Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazole Compounds for Commercial Scale-up
Introduction to Patent CN111423381B: A Breakthrough in Heterocyclic Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups, due to their profound impact on drug pharmacokinetics. Patent CN111423381B discloses a highly efficient preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing critical challenges in modern medicinal chemistry. This technology leverages a sophisticated palladium-catalyzed multicomponent reaction that integrates trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts into a single synthetic operation. The introduction of the trifluoromethyl moiety significantly enhances the electronegativity, metabolic stability, and lipophilicity of the parent molecule, making these intermediates invaluable for the development of next-generation active pharmaceutical ingredients (APIs). By operating under remarkably mild conditions at 30°C, this process not only ensures high reaction efficiency but also minimizes energy consumption, presenting a compelling value proposition for both research and development teams and large-scale manufacturing facilities seeking reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of trifluoromethyl-substituted nitrogen heterocycles has been fraught with significant operational hazards and economic inefficiencies. Conventional routes often rely on direct trifluoromethylation using synthons such as trifluorodiazoethane, which is notoriously unstable, explosive, and difficult to handle safely on a large scale. Furthermore, existing methods frequently require harsh reaction conditions, including elevated temperatures and pressures, which can lead to poor substrate tolerance and the formation of complex impurity profiles that are costly to remove. The reliance on specialized, high-pressure equipment for carbon monoxide insertion further complicates the process engineering, creating bottlenecks in cost reduction in API manufacturing. These limitations have historically restricted the widespread adoption of trifluoromethyl-imidazole scaffolds in commercial drug synthesis, necessitating a safer and more versatile alternative that can accommodate diverse functional groups without compromising yield or purity.
The Novel Approach
The methodology described in patent CN111423381B represents a paradigm shift by utilizing cheap and readily available starting materials to construct the target scaffold through a transition metal-catalyzed cascade. Instead of hazardous gases, the process employs trifluoroethylimidoyl chloride and diaryl iodonium salts, which are stable solids that can be easily weighed and handled in standard reactor vessels. The reaction proceeds smoothly in common organic solvents like tetrahydrofuran (THF) at a constant temperature of 30°C, eliminating the need for energy-intensive heating or cooling cycles. This approach not only simplifies the operational workflow but also dramatically expands the substrate scope, allowing for the synthesis of diversified imidazole derivatives with various substituents at the 1 and 5 positions. 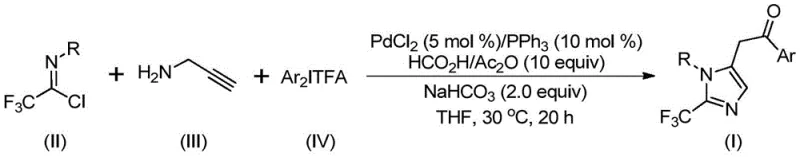
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The core of this innovative synthesis lies in a complex yet elegant catalytic cycle driven by palladium chloride and triphenylphosphine. The mechanism initiates with the formation of an intermolecular carbon-nitrogen bond promoted by the alkaline additive, sodium bicarbonate, yielding a trifluoroacetamidine intermediate. This species undergoes isomerization followed by palladation of the alkyne moiety from the propargylamine, generating a crucial alkenyl palladium intermediate. Subsequent isomerization converts this into an alkyl palladium species, setting the stage for the pivotal carbonylation step. Uniquely, the carbon monoxide required for this transformation is generated in situ from the decomposition of formic acid and acetic anhydride, thereby avoiding the logistical nightmares associated with storing and handling high-pressure CO gas. The resulting acyl palladium intermediate then undergoes oxidative addition with the diaryl iodonium salt to form a tetravalent palladium complex, which finally collapses via reductive elimination to release the desired 2-trifluoromethyl-substituted imidazole product and regenerate the active catalyst.
From a quality control perspective, this mechanistic pathway offers superior impurity management compared to radical-based trifluoromethylation methods. The stepwise nature of the palladium cycle ensures high regioselectivity, minimizing the formation of structural isomers that often plague heterocyclic synthesis. The use of mild conditions prevents thermal degradation of sensitive functional groups, such as nitro or halogen substituents, which might otherwise decompose under harsher protocols. This high level of chemoselectivity translates directly into a cleaner crude reaction mixture, reducing the burden on downstream purification processes like column chromatography. For R&D directors, understanding this mechanism is vital for troubleshooting and optimizing the process for specific substrates, ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications for residual metals and organic impurities.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
To implement this synthesis effectively, precise control over stoichiometry and reaction parameters is essential. The patent outlines a robust protocol where palladium chloride (5 mol%) and triphenylphosphine (10 mol%) serve as the catalytic system, balanced by sodium bicarbonate as a base. The reaction mixture typically includes acetic anhydride and formic acid as the CO source, combined with the three primary building blocks in a THF solvent system. While the general procedure is straightforward, scaling this reaction requires attention to mixing efficiency and temperature uniformity to maintain the high yields observed in the laboratory examples, which range from 65% to an impressive 97%. The detailed standardized synthesis steps, including specific workup procedures and purification guidelines, are provided below to assist technical teams in replicating these results.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture.
- Stir the reaction at 30°C for 16-24 hours, then filter and purify via column chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this patented technology are substantial, primarily driven by the accessibility and cost profile of the raw materials. Unlike specialized fluorinating agents that command premium prices and suffer from volatile supply chains, the key precursors—trifluoroethylimidoyl chloride and diaryl iodonium salts—are derived from abundant aromatic amines and boronic acids. This reliance on commodity chemicals significantly de-risks the supply chain, ensuring continuity of supply even during market fluctuations. Furthermore, the elimination of high-pressure equipment and hazardous gas handling reduces the capital expenditure (CAPEX) required for facility upgrades, allowing manufacturers to utilize existing infrastructure for production. The mild reaction temperature of 30°C also contributes to lower operational expenditures (OPEX) by minimizing energy consumption for heating or cryogenic cooling, aligning with modern sustainability goals.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by replacing expensive and dangerous trifluoromethyl sources with stable, low-cost alternatives. The use of a simple palladium chloride catalyst system, rather than exotic ligands or high-loading noble metals, further drives down the cost of goods sold (COGS). Additionally, the high atom economy and reduced need for extensive purification steps due to cleaner reaction profiles contribute to overall manufacturing efficiency.
- Enhanced Supply Chain Reliability: By utilizing starting materials that are widely available in the global chemical market, this method mitigates the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failure or strict safety shutdowns associated with high-risk chemistry. This reliability is crucial for maintaining consistent inventory levels of critical API intermediates.
- Scalability and Environmental Compliance: The protocol has been demonstrated to work effectively on the gram scale with potential for expansion to multi-kilogram batches. The avoidance of toxic gases and the use of standard organic solvents simplify waste treatment and environmental compliance, facilitating smoother regulatory approvals for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction scope, safety, and scalability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this technology into their existing production pipelines.
Q: What are the key advantages of this Pd-catalyzed method over traditional trifluoromethylation?
A: This method avoids hazardous reagents like trifluorodiazoethane, utilizing stable and cheap trifluoroethylimidoyl chlorides instead. It operates under mild conditions (30°C) with excellent substrate compatibility.
Q: What is the role of formic acid and acetic anhydride in this reaction?
A: Formic acid and acetic anhydride act as a carbon monoxide (CO) surrogate system, generating CO in situ to facilitate the critical carbonylation step without requiring high-pressure gas cylinders.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent demonstrates gram-level synthesis with high yields (up to 97%) and uses commercially available catalysts and solvents, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient fluorination technologies in the development of modern therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering high-purity 2-trifluoromethyl imidazole compounds that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in palladium-catalyzed transformations allows us to optimize this specific patent technology for maximum yield and minimal impurity generation, providing our partners with a competitive edge in the marketplace.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient route. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to initiate a dialogue regarding route feasibility assessments for your proprietary molecules, ensuring your supply chain is built on a foundation of innovation and reliability.
