Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Applications
Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Applications
The landscape of heterocyclic chemistry is constantly evolving, driven by the need for safer, more efficient, and environmentally benign synthetic routes. A significant breakthrough in this domain is documented in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. These structural motifs are ubiquitous in medicinal chemistry, serving as critical scaffolds in drug candidates targeting beta-3 adrenergic receptors and various agrochemical applications. The patent introduces a transformative approach that bypasses the traditional reliance on hazardous azides and expensive transition metal catalysts. By utilizing a base-promoted reaction between trifluoroethylimidoyl chloride and diazo compounds, this technology offers a streamlined pathway to high-purity intermediates. For R&D directors and procurement specialists, this represents a pivotal shift towards processes that enhance safety profiles while maintaining high reaction efficiency and broad substrate tolerance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has been fraught with significant safety and operational challenges. The literature predominantly describes two main strategies, both of which carry inherent risks that complicate large-scale manufacturing. The first involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation. The second relies on organocatalytic 1,3-dipolar cycloaddition involving azides and trifluoromethyl ketones. As illustrated in the general reaction schemes below, these conventional pathways invariably require the handling of organic azides, which are notorious for their toxicity and potential explosiveness. Furthermore, the use of copper catalysts introduces the burden of residual heavy metal removal, a critical quality control hurdle for pharmaceutical intermediates that can drastically increase production costs and lead times.
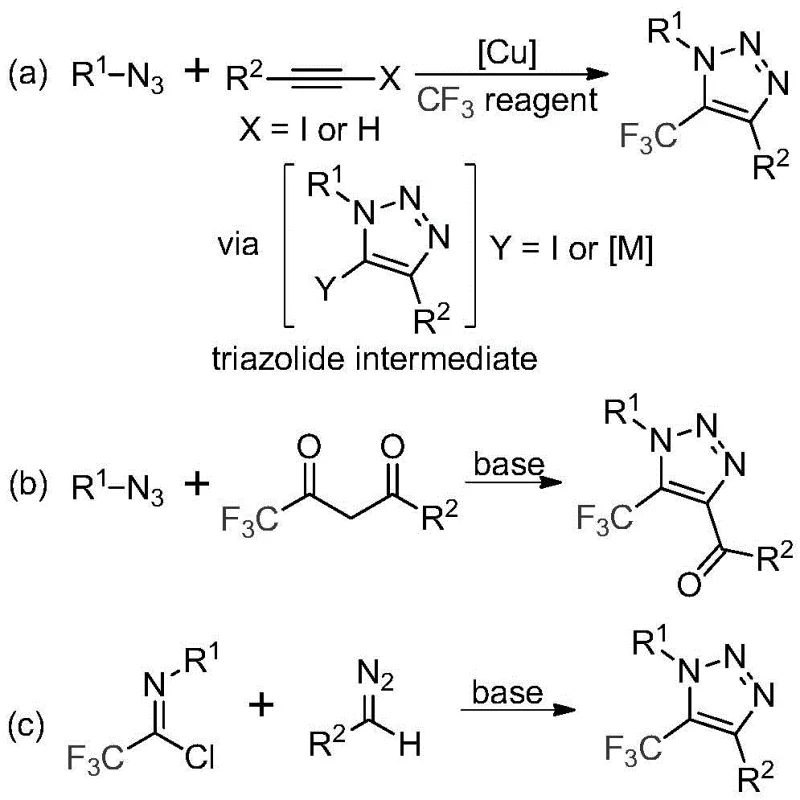
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN113121462B presents a metal-free and azide-free alternative that fundamentally reshapes the synthetic strategy. The core innovation lies in the direct coupling of trifluoroethylimidoyl chloride with diazo compounds under mild basic conditions. This reaction proceeds efficiently without the need for transition metals or dangerous azide reagents. As depicted in the specific reaction scheme for this patent, the process utilizes cesium carbonate as a promoter in acetonitrile, operating at moderate temperatures between 50°C and 70°C. This not only mitigates safety risks associated with explosive precursors but also simplifies the downstream purification process by eliminating the need for complex metal scavenging steps. The result is a robust, operationally simple protocol that delivers the desired 5-trifluoromethyl-1,2,3-triazole core with excellent functional group compatibility.
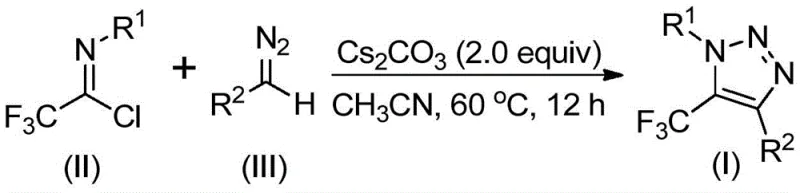
Mechanistic Insights into Base-Promoted Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific derivatives. The reaction is believed to initiate with a base-promoted intermolecular nucleophilic addition-elimination sequence. Specifically, the diazo compound acts as a nucleophile, attacking the electrophilic carbon of the trifluoroethylimidoyl chloride. This step facilitates the formation of a critical carbon-carbon bond while eliminating a chloride ion. Following this initial coupling, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This cyclization event closes the triazole ring, establishing the characteristic five-membered nitrogen-containing heterocycle. The presence of the trifluoromethyl group at the 5-position is retained throughout this sequence, ensuring the final product possesses the desired electronic and metabolic properties imparted by the fluorine atoms.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based trifluoromethylation methods. Because the trifluoromethyl group is pre-installed on the imidoyl chloride starting material rather than introduced via a reactive radical species, the formation of random trifluoromethylated byproducts is significantly minimized. The use of cesium carbonate, a mild inorganic base, further ensures that sensitive functional groups on the aromatic rings of R1 and R2 remain intact. This high level of chemoselectivity is vital for synthesizing complex pharmaceutical intermediates where protecting group strategies can add unnecessary steps and cost. The reaction conditions allow for the tolerance of halogens, alkoxy groups, and even phospholipid moieties, expanding the chemical space accessible to process chemists.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and purity. The patent outlines a straightforward procedure where cesium carbonate, trifluoroethylimidoyl chloride, and the diazo compound are combined in an organic solvent such as acetonitrile. The molar ratio is optimized to ensure complete conversion, typically employing a slight excess of the diazo compound and base relative to the imidoyl chloride. The reaction mixture is then heated to a controlled temperature, optimally around 60°C, and maintained for a duration of 8 to 16 hours. Upon completion, the workup involves simple filtration to remove inorganic salts, followed by standard purification techniques like column chromatography. For detailed standardized operating procedures and specific stoichiometric ratios for various substrates, please refer to the guide below.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C (optimally 60°C) and stir for 8-16 hours.
- Filter the mixture, concentrate, and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic benefits beyond mere chemical elegance. The elimination of hazardous azides and heavy metal catalysts directly addresses two of the most persistent pain points in chemical sourcing: safety compliance and purification costs. By shifting to a metal-free protocol, manufacturers can avoid the expensive and time-consuming processes associated with removing trace copper residues to meet stringent pharmaceutical standards. Furthermore, the starting materials, including trifluoroethylimidoyl chloride and various diazo compounds, are described as cheap and readily available, suggesting a stable and cost-effective supply chain foundation. This stability is crucial for long-term project planning and risk mitigation in the volatile fine chemicals market.
- Cost Reduction in Manufacturing: The economic implications of switching to this metal-free methodology are profound. Traditional copper-catalyzed routes often necessitate the use of specialized ligands and rigorous metal scavenging resins, both of which add significant line items to the bill of materials. By utilizing inexpensive cesium carbonate as the promoter, the direct material costs are substantially lowered. Additionally, the simplified workup procedure, which avoids complex extraction protocols required for metal removal, reduces labor hours and solvent consumption. This cumulative effect leads to a drastic simplification of the manufacturing process, driving down the overall cost of goods sold (COGS) for these valuable triazole intermediates without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the regulatory restrictions surrounding explosive precursors like organic azides. The storage and transport of such materials require specialized facilities and permits, creating bottlenecks and potential delays. This new method circumvents these logistical hurdles entirely by employing stable, non-explosive reagents. The ability to source starting materials from general chemical suppliers rather than specialized hazardous material vendors enhances flexibility and reduces lead times. Moreover, the robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental controls, ensuring consistent output and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry principles, the environmental footprint of a synthesis route is a key decision factor. This process aligns well with sustainability goals by avoiding toxic heavy metals and reducing the generation of hazardous waste streams. The patent explicitly notes that the method can be easily extended to the gram level and potentially industrial scale, indicating strong scalability. The use of common solvents like acetonitrile, which can be readily recovered and recycled, further minimizes environmental impact. For companies aiming to reduce their carbon footprint and adhere to increasingly strict environmental regulations, adopting this cleaner technology provides a competitive advantage in the marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation. They are designed to provide clarity on the practical aspects of adopting this route for your specific development projects. Understanding these nuances is essential for making informed decisions about process integration and resource allocation.
Q: Why is this metal-free method safer than traditional triazole synthesis?
A: Traditional methods often rely on toxic and explosive organic azides or heavy metal catalysts like copper. This novel route utilizes stable diazo compounds and imidoyl chlorides, eliminating explosion risks and heavy metal contamination concerns.
Q: What is the substrate scope for R1 and R2 groups?
A: The method demonstrates broad tolerance. R1 can be alkyl, substituted or unsubstituted aryl (e.g., phenyl, 4-Me-Ph, 4-Cl-Ph). R2 accommodates aroyl, phospholipids, alkoxycarbonyl, or trifluoromethyl groups, allowing for diverse functionalization.
Q: Is this process suitable for industrial scale-up?
A: Yes, the patent explicitly states the method can be easily extended to the gram level and potentially industrial scale. It uses cheap reagents like cesium carbonate and common solvents like acetonitrile, facilitating cost-effective manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to accelerate drug discovery and development. Our team of expert chemists has thoroughly analyzed the potential of the metal-free route described in CN113121462B and is fully equipped to leverage this methodology for your custom synthesis needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. Our state-of-the-art facilities are supported by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole intermediate meets the highest global standards for pharmaceutical applications.
We invite you to collaborate with us to explore how this innovative chemistry can optimize your supply chain and reduce your overall project costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data for our existing inventory or to discuss route feasibility assessments for your novel targets. Let us be your partner in turning complex chemical challenges into commercial successes.
