and substantial decrease in raw material costs. Moreover, the elimination of volatile organic compounds (VOCs) as solvents drastically reduces expenditure on solvent purchase, recovery, and hazardous waste disposal. The mild reaction conditions also lower energy consumption by removing the need for heating mantles or oil baths, contributing to a leaner manufacturing overhead. These cumulative savings enhance the overall margin profile for quinazolinone-based projects, making them more attractive for large-scale production campaigns.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven by the use of earth-abundant catalysts and the avoidance of stoichiometric oxidants. Traditional methods often consume large quantities of reagents that end up as waste, whereas this catalytic cycle regenerates the active species. By removing the need for complex metal scavenging resins or extensive chromatography to remove heavy metals, the downstream processing costs are significantly minimized. This streamlined workflow translates to a lower cost per kilogram of the final API intermediate, providing a competitive edge in pricing negotiations with generic drug manufacturers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on universally available starting materials. Alcohols and anthranilamides are bulk chemicals with stable global supply chains, unlike specialized organometallic catalysts that may face sourcing bottlenecks. The robustness of the reaction against moisture and air further simplifies logistics, as strict anhydrous conditions are not required. This reduces the risk of batch failures due to environmental factors and ensures consistent delivery schedules. Manufacturers can maintain higher inventory turnover rates and respond more agilely to fluctuating market demands for quinazolinone derivatives.
- Scalability and Environmental Compliance: Scaling this photochemical process is straightforward due to the modular nature of LED lighting arrays, which can be easily expanded to accommodate larger reactor volumes without altering the reaction kinetics. The use of water as a solvent aligns perfectly with increasingly stringent environmental regulations regarding industrial effluent. Facilities adopting this technology can achieve better compliance scores and reduce their carbon footprint, which is a key metric for modern ESG (Environmental, Social, and Governance) reporting. This future-proofs the manufacturing asset against tightening regulatory landscapes and potential carbon taxes.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this photocatalytic technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing clarity for process chemists evaluating this route for adoption. Understanding these nuances is critical for assessing the feasibility of transferring this chemistry from bench scale to commercial production.
Q: What are the primary advantages of using water as a solvent in this synthesis?
A: Using water eliminates the need for toxic organic solvents, significantly reducing environmental impact and waste disposal costs while simplifying the workup procedure compared to traditional methods.
Q: Can this photocatalytic method tolerate diverse functional groups?
A: Yes, the method demonstrates excellent functional group tolerance, successfully accommodating substituents such as halogens, nitro groups, and alkoxy groups on both the benzamide and alcohol substrates.
Q: Is noble metal catalyst required for this oxidation process?
A: No, this innovative protocol utilizes inexpensive potassium iodide (KI) as the photocatalyst, avoiding the high costs and potential contamination issues associated with precious metal catalysts like Ruthenium or Iridium.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting green and efficient synthetic routes like the one described in CN111777564B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of handling complex heterocyclic syntheses with stringent purity specifications. We are committed to delivering high-purity quinazolinone intermediates that meet the exacting standards of the global pharmaceutical industry, leveraging our expertise to optimize yield and minimize impurities.
We invite you to collaborate with us to explore the full potential of this aqueous photocatalytic technology for your specific pipeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project's volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply chain for your critical drug candidates.
