Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Production: A Safe, Scalable, and Metal-Free Route for Global Pharma
Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Production: A Safe, Scalable, and Metal-Free Route for Global Pharma
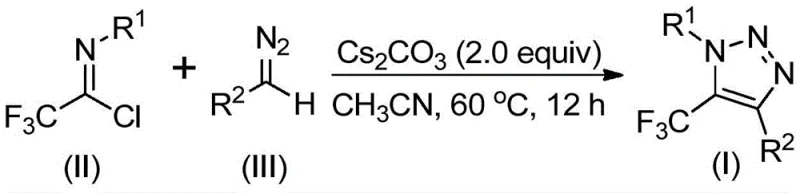
The landscape of heterocyclic chemistry is constantly evolving, driven by the urgent need for safer, more efficient, and environmentally benign synthetic methodologies. A significant breakthrough in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. These structural motifs are ubiquitous in modern medicinal chemistry, serving as critical scaffolds in drug candidates targeting various biological receptors, including potent beta-3 adrenergic agonists. The patent introduces a transformative approach that bypasses the traditional reliance on hazardous azides and expensive transition metal catalysts. Instead, it leverages a base-promoted cyclization between readily available trifluoroethylimidoyl chlorides and diazo compounds. This strategic shift not only enhances laboratory safety profiles but also streamlines the manufacturing workflow, offering a compelling value proposition for R&D directors and procurement managers seeking reliable pharmaceutical intermediate suppliers who prioritize both quality and operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has been dominated by two primary strategies, both of which carry significant drawbacks for large-scale industrial application. The first involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation. This pathway is fraught with peril due to the inherent instability and explosive nature of organic azides, posing severe safety risks during storage and handling. Furthermore, the reliance on copper catalysts necessitates rigorous downstream purification to meet stringent regulatory limits on heavy metal residues in active pharmaceutical ingredients (APIs). The second conventional route utilizes organocatalytic 1,3-dipolar cycloaddition involving azides and trifluoromethyl ketones, which similarly suffers from the safety liabilities associated with azide chemistry. These legacy methods often require harsh conditions, specialized equipment for hazard mitigation, and complex workup procedures, all of which contribute to inflated production costs and extended lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these hazardous legacy protocols, the methodology outlined in CN113121462B offers a streamlined, metal-free alternative that fundamentally alters the risk-reward profile of triazole synthesis. By utilizing trifluoroethylimidoyl chlorides and diazo compounds as starting materials, the process completely eliminates the need for explosive azides and toxic transition metals. The reaction proceeds smoothly under mild thermal conditions, typically between 50°C and 70°C, using cesium carbonate as a benign base promoter. This approach not only mitigates safety concerns but also simplifies the reaction setup, allowing for easier operation and application in standard chemical manufacturing facilities. The versatility of this method is demonstrated by its tolerance for a wide range of functional groups on both the imidoyl chloride and the diazo components, enabling the synthesis of diverse derivatives with high efficiency. This represents a paradigm shift towards greener, safer, and more cost-effective manufacturing practices.
Mechanistic Insights into Base-Promoted Cyclization
From a mechanistic perspective, this transformation is a fascinating example of how simple reagents can orchestrate complex bond formations without the aid of transition metals. The reaction is hypothesized to initiate with a base-promoted intermolecular nucleophilic addition-elimination process. Specifically, the diazo compound, activated by the basic environment provided by cesium carbonate, attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step facilitates the formation of a crucial carbon-carbon bond while eliminating a chloride ion. Following this initial coupling, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This cyclization step is critical for closing the five-membered triazole ring, ultimately yielding the stable 5-trifluoromethyl-1,2,3-triazole scaffold. The elegance of this mechanism lies in its atom economy and the avoidance of radical pathways that often lead to complex impurity profiles in metal-catalyzed systems.
Furthermore, the choice of cesium carbonate plays a pivotal role in controlling the reaction trajectory and minimizing impurities. As a mild yet effective base, it deprotonates the diazo species sufficiently to enable nucleophilic attack without causing excessive decomposition of the sensitive diazo functionality. The use of aprotic solvents like acetonitrile further stabilizes the ionic intermediates and ensures high conversion rates. This controlled environment significantly reduces the formation of side products, resulting in a cleaner crude reaction mixture. For R&D teams, this means less time spent on optimizing purification protocols and more time focusing on biological evaluation. The ability to fine-tune the electronic properties of the starting materials—by varying substituents on the aryl rings of either the imidoyl chloride or the diazo compound—allows for precise modulation of the final product's physicochemical properties, a key requirement in lead optimization campaigns.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
The practical implementation of this synthesis is remarkably straightforward, making it accessible for both laboratory discovery and pilot-scale production. The protocol involves combining the key starting materials—trifluoroethylimidoyl chloride and the specific diazo compound—with cesium carbonate and 4Å molecular sieves in an organic solvent. The molecular sieves serve to scavenge trace moisture, which is critical for maintaining the stability of the reagents and maximizing yield. The mixture is then heated to a moderate temperature, typically around 60°C, and stirred for a period ranging from 8 to 16 hours. This duration ensures complete consumption of the starting materials while preventing thermal degradation of the product. Upon completion, the workup is minimal: filtration to remove solids, followed by standard silica gel chromatography. This simplicity underscores the method's potential for rapid adoption in process chemistry workflows.
- Prepare the reaction mixture by adding cesium carbonate, molecular sieves, trifluoroethylimidoyl chloride, and the diazo compound into an aprotic organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature range of 50-70°C and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 5-trifluoromethyl-1,2,3-triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic benefits that extend far beyond the laboratory bench. The elimination of hazardous azides removes a major bottleneck in chemical logistics, as it negates the need for specialized storage facilities and expensive safety protocols associated with explosive precursors. This inherently safer chemistry reduces insurance premiums and operational overheads, contributing to substantial cost savings in pharmaceutical intermediate manufacturing. Moreover, the reliance on commercially available, off-the-shelf starting materials like aromatic amines and simple diazo precursors ensures a robust and resilient supply chain. Unlike specialized catalysts that may suffer from supply disruptions or long lead times, the reagents for this process are commodity chemicals, guaranteeing supply continuity even in volatile market conditions.
- Cost Reduction in Manufacturing: The economic implications of switching to this metal-free protocol are profound. By removing the necessity for expensive transition metal catalysts such as copper or palladium, manufacturers can drastically reduce raw material costs. More importantly, the absence of heavy metals simplifies the purification process, eliminating the need for costly metal scavenging resins and extensive analytical testing for residual metals. This streamlined downstream processing reduces solvent consumption and waste generation, leading to a significantly lower cost of goods sold (COGS). The high reaction efficiency and yields reported in the patent further enhance the economic viability, ensuring that raw materials are converted into valuable products with minimal waste.
- Enhanced Supply Chain Reliability: Supply chain resilience is paramount in the pharmaceutical industry, where delays can halt clinical trials or commercial launches. This synthesis method bolsters reliability by utilizing starting materials that are widely produced and easily sourced from multiple global vendors. The robustness of the reaction conditions—tolerating a broad scope of substrates and functioning effectively in common solvents like acetonitrile—means that production is less susceptible to variations in reagent quality or minor fluctuations in process parameters. This flexibility allows for agile manufacturing responses to changing demand, reducing lead time for high-purity pharmaceutical intermediates and ensuring that critical drug development timelines are met without compromise.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry, this method aligns perfectly with environmental, social, and governance (ESG) goals. The process generates less hazardous waste compared to azide-based routes and avoids the environmental burden of mining and refining transition metals. The scalability of the reaction, demonstrated from milligram to gram scales in the patent data, suggests a smooth path to kilogram and ton-scale production. The mild conditions reduce energy consumption, and the simplified workup minimizes the volume of organic solvents required for purification. These factors collectively ease the regulatory burden for environmental compliance, facilitating faster approval for commercial scale-up of complex pharmaceutical intermediates and supporting sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this innovative synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical implementation and benefits of the process. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this method into their existing production pipelines or sourcing strategies for next-generation drug candidates.
Q: Why is this new synthesis method safer than traditional copper-catalyzed routes?
A: Traditional methods often rely on toxic and potentially explosive organic azides and transition metal catalysts. This novel protocol utilizes stable diazo compounds and imidoyl chlorides under metal-free conditions, significantly reducing safety hazards and eliminating the need for expensive heavy metal removal steps.
Q: What are the key commercial advantages of using cesium carbonate in this reaction?
A: Cesium carbonate acts as an efficient, inexpensive base promoter that facilitates the reaction under mild temperatures (50-70°C). Its use avoids the complexity of ligand optimization required in metal catalysis and simplifies the downstream purification process, leading to lower operational costs.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly demonstrates that the method is easily scalable to gram levels and beyond. The use of commercially available starting materials, mild reaction conditions, and simple workup procedures makes it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in CN113121462B for advancing drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality supply regardless of project phase. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole intermediate adheres to the highest international standards. We are committed to leveraging our technical expertise to optimize this metal-free route, delivering cost-effective solutions that accelerate your time to market.
We invite you to collaborate with us to explore how this advanced chemistry can enhance your portfolio. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating exactly how this route can improve your margins. Please contact our technical procurement team today to request specific COA data for our available triazole derivatives and to discuss detailed route feasibility assessments. Let us be your partner in turning complex chemical challenges into commercial successes.
