Advanced Pd-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Scalable Pharmaceutical Manufacturing
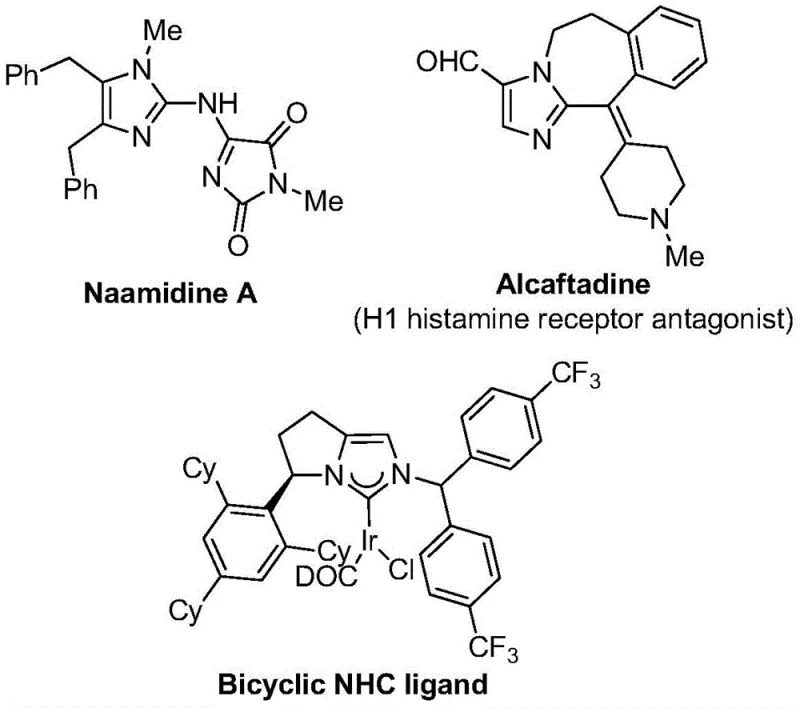
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds, particularly those incorporating fluorine atoms which are pivotal for modulating biological activity. A significant breakthrough in this domain is documented in Chinese Patent CN111423381A, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted imidazole compounds. This technology addresses critical challenges in modern medicinal chemistry by providing a streamlined pathway to access these valuable motifs under remarkably mild conditions. The imidazole core is a ubiquitous structure found in numerous bioactive molecules, ranging from antifungal agents to histamine receptor antagonists, as illustrated by the diverse molecular architectures shown in the reference data. By integrating a trifluoromethyl group directly onto this scaffold, chemists can drastically enhance the metabolic stability and lipophilicity of drug candidates, thereby improving their pharmacokinetic profiles. This patent represents a strategic advancement for any organization aiming to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity building blocks for next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with significant operational hazards and synthetic inefficiencies. Conventional literature often relies on the use of trifluorodiazoethane or similar high-energy synthons which pose severe safety risks due to their explosive nature and instability. Furthermore, many traditional protocols require harsh reaction conditions, including elevated temperatures and strong bases, which frequently lead to poor functional group tolerance and the generation of complex impurity profiles. These limitations not only increase the cost of goods sold through expensive safety mitigation measures but also complicate the purification process, resulting in lower overall yields. For procurement managers and supply chain heads, these factors translate into volatile pricing and unreliable delivery schedules for critical intermediates. The reliance on unstable reagents also restricts the scope of substrates that can be utilized, limiting the chemical diversity available for drug discovery campaigns and hindering the rapid iteration required in modern R&D pipelines.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a transition metal palladium-catalyzed carbonylation cascade reaction that operates with exceptional safety and efficiency. By employing cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts, the process eliminates the need for hazardous diazo compounds. The reaction proceeds smoothly at a mild temperature of 30°C, utilizing a formic acid and acetic anhydride mixture as a safe carbon monoxide surrogate. This shift in methodology offers profound advantages for cost reduction in pharmaceutical intermediate manufacturing, as it simplifies the reactor requirements and reduces energy consumption. Moreover, the method exhibits excellent substrate compatibility, allowing for the introduction of various substituents including alkyl, halogen, and nitro groups without compromising reaction efficiency. This flexibility enables the synthesis of a diverse library of 2-trifluoromethyl substituted imidazoles, providing R&D directors with a powerful tool for structure-activity relationship studies while ensuring that the supply chain remains robust and scalable.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The underlying mechanism of this transformation is a sophisticated multi-step catalytic cycle that elegantly constructs the imidazole ring while installing the trifluoromethyl group. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, generating a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization to facilitate the palladium-catalyzed alkynyl amination, forming a key alkenyl palladium intermediate. Through a series of isomerization steps, this evolves into an alkyl palladium species which is then subjected to carbonylation. The carbon monoxide required for this step is generated in situ from the decomposition of the formic acid and acetic anhydride mixture, avoiding the need for handling toxic CO gas cylinders. The resulting acyl palladium intermediate then undergoes oxidative addition with the diaryliodonium salt to form a high-valent tetravalent palladium species. Finally, a reductive elimination step releases the desired 2-trifluoromethyl substituted imidazole product and regenerates the active palladium catalyst, completing the cycle. This mechanistic understanding is crucial for process chemists aiming to optimize reaction parameters and ensure consistent quality in commercial production.
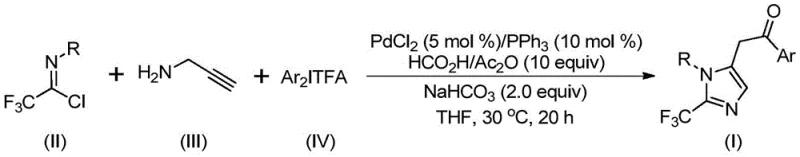
Controlling the impurity profile in such complex cascade reactions is paramount for meeting the stringent purity specifications required by regulatory bodies. The use of specific additives like sodium bicarbonate helps to neutralize acidic byproducts and maintain the optimal pH for the catalytic cycle, thereby minimizing side reactions such as hydrolysis of the imidoyl chloride. Furthermore, the choice of ligand, specifically triphenylphosphine in this protocol, plays a critical role in stabilizing the palladium center and facilitating the oxidative addition step with the diaryliodonium salt. By carefully tuning the molar ratios of the catalyst, ligand, and base, manufacturers can suppress the formation of homocoupling byproducts or incomplete cyclization intermediates. The patent data indicates that the reaction tolerates a wide range of electronic environments on the aryl rings, suggesting that the catalytic system is robust enough to handle electron-rich and electron-deficient substrates alike. This broad tolerance ensures that the final product stream is clean, reducing the burden on downstream purification units and enhancing the overall economic viability of the process for large-scale applications.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
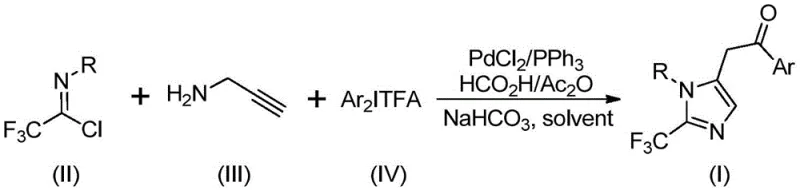
Implementing this synthesis route in a laboratory or pilot plant setting requires precise adherence to the optimized conditions described in the patent to achieve the reported high yields. The procedure involves charging a reaction vessel with the palladium catalyst system, the CO source, and the specific amine and salt precursors in an aprotic solvent such as tetrahydrofuran. The mild thermal requirements allow the reaction to proceed effectively at 30°C over a period of 16 to 24 hours, ensuring complete conversion of the starting materials. Detailed standardized operating procedures for this synthesis are provided below to guide technical teams in replicating these results with high fidelity.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction vessel under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers transformative benefits that extend far beyond simple chemical synthesis. The primary advantage lies in the substantial cost savings achieved through the utilization of commodity chemicals as starting materials. Trifluoroethylimidoyl chloride can be rapidly synthesized from inexpensive aromatic amines, while propargylamine and diaryliodonium salts are commercially available in bulk quantities. This reliance on abundant feedstocks decouples the production cost from the volatility associated with specialized or proprietary reagents, leading to a more stable pricing structure for the final intermediate. Additionally, the elimination of hazardous reagents like trifluorodiazoethane removes the need for expensive explosion-proof infrastructure and specialized waste disposal protocols, further driving down the operational expenditure. These factors combine to create a highly competitive cost position for manufacturers who adopt this technology, allowing them to offer more attractive pricing to their downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of high-cost, dangerous reagents with stable, low-cost alternatives. By utilizing a palladium catalyst system that operates at low loading levels and employs a safe in situ CO source, the process minimizes both material and utility costs. The mild reaction temperature of 30°C significantly reduces energy consumption compared to traditional high-temperature reflux methods, contributing to a lower carbon footprint and reduced utility bills. Furthermore, the high reaction efficiency and yields reported in the patent examples mean that less raw material is wasted, maximizing the atom economy of the process. These cumulative efficiencies result in a drastic simplification of the cost structure, enabling significant margin improvement for producers of these valuable heterocyclic intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source suppliers for exotic reagents, but this method mitigates that risk by using widely available starting materials. The starting aromatic amines and simple alkynes are produced by multiple global chemical manufacturers, ensuring that supply disruptions are unlikely to halt production. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the rate of batch failures and ensuring consistent output. For supply chain heads, this translates to reduced lead times for high-purity pharmaceutical intermediates and the ability to maintain safety stock levels with greater confidence. The scalability of the method, demonstrated by its successful expansion to the gram level in the patent, assures partners that the technology can seamlessly transition from pilot scale to multi-ton commercial production without unforeseen bottlenecks.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent, the ability to manufacture complex chemicals with minimal waste is a critical competitive advantage. This palladium-catalyzed route generates fewer hazardous byproducts compared to traditional diazo-based methods, simplifying the wastewater treatment process and reducing the volume of hazardous solid waste. The use of tetrahydrofuran as a solvent, which can be recovered and recycled, further aligns the process with green chemistry principles. The commercial scale-up of complex pharmaceutical intermediates using this method is facilitated by the absence of gas handling requirements for carbon monoxide, as the CO is generated safely from liquid precursors. This inherent safety feature allows for the use of standard glass-lined or stainless steel reactors, accelerating the timeline for technology transfer and commercial validation. Consequently, manufacturers can bring new products to market faster while maintaining full compliance with global environmental and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and strategic benefits for potential partners and technical stakeholders evaluating this route for their supply chains.
Q: What are the advantages of introducing a trifluoromethyl group into imidazole compounds?
A: Introducing a trifluoromethyl group significantly improves the electronegativity, bioavailability, metabolic stability, and lipophilicity of the parent molecule, making it highly valuable for drug design.
Q: Why is this Pd-catalyzed method superior to traditional trifluoromethylation techniques?
A: Unlike traditional methods that often rely on hazardous trifluorodiazoethane, this method uses stable trifluoroethylimidoyl chloride and operates under mild conditions (30°C) with high substrate compatibility.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and uses cheap, readily available starting materials, indicating strong potential for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert process chemists has extensively evaluated the technology described in CN111423381A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity 2-trifluoromethyl imidazole derivatives that meet the rigorous quality standards demanded by the global pharmaceutical industry. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of intermediate we produce is fully characterized and free from critical impurities. By leveraging our deep expertise in palladium catalysis and fluorine chemistry, we can guarantee a consistent and reliable supply of these essential building blocks for your most challenging projects.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your specific manufacturing needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and project timelines. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Together, we can drive efficiency and innovation in the production of next-generation therapeutic agents.
