Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Scalable Pharmaceutical Production
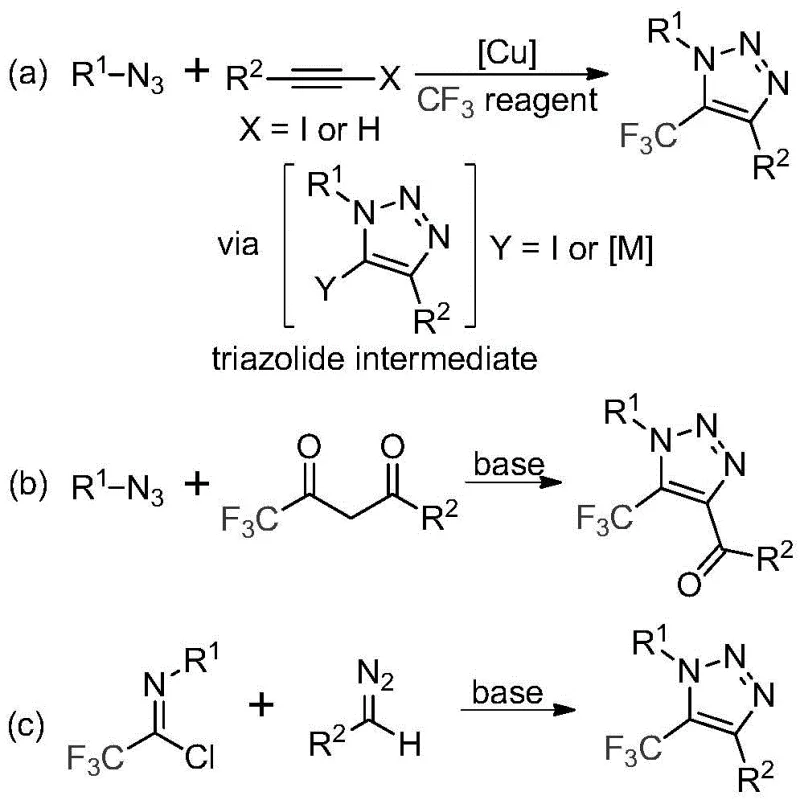
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to incorporate trifluoromethyl groups into heterocyclic scaffolds, driven by the profound impact of fluorine on metabolic stability and lipophilicity. Patent CN113121462B introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds that fundamentally shifts the paradigm away from hazardous and complex traditional routes. This technology leverages a base-promoted cyclization strategy that bypasses the need for toxic transition metal catalysts and explosive organic azides, offering a safer and more economically viable pathway for producing high-purity pharmaceutical intermediates. By utilizing readily available trifluoroethylimidoyl chlorides and diazo compounds, this invention addresses critical pain points in process chemistry, specifically regarding safety protocols and waste management. The ability to synthesize these valuable heterocycles under mild conditions represents a significant leap forward for process development teams aiming to streamline their supply chains for complex drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed azide-alkyne cycloaddition (CuAAC) reactions or organocatalytic variants involving trifluoromethyl ketones. These conventional pathways present severe logistical and safety challenges for large-scale manufacturing, primarily due to the inherent instability and explosive nature of organic azides required as starting materials. Furthermore, the reliance on copper catalysts necessitates rigorous downstream purification steps to remove trace heavy metals, which is a critical regulatory requirement for active pharmaceutical ingredients (APIs). The presence of residual copper can catalyze degradation pathways in the final drug product, leading to stability issues and potential toxicity concerns. Additionally, the synthesis of specific trifluoromethyl reagents often involves multi-step sequences with poor atom economy, driving up the cost of goods and generating significant chemical waste that complicates environmental compliance.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN113121462B utilizes a metal-free, base-promoted reaction between trifluoroethylimidoyl chlorides and diazo compounds to construct the triazole ring efficiently. This innovative approach completely eliminates the need for handling hazardous organic azides, thereby drastically improving the safety profile of the manufacturing process and reducing the need for specialized explosion-proof infrastructure. The reaction proceeds smoothly in common aprotic solvents like acetonitrile at moderate temperatures, facilitating easy scale-up from gram to kilogram quantities without compromising yield or purity. By avoiding transition metals entirely, the process simplifies the workup procedure, removing the costly and time-consuming steps associated with metal scavenging and chelation. This streamlined workflow not only accelerates the timeline from laboratory discovery to commercial production but also aligns perfectly with green chemistry principles by minimizing waste and energy consumption.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation involves a sophisticated sequence of nucleophilic attacks and cyclization events that are carefully orchestrated by the choice of base and solvent. The reaction initiates with the base-promoted intermolecular nucleophilic addition of the diazo compound to the electrophilic carbon of the trifluoroethylimidoyl chloride. This step is critical as it establishes the initial carbon-carbon bond connectivity required for ring closure, driven by the strong electron-withdrawing nature of the trifluoromethyl group which activates the imidoyl chloride towards nucleophilic attack. Following this addition-elimination process, the intermediate undergoes an intramolecular 5-endo-dig cyclization, a kinetically favorable pathway that rapidly forms the five-membered triazole ring system. The use of cesium carbonate as the promoter is particularly advantageous due to its optimal basicity and solubility profile in organic media, which ensures efficient deprotonation without causing excessive decomposition of the sensitive diazo species.
From an impurity control perspective, the mild reaction conditions ranging from 50°C to 70°C play a pivotal role in maintaining high product fidelity. Harsh thermal conditions often lead to the decomposition of diazo compounds into carbene species, which can result in non-specific insertion reactions and complex byproduct mixtures that are difficult to separate. By operating within this温和 temperature window, the process minimizes side reactions such as dimerization of the diazo compound or hydrolysis of the imidoyl chloride. Furthermore, the selection of acetonitrile as the preferred solvent enhances the solubility of both the inorganic base and the organic substrates, creating a homogeneous reaction environment that promotes consistent kinetics. This precise control over reaction parameters ensures that the final 5-trifluoromethyl-1,2,3-triazole products are obtained with high purity, reducing the burden on downstream purification units and maximizing overall process efficiency.
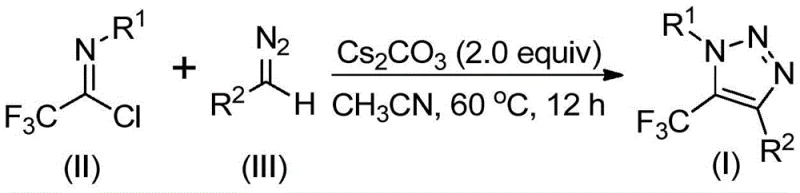
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, making it highly accessible for process chemists aiming to replicate these results in a pilot or production setting. The protocol involves charging a reactor with cesium carbonate, the specific trifluoroethylimidoyl chloride derivative, and the corresponding diazo compound in a measured volume of dry acetonitrile. The mixture is then heated to a controlled temperature of 60°C and maintained under stirring for approximately 12 hours to ensure full conversion of the starting materials. Upon completion, the reaction mixture is cooled, filtered to remove inorganic salts, and the filtrate is concentrated before undergoing standard silica gel chromatography to isolate the pure triazole product. For detailed standardized synthetic steps and specific stoichiometric ratios optimized for different substrates, please refer to the guide below.
- Combine cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis technology offers transformative benefits that extend far beyond simple yield improvements. The elimination of copper catalysts removes a significant cost center associated with expensive metal salts and the specialized scavenger resins required for their removal, directly contributing to a lower cost of goods sold (COGS). Moreover, the avoidance of organic azides mitigates the need for costly safety engineering controls and insurance premiums associated with handling energetic materials, further enhancing the economic attractiveness of this route. The use of commodity chemicals like cesium carbonate and acetonitrile ensures a stable and reliable supply chain, insulating manufacturers from the volatility often seen with specialized organometallic reagents. This robustness allows for more accurate forecasting and inventory management, reducing the risk of production delays caused by raw material shortages.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts eliminates the need for expensive metal scavengers and activated carbon treatment steps, which are standard requirements in copper-catalyzed processes to meet strict regulatory limits for heavy metals in APIs. This simplification of the downstream processing workflow significantly reduces the consumption of auxiliary materials and shortens the overall batch cycle time. Additionally, the high atom economy of the reaction means that less raw material is wasted as byproducts, leading to substantial savings in raw material procurement costs over the lifecycle of the product. The ability to run the reaction at moderate temperatures also lowers energy consumption compared to high-temperature reflux conditions often required for less reactive substrates.
- Enhanced Supply Chain Reliability: The starting materials for this process, specifically trifluoroethylimidoyl chlorides and diazo compounds, are derived from widely available industrial feedstocks, ensuring a consistent and secure supply source. Unlike specialized azide reagents which may have limited suppliers and long lead times, these precursors can be sourced from multiple global vendors, reducing dependency on single-source suppliers. The stability of these reagents allows for bulk purchasing and long-term storage without significant degradation, enabling companies to take advantage of economies of scale. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, having been validated from small-scale laboratory experiments to gram-level preparations without loss of efficiency, indicating a clear path to multi-kilogram and ton-scale production. The absence of heavy metals simplifies waste stream management, as the effluent does not require complex treatment for metal removal before discharge, aligning with increasingly stringent environmental regulations. The use of acetonitrile, a solvent with well-established recovery and recycling protocols, further supports sustainability goals by minimizing solvent waste. This environmentally friendly profile enhances the corporate social responsibility standing of the manufacturer and facilitates easier regulatory approval in markets with strict green chemistry mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the detailed data provided in the patent documentation. These insights are intended to clarify the operational advantages and scope of applicability for potential licensees or manufacturing partners. Understanding these nuances is essential for evaluating the fit of this technology within existing production portfolios and R&D pipelines.
Q: Does this synthesis method require hazardous organic azides?
A: No, unlike traditional CuAAC methods, this patent describes a route using stable diazo compounds and imidoyl chlorides, eliminating the safety risks associated with explosive organic azides.
Q: Is transition metal removal necessary for the final product?
A: No, the process is metal-free and uses cesium carbonate as a promoter, which significantly simplifies downstream purification and avoids heavy metal contamination issues common in copper-catalyzed reactions.
Q: What is the substrate scope for R1 and R2 groups?
A: The method tolerates a wide range of substituents including alkyl, substituted aryl, aroyl, phospholipids, and alkoxycarbonyl groups, allowing for diverse functionalization of the triazole core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in handling fluorinated compounds and heterocyclic chemistry positions us as an ideal partner for companies seeking to leverage this metal-free triazole synthesis technology for their drug development programs.
We invite you to engage with our technical procurement team to discuss how this novel route can be integrated into your supply chain to achieve significant operational efficiencies. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and accelerate your time to market.
