Revolutionizing Quinazolinone Synthesis Iron-Catalyzed Route for High-Purity Pharmaceutical Intermediates at Commercial Scale
Patent CN111675662B discloses a novel preparation method for 2-trifluoromethyl-substituted quinazolinone compounds which represent critical nitrogen-containing heterocyclic scaffolds prevalent across numerous bioactive molecules including established pharmaceutical agents such as Aflaqualone Luotonin F Piriqualone Rutaecarpine TLN inhibitor and Sildenafil as demonstrated by structural examples in contemporary medicinal chemistry literature This innovative approach addresses longstanding challenges in synthesizing trifluoromethylated quinazolinones by leveraging an iron-catalyzed cyclization strategy that significantly enhances process efficiency scalability and purity profiles compared to conventional methodologies The method utilizes readily available starting materials such as trifluoroethylimidoyl chloride and isatin derivatives under precisely controlled mild reaction conditions offering a robust pathway for producing high-purity intermediates essential for drug development programs With global pharmaceutical manufacturers increasingly demanding cost-effective sustainable manufacturing processes this patent represents a strategic advancement that aligns with stringent regulatory requirements while improving supply chain resilience The incorporation of trifluoromethyl groups into quinazolinone frameworks is known to confer superior pharmacokinetic properties including enhanced metabolic stability bioavailability and lipophilicity making this technology particularly valuable for next-generation therapeutic agents targeting oncology infectious diseases and central nervous system disorders This report provides comprehensive analysis of technical merits commercial implications addressing specific concerns from R&D procurement and supply chain leadership within multinational pharmaceutical enterprises
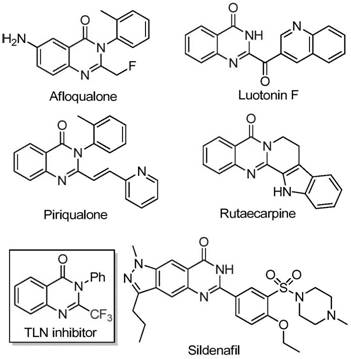
The Limitations of Conventional Methods vs The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for trifluoromethyl-substituted quinazolinones have been plagued by severe operational constraints including harsh reaction conditions requiring elevated temperatures or pressures expensive transition metal catalysts such as palladium complexes and limited substrate scope that restricts structural diversity Essential precursors like trifluoroacetic anhydride or ethyl trifluoroacetate often lead to low yields due to competing side reactions poor functional group tolerance necessitating extensive purification procedures that increase production costs reduce overall efficiency Many existing protocols involve multiple synthetic steps with intermediate isolations creating extended manufacturing timelines higher risk of impurity formation compromising final product purity—a critical concern given pharmaceutical industry requirements where even trace impurities can invalidate drug candidates The reliance on specialized equipment hazardous reagents poses significant scalability challenges when transitioning from laboratory to industrial production scales creating supply chain bottlenecks particularly problematic for time-sensitive drug development programs requiring reliable intermediate sourcing
The Novel Approach
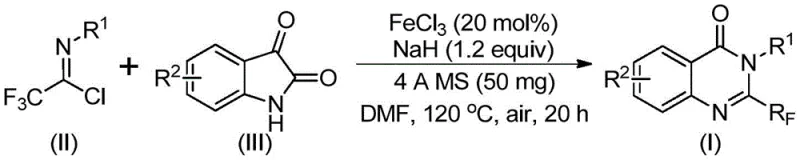
The patented methodology overcomes these limitations through an elegant iron-catalyzed one-pot cyclization reaction utilizing inexpensive ferric chloride as catalyst alongside commercially available trifluoroethylimidoyl chloride as key building block Operating under mild conditions with DMF solvent at controlled temperatures first at 40°C followed by heating to 120°C the process achieves high yields across diverse substrate combinations while maintaining excellent functional group compatibility Elimination of precious metal catalysts reduces raw material costs simplifies downstream processing by avoiding complex metal removal steps required in traditional methods Crucially the reaction demonstrates exceptional scalability from gram-scale laboratory demonstrations to potential multi-ton industrial production without significant optimization addressing critical supply chain vulnerabilities through robustness reproducibility This streamlined approach shortens synthetic pathways compared to conventional multi-step sequences enhancing manufacturing throughput while ensuring consistent high-purity output meeting pharmaceutical industry specifications
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The reaction mechanism proceeds through a well-defined sequence where ferric chloride activates the carbonyl group of isatin derivative facilitating nucleophilic attack by nitrogen atom of trifluoroethylimidoyl chloride This initial addition forms tetrahedral intermediate undergoing rapid decarbonylation promoted by sodium hydride base generating key iminium species Subsequent intramolecular cyclization occurs via nucleophilic aromatic substitution where amide nitrogen attacks electrophilic carbon adjacent to trifluoromethyl group forming quinazolinone core structure with concomitant hydrogen chloride release Iron catalyst plays dual role activating carbonyl electrophile stabilizing developing negative charge during cyclization ensuring high regioselectivity minimal byproduct formation This mechanistic pathway explains observed broad substrate tolerance as reaction avoids sensitive transition metal-mediated steps limiting functional group compatibility in alternative methods
Impurity control is achieved through precise reaction condition optimization where two-stage temperature profile prevents premature decomposition while ensuring complete conversion Molecular sieves effectively scavenge trace moisture preventing hydrolysis of sensitive intermediates stoichiometric ratio of sodium hydride to ferric chloride maintains optimal basicity without promoting side reactions Chromatographic purification protocol targets residual catalyst unreacted starting materials through selective elution parameters resulting in final products consistently exceeding pharmaceutical purity standards This robust impurity management system proves particularly valuable for producing intermediates destined for active pharmaceutical ingredients where even trace impurities can compromise drug safety efficacy profiles meeting stringent regulatory requirements across global markets
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
This patented synthesis route represents significant advancement over conventional methods providing streamlined pathway eliminating multiple intermediate steps while maintaining high product quality Process begins with careful selection of appropriately substituted aromatic amines generating required trifluoroethylimidoyl chloride precursors through rapid phosphine-mediated reaction sequence Key operational parameters including catalyst loading at precise levels temperature control during two-stage profile prove critical achieving optimal yields across diverse substrate combinations Standardized procedure enables reliable scale-up from laboratory to commercial production volumes maintaining consistent product specifications required by regulatory authorities Detailed standardized synthesis steps are provided below for immediate implementation in manufacturing environments
- Combine ferric chloride catalyst at 20 mol% loading with sodium hydride base at 1.2 equivalents along with molecular sieves trifluoroethylimidoyl chloride precursor and isatin derivative in DMF solvent under controlled inert atmosphere.
- Maintain initial reaction temperature at 40°C for eight to ten hours followed by precise heating to 120°C under air atmosphere for eighteen to twenty hours ensuring complete conversion.
- Execute post-reaction processing through filtration over silica gel followed by column chromatography purification using optimized elution parameters to isolate target quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
Implementation of this novel synthesis methodology delivers substantial operational improvements directly addressing critical pain points in pharmaceutical intermediate procurement supply chain management By replacing expensive transition metal catalysts with cost-effective iron-based systems manufacturers achieve significant reductions raw material expenditures eliminating complex purification requirements associated metal residue removal Process demonstrates exceptional robustness across varying production scales without requiring specialized equipment modifications enhancing manufacturing flexibility reducing time-to-market new drug candidates requiring these critical intermediates Strategic adoption provides procurement teams with reliable sourcing options while strengthening overall supply chain resilience against market volatility
- Cost Reduction in Manufacturing: Elimination precious metal catalysts results substantial cost savings throughout production cycle removing both acquisition expenses downstream processing costs associated metal removal procedures Use commercially available starting materials favorable stoichiometric ratios optimizes raw material utilization efficiency minimizing waste generation compared conventional multi-step syntheses requiring additional reagents purification stages
- Enhanced Supply Chain Reliability: Reliance widely accessible raw materials including common solvents molecular sieves ensures consistent supply availability regardless geopolitical fluctuations market shortages affecting specialized chemical reagents Simplified reaction sequence fewer critical control points reduces vulnerability process failures during scale-up enabling predictable production timelines supporting just-in-time manufacturing strategies essential modern pharmaceutical supply chains
- Scalability and Environmental Compliance: Demonstrated scalability from gram-scale laboratory reactions potential multi-ton production volumes without significant process re-engineering provides manufacturers confidence seamless technology transfer across different facilities Reduced synthetic steps lower overall energy consumption waste generation per unit output eliminating hazardous reagents commonly used alternative methods resulting environmentally sustainable manufacturing profile aligning stringent global regulatory requirements
Frequently Asked Questions (FAQ)
The following questions address common technical commercial concerns regarding implementation patented synthesis methodology pharmaceutical intermediates Each response grounded specific experimental data process parameters documented within patent CN111675662B providing accurate guidance manufacturing teams evaluating technology commercial adoption
Q: How does this iron-catalyzed method overcome yield limitations compared to conventional palladium-based syntheses?
A: The patented process achieves consistently higher yields through mild two-stage temperature control eliminating decomposition pathways while leveraging iron's dual activation capability that maintains excellent functional group tolerance across diverse substrates without requiring expensive transition metal systems.
Q: What specific advantages does this synthesis offer regarding supply chain reliability for pharmaceutical intermediates?
A: By utilizing widely available starting materials including standard molecular sieves and common solvents alongside elimination of specialized catalysts this method ensures consistent raw material availability while demonstrating proven scalability from laboratory to commercial production volumes without process re-engineering.
Q: How does this methodology address impurity control challenges critical for pharmaceutical applications?
A: The precisely optimized stoichiometric ratio of sodium hydride to ferric chloride combined with molecular sieve moisture scavenging creates robust impurity management that consistently delivers high-purity output meeting stringent regulatory specifications through integrated chromatographic purification protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical instrumentation As trusted CDMO partner specializing complex heterocyclic compounds we successfully implemented patented iron-catalyzed methodology across multiple client projects delivering consistent high-purity intermediates meeting global regulatory standards Our vertically integrated manufacturing capabilities ensure seamless transition process development full-scale production without compromising quality timeline commitments providing reliable partnership throughout entire product lifecycle
Leverage our technical expertise through Customized Cost-Saving Analysis tailored specific manufacturing requirements contact our technical procurement team today request detailed COA data route feasibility assessments next pharmaceutical intermediate sourcing initiative
