Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-Up
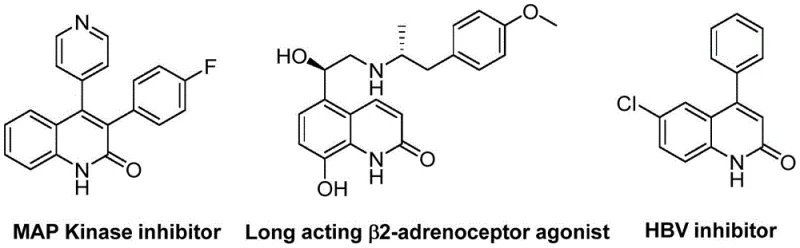
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN113045489B, which discloses a highly efficient preparation method for 3-arylquinolin-2(1H)-one derivatives. These derivatives are not merely academic curiosities; they represent a cornerstone structural motif found in a vast array of therapeutic agents, including antibiotics, antiplatelet drugs, antitumor agents, and endothelin receptor antagonists. The biological significance of this scaffold is underscored by its presence in potent MAP Kinase inhibitors and long-acting β2-adrenoceptor agonists, highlighting the urgent demand for reliable synthetic routes that can support the rigorous purity standards required by global regulatory bodies.
The innovation presented in this patent addresses several longstanding challenges in heterocyclic chemistry by leveraging a palladium-catalyzed aminocarbonylation strategy. Unlike traditional approaches that often rely on hazardous gaseous carbon monoxide or multi-step sequences with poor atom economy, this novel protocol utilizes benzisoxazole as a unique dual-purpose reagent. It acts simultaneously as the nitrogen source and the formyl source, thereby streamlining the synthetic pathway. This strategic design not only enhances operational safety by avoiding high-pressure CO gas but also significantly improves the overall process mass intensity (PMI), a key metric for green chemistry and cost-effective manufacturing. For R&D directors and process chemists, this represents a paradigm shift towards more sustainable and economically viable production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)-one derivatives has relied heavily on classical named reactions such as the Vilsmeier-Haack, Knorr, and Friedlander condensations. While these methods are well-documented in literature, they frequently suffer from significant drawbacks when applied to modern industrial settings. For instance, the Vilsmeier-Haack reaction typically requires the use of phosphorus oxychloride (POCl3) and dimethylformamide (DMF), generating substantial amounts of corrosive waste and posing severe environmental disposal challenges. Furthermore, these traditional routes often exhibit limited functional group tolerance, necessitating extensive protection and deprotection strategies that inflate both the cost of goods sold (COGS) and the production lead time. Transition metal-catalyzed improvements have been attempted, yet many still depend on the direct use of carbon monoxide gas, which introduces complex engineering requirements for high-pressure reactors and stringent safety protocols to prevent leakage and exposure.
The Novel Approach
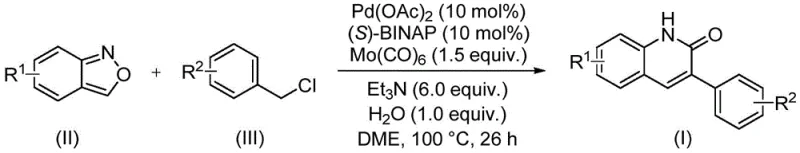
The methodology outlined in patent CN113045489B offers a transformative solution by employing a palladium-catalyzed carbonylative coupling between benzisoxazoles and benzyl chlorides. This approach elegantly bypasses the need for external CO gas by utilizing molybdenum hexacarbonyl [Mo(CO)6] as a solid, easy-to-handle CO surrogate. The reaction proceeds under relatively mild thermal conditions (90~110°C) in a sealed tube, utilizing a catalytic system composed of palladium acetate and the chiral ligand (S)-BINAP. This setup allows for the direct construction of the quinolinone core with exceptional efficiency, achieving isolated yields as high as 97% in optimized examples. The versatility of this method is further demonstrated by its broad substrate scope, accommodating various electron-withdrawing and electron-donating groups on both the benzisoxazole and benzyl chloride partners, thus providing a powerful tool for the rapid generation of diverse chemical libraries for drug discovery programs.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The mechanistic underpinning of this transformation involves a sophisticated catalytic cycle initiated by the oxidative addition of the benzyl chloride to the active Pd(0) species generated in situ from Pd(OAc)2 and (S)-BINAP. This step forms a benzyl-palladium(II) complex, which subsequently undergoes insertion of carbon monoxide released from the thermal decomposition of Mo(CO)6. The resulting acyl-palladium intermediate is then intercepted by the nitrogen atom of the benzisoxazole ring. Crucially, the benzisoxazole moiety undergoes a ring-opening process that delivers both the nitrogen atom and the carbonyl carbon necessary for the lactam formation, effectively acting as a masked isocyanate equivalent. This intramolecular cyclization is followed by reductive elimination to release the final 3-arylquinolin-2(1H)-one product and regenerate the Pd(0) catalyst, closing the loop. The presence of triethylamine as a base and water as an additive plays a vital role in facilitating the ring-opening of the benzisoxazole and neutralizing the HCl byproduct, ensuring the catalytic turnover remains high throughout the 26-hour reaction period.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal cyclizations. The use of a well-defined palladium catalyst system ensures high regioselectivity, minimizing the formation of positional isomers that are notoriously difficult to separate. Furthermore, the mild reaction temperature of 100°C helps suppress side reactions such as polymerization or over-carbonylation, which are common pitfalls in harsher acidic or basic conditions. The patent data indicates that even with sterically hindered substrates or those containing sensitive functional groups like cyano or trifluoromethyl moieties, the reaction maintains high fidelity. This robustness is critical for pharmaceutical manufacturing, where the impurity profile must be tightly controlled to meet ICH guidelines. The ability to tolerate halogens (Cl, F) on the aromatic rings also opens up opportunities for downstream cross-coupling reactions, allowing medicinal chemists to further elaborate the molecular structure without needing to reintroduce these handles through additional synthetic steps.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, precise adherence to the stoichiometric ratios and reaction parameters described in the patent is essential. The process begins with the careful weighing of the palladium catalyst, ligand, and CO source, followed by the addition of the organic substrates and solvent. The reaction is typically conducted in 1,2-dimethoxyethane (DME), which provides excellent solubility for the reactants and stability for the catalytic species. Maintaining the temperature at 100°C for a duration of 26 hours is critical to drive the reaction to completion, as shorter times may result in incomplete conversion of the starting benzyl chloride. Upon completion, the workup procedure is straightforward, involving simple filtration to remove insoluble salts and metal residues, followed by silica gel treatment and column chromatography. This simplicity in downstream processing is a major operational benefit, reducing the burden on purification teams and accelerating the timeline from reaction to pure compound.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for 26 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route presents compelling economic and logistical benefits that directly impact the bottom line. The primary driver for cost reduction lies in the selection of starting materials; benzisoxazoles and substituted benzyl chlorides are commodity chemicals that are widely available from multiple global suppliers, ensuring a competitive pricing landscape and mitigating the risk of single-source dependency. Unlike processes that require specialized high-pressure equipment for handling carbon monoxide gas, this method operates in standard glass-lined or stainless steel reactors capable of moderate temperatures, significantly lowering the capital expenditure (CAPEX) required for technology transfer and scale-up. The elimination of toxic gas handling also reduces insurance premiums and safety compliance costs, contributing to a leaner operational budget.
- Cost Reduction in Manufacturing: The economic viability of this process is further enhanced by the high atom economy and the use of Mo(CO)6 as a safe, solid CO source. By avoiding the infrastructure costs associated with gaseous CO delivery and scrubbing systems, manufacturers can achieve substantial cost savings in facility maintenance and operation. Additionally, the high yields reported (often exceeding 90%) mean that less raw material is wasted per kilogram of product produced, directly lowering the variable cost of production. The simplified workup procedure, which avoids complex aqueous extractions or crystallizations in favor of direct chromatography or filtration, reduces solvent consumption and waste disposal fees, aligning with both financial and environmental sustainability goals.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the commercial availability of all reagents. Since the reaction tolerates a wide range of functional groups, manufacturers can source generic grades of benzyl chlorides without needing ultra-high purity specifications, which are often subject to longer lead times and higher price volatility. The use of a heterogeneous-style workup (filtration) facilitates easier scale-up compared to emulsion-prone liquid-liquid extractions, ensuring consistent batch-to-batch quality and predictable production schedules. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, preventing costly production stoppages due to intermediate shortages.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method scores highly due to the avoidance of corrosive reagents like POCl3 and toxic gases. The waste stream is primarily organic solvent and silica gel, which are easier to treat and incinerate compared to heavy metal sludge or acidic aqueous waste. This simplifies the permitting process for new manufacturing lines and reduces the long-term liability associated with hazardous waste management. The scalability of the process is proven by its successful application across a diverse library of substrates in the patent examples, suggesting that translation from gram-scale R&D to multi-ton commercial production can be achieved with minimal process re-engineering, thereby accelerating time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and strategic value of this synthetic route for industry stakeholders. Understanding these nuances is essential for making informed decisions about process adoption and technology licensing.
Q: What is the primary advantage of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl (C1) source, eliminating the need for external toxic carbon monoxide gas and simplifying the reaction stoichiometry.
Q: What are the optimal reaction conditions described in the patent?
A: The optimal conditions involve using Pd(OAc)2 (10 mol%), (S)-BINAP (10 mol%), and Mo(CO)6 (1.5 equiv.) in DME solvent at 100°C for 26 hours, yielding up to 97% efficiency.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes commercially available starting materials like benzyl chlorides and avoids high-pressure gas equipment, making it highly scalable and safer for industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
As the global demand for complex heterocyclic intermediates continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies and reliable supply chains. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver products with stringent purity specifications. Our rigorous QC labs are equipped to handle the analytical challenges of complex quinolinone derivatives, ensuring that every batch meets the exacting standards required by international pharmaceutical regulators. By integrating advanced catalytic methods like the one described in CN113045489B into our portfolio, we offer our clients a competitive edge through faster development timelines and superior cost structures.
We invite potential partners to engage with our technical procurement team to discuss how this specific technology can be tailored to your project needs. Whether you require a Customized Cost-Saving Analysis for an existing route or need to evaluate the feasibility of scaling this novel Pd-catalyzed process, our experts are ready to assist. Please contact us to request specific COA data for related intermediates and comprehensive route feasibility assessments, and let us help you optimize your supply chain for the next generation of therapeutic innovations.
