Scalable One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Pharmaceutical Applications
Scalable One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Pharmaceutical Applications
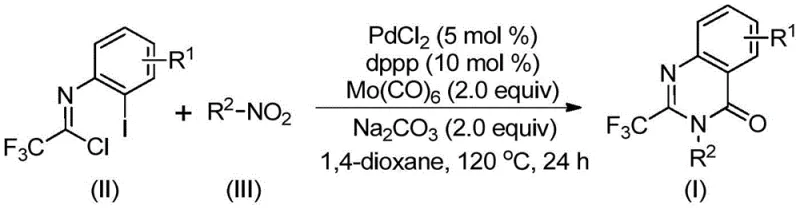
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, safer, and cost-effective synthetic routes for bioactive heterocycles. A significant breakthrough in this domain is documented in Chinese Patent CN112480015B, which discloses a robust multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. These privileged scaffolds are ubiquitous in medicinal chemistry, serving as core structures for a wide array of therapeutic agents exhibiting antifungal, antibacterial, antiviral, and anticancer properties. The introduction of the trifluoromethyl group is particularly strategic, as it enhances metabolic stability, lipophilicity, and bioavailability of the parent molecule. This patent presents a transformative approach that leverages palladium catalysis to construct these complex architectures from readily available nitro compounds and trifluoroethylimidoyl chlorides, addressing critical bottlenecks in traditional synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core, especially with specific trifluoromethyl substitution patterns, has been fraught with synthetic challenges that hinder industrial scalability. Conventional methodologies often rely on harsh reaction conditions, such as the use of high-pressure carbon monoxide gas, which necessitates expensive and specialized autoclave equipment, posing significant safety hazards in a manufacturing environment. Furthermore, many existing routes require pre-activated substrates, such as 2-bromoformylanilines or acid anhydrides, which are not only costly to procure but also generate substantial chemical waste during their preparation. Traditional catalysts based on ruthenium or iron often suffer from limited substrate scope and lower turnover frequencies, resulting in inconsistent yields and difficult purification processes that increase the overall cost of goods sold (COGS) for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology outlined in CN112480015B offers a streamlined, one-pot solution that drastically simplifies the synthetic workflow. By utilizing molybdenum hexacarbonyl [Mo(CO)6] as a safe and convenient solid surrogate for carbon monoxide, the process eliminates the need for high-pressure gas infrastructure. The reaction employs a palladium catalyst system, specifically PdCl2 paired with the dppp ligand, to facilitate a cascade transformation directly from cheap nitro compounds. This approach not only reduces the number of unit operations but also exhibits remarkable functional group tolerance, allowing for the synthesis of diverse derivatives without extensive protecting group strategies. The ability to convert simple, commodity-grade starting materials into high-value intermediates in a single vessel represents a paradigm shift towards greener and more economical chemical manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific analogs. The reaction is believed to proceed through a sophisticated catalytic cycle initiated by the reduction of the nitro group. Initially, the Mo(CO)6 serves a dual purpose: it acts as the carbon monoxide source for the carbonylation step and potentially facilitates the reduction of the nitro compound to the corresponding amine in situ. Once the amine is generated, it undergoes a base-promoted nucleophilic attack on the trifluoroethylimidoyl chloride, forming a trifluoroacetamidine intermediate. This step is critical as it sets the stage for the subsequent cyclization.
Following the formation of the amidine derivative, the palladium catalyst enters the cycle by oxidative addition into the carbon-iodine bond of the aromatic ring. The released carbon monoxide from the thermal decomposition of Mo(CO)6 then inserts into the carbon-palladium bond to generate an acyl-palladium species. Intramolecular coordination of the nitrogen atom to the palladium center, followed by reductive elimination, closes the quinazolinone ring and regenerates the active Pd(0) catalyst. This intricate dance of bond formations ensures high chemoselectivity, minimizing the formation of side products such as ureas or simple amides, thereby simplifying the impurity profile and easing the burden on downstream purification teams.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize yield and purity. The patent details a standardized protocol where reagents are combined in a Schlenk tube under inert atmosphere, highlighting the sensitivity of the palladium catalyst to oxygen. The choice of solvent is pivotal, with 1,4-dioxane identified as the optimal medium due to its ability to dissolve all components effectively while stabilizing the transition states. The reaction temperature is maintained at 120°C, a balance between providing sufficient energy for the carbonyl insertion and preventing thermal degradation of sensitive functional groups. For a comprehensive understanding of the stoichiometry and specific conditions required for different substrates, refer to the detailed procedural guide below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material supply chain. By utilizing nitro compounds, which are commodity chemicals available in bulk quantities globally, manufacturers can mitigate the risks associated with sourcing specialized, low-volume precursors. This shift to abundant feedstocks ensures a more resilient supply chain, less susceptible to market fluctuations or geopolitical disruptions that often plague the availability of exotic building blocks.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of high-pressure equipment requirements. Removing the need for specialized autoclaves significantly lowers capital expenditure (CAPEX) for new production lines and reduces maintenance costs for existing facilities. Furthermore, the use of Mo(CO)6 as a solid CO source avoids the logistical complexities and safety costs associated with transporting and storing high-pressure carbon monoxide cylinders. The high atom economy of the one-pot design also means less solvent usage and reduced waste disposal costs, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system ensures consistent batch-to-batch quality, a critical factor for maintaining uninterrupted supply to downstream API manufacturers. The broad substrate scope means that a single manufacturing platform can be adapted to produce a library of different quinazolinone derivatives simply by swapping the nitro compound input. This flexibility allows suppliers to respond rapidly to changing market demands or custom synthesis requests without retooling entire production lines, thereby shortening lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with modern green chemistry principles. The avoidance of toxic gas handling and the use of relatively benign solvents simplify regulatory compliance and permit acquisition. The post-treatment process, involving simple filtration and standard chromatography, is easily scalable from gram to kilogram scales without encountering the mixing or heat transfer issues common in heterogeneous high-pressure reactions. This scalability ensures that the process can grow seamlessly from pilot plant trials to full commercial production.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled answers to common inquiries based on the patent data. These insights address concerns regarding safety, substrate versatility, and purification, providing a clear picture of what partners can expect when integrating this technology into their supply chains. Understanding these nuances is essential for making informed decisions about process adoption and vendor selection.
Q: What are the advantages of using Mo(CO)6 over high-pressure CO gas in this synthesis?
A: Using Mo(CO)6 as a solid carbon monoxide substitute eliminates the need for specialized high-pressure autoclaves, significantly reducing equipment costs and safety risks associated with handling toxic CO gas while maintaining high reaction efficiency.
Q: Does this method tolerate diverse functional groups on the nitro compound substrate?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating various substituents such as halogens (F, Cl, Br), alkyl groups (methyl), and electron-withdrawing groups (CF3) without compromising yield.
Q: Is the purification process complex for large-scale production?
A: The post-treatment is straightforward, involving simple filtration followed by standard silica gel column chromatography, which is a widely established and scalable technique in industrial organic synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the multi-component one-pot synthesis described in CN112480015B for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to industrial reality. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic route for your next drug development program. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient methodology can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom targets, ensuring a secure and competitive supply chain for your critical intermediates.
