Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing privileged heterocyclic scaffolds. Among these, the quinolin-2(1H)-one core stands out due to its prevalence in bioactive molecules ranging from antibiotics to antitumor agents. A significant technological breakthrough in this domain is detailed in patent CN113045489B, which discloses a novel preparation method for 3-arylquinolin-2(1H)-one derivatives. This innovation leverages a sophisticated palladium-catalyzed aminocarbonylation strategy that fundamentally alters the synthetic landscape by utilizing benzisoxazole as a dual-function reagent. Unlike traditional approaches that often rely on hazardous gases or multi-step sequences, this method integrates the nitrogen and formyl components into a single, stable heterocyclic starting material. The versatility of this approach is evidenced by the successful synthesis of diverse analogs, including structures resembling MAP Kinase inhibitors and HBV inhibitors, highlighting its immediate relevance to modern drug discovery pipelines.
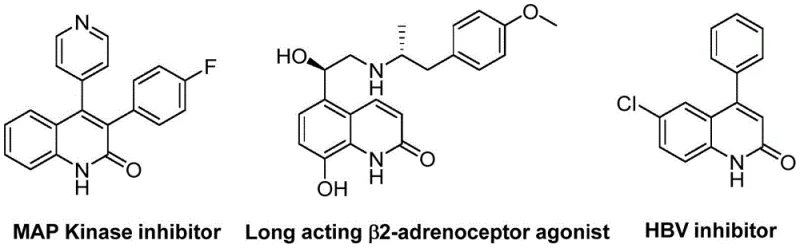
For R&D directors evaluating new routes, the structural diversity achievable through this protocol is paramount. The ability to tolerate a wide array of functional groups on both the benzisoxazole and the benzyl chloride partners allows for the rapid generation of chemical libraries. This is critical for structure-activity relationship (SAR) studies where subtle electronic or steric modifications can dictate biological efficacy. Furthermore, the reaction conditions are remarkably mild, operating at 100°C in ethylene glycol dimethyl ether (DME), which avoids the decomposition of sensitive functional groups often seen in harsher Friedlander or Vilsmeier-Haack reactions. By streamlining the synthesis of these complex heterocycles, this technology offers a reliable pharmaceutical intermediate supplier pathway that aligns with the rigorous purity and efficiency standards demanded by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolinone skeleton has relied on classical named reactions such as the Knorr, Friedlander, or Vilsmeier-Haack condensations. While these methods are well-established, they suffer from significant drawbacks when applied to modern, complex molecule synthesis. For instance, the Friedlander reaction typically requires high temperatures and strong acidic or basic conditions, which can lead to the degradation of acid-sensitive or base-sensitive substituents on the aromatic rings. Additionally, these traditional routes often necessitate the use of specialized aniline derivatives that may not be readily commercially available, forcing procurement teams to engage in custom synthesis of starting materials, thereby inflating costs and extending lead times. Another critical limitation is the reliance on toxic reagents like phosphorus oxychloride in Vilsmeier-Haack reactions, which generates substantial hazardous waste and complicates environmental compliance and waste disposal protocols in large-scale manufacturing facilities.
The Novel Approach
The methodology described in the patent data represents a paradigm shift by employing a transition metal-catalyzed cascade reaction that overcomes these historical bottlenecks. By utilizing benzisoxazole and benzyl chloride compounds as the primary building blocks, the process eliminates the need for pre-functionalized anilines. The reaction proceeds via a palladium-catalyzed mechanism where the benzisoxazole ring acts uniquely as both the nitrogen donor and the source of the carbonyl carbon, effectively internalizing the carbonylation step. This not only simplifies the reagent profile but also enhances safety by avoiding the direct handling of high-pressure carbon monoxide gas, which is traditionally required for carbonylation reactions. The use of a solid carbonyl source like molybdenum hexacarbonyl in conjunction with the benzisoxazole framework ensures a controlled release of reactive species, leading to cleaner reaction profiles and higher selectivity for the desired 3-aryl substitution pattern.
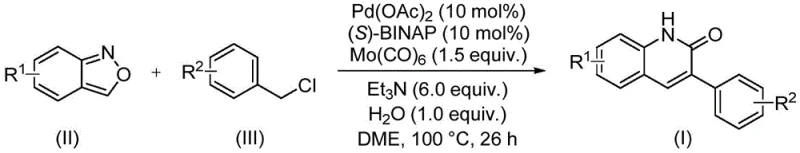
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
Understanding the mechanistic underpinnings of this transformation is essential for process optimization and scale-up. The reaction initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species, generated in situ from palladium acetate and the chiral ligand (S)-BINAP. This step forms a benzyl-palladium(II) intermediate, which is then poised for coordination with the benzisoxazole substrate. The unique reactivity of benzisoxazole allows for ring-opening under these catalytic conditions, releasing the nitrogen atom to coordinate with the palladium center while simultaneously providing the necessary carbon framework for the quinolinone ring closure. The presence of molybdenum hexacarbonyl serves as a supplementary carbonyl source or facilitates the carbonylation event, ensuring the efficient formation of the lactam carbonyl group at the 2-position. Triethylamine acts as a base to neutralize the hydrochloric acid byproduct, driving the equilibrium forward, while the addition of water plays a crucial role in facilitating the hydrolysis or proton transfer steps necessary for the final aromatization and product release.
From an impurity control perspective, this mechanism offers distinct advantages. The high regioselectivity observed, consistently yielding the 3-aryl isomer, minimizes the formation of regioisomeric byproducts that are notoriously difficult to separate via crystallization or chromatography. The tolerance for various substituents, such as halogens (Cl, F), alkoxy groups (OMe), and electron-withdrawing groups (CN, CF3), suggests that the catalytic cycle is robust against electronic perturbations. This stability reduces the risk of side reactions like homocoupling of the benzyl chloride or polymerization of the benzisoxazole. For quality control teams, this means a simpler impurity profile and a more straightforward purification process, typically involving standard silica gel column chromatography to achieve high-purity pharmaceutical intermediates suitable for downstream coupling reactions.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized reaction parameters to maximize yield and reproducibility. The protocol involves charging a sealed reaction vessel with the catalyst system comprising palladium acetate and (S)-BINAP, along with molybdenum hexacarbonyl as the carbonyl source. The stoichiometry is critical, with a molar ratio of benzisoxazole to benzyl chloride typically maintained around 1:2.5 to ensure complete consumption of the limiting reagent. The reaction is conducted in DME solvent at 100°C for approximately 26 hours. Detailed standardized operating procedures regarding reagent addition order, stirring rates, and workup protocols are essential for consistent results.
- Charge a sealed tube with palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and the specific benzyl chloride compound.
- Add ethylene glycol dimethyl ether (DME) as the solvent and stir the mixture to ensure homogeneity before heating.
- Heat the reaction mixture to 100°C for 26 hours, then perform filtration and silica gel chromatography to isolate the pure derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic and logistical implications of this synthetic route are profound. The shift towards using commodity chemicals like benzyl chlorides and benzisoxazoles drastically reduces the dependency on custom-synthesized, high-cost starting materials. This accessibility translates directly into cost reduction in pharmaceutical intermediate manufacturing, as the raw material basket is composed of bulk chemicals available from multiple global vendors. Furthermore, the elimination of high-pressure carbon monoxide gas cylinders removes a significant safety hazard and regulatory burden associated with storage and handling, thereby lowering insurance and facility compliance costs. The operational simplicity of the reaction, which does not require specialized high-pressure autoclaves beyond standard sealed tubes or reactors capable of 100°C, allows for implementation in existing multipurpose production units without major capital expenditure.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the high atom economy and the use of inexpensive catalysts relative to the value of the final product. By combining the nitrogen and carbonyl sources into the benzisoxazole structure, the process reduces the total number of reagents required per batch. This consolidation leads to substantial cost savings in raw material procurement and waste treatment. Additionally, the high yields reported, often exceeding 90% for unsubstituted or electron-neutral substrates, mean that less starting material is wasted, further enhancing the overall process mass intensity (PMI) metrics which are critical for green chemistry assessments and cost accounting.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the widespread availability of the key starting materials. Benzyl chlorides with various substituents are staple products in the fine chemical industry, ensuring that supply disruptions for a specific precursor can be easily mitigated by sourcing alternative grades or suppliers. The robustness of the reaction conditions also implies a lower risk of batch failure due to sensitive parameter fluctuations. This reliability reduces lead time for high-purity pharmaceutical intermediates, allowing manufacturing schedules to be met with greater confidence and reducing the need for excessive safety stock inventory.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the homogeneous nature of the catalytic system and the moderate temperature requirements. The absence of gaseous reagents simplifies reactor design and operation at scale. From an environmental standpoint, the process generates fewer hazardous byproducts compared to classical methods involving phosphorus halides or strong mineral acids. The primary waste streams consist of organic solvents and salt byproducts which are easier to treat and dispose of in accordance with modern environmental regulations, supporting the company's sustainability goals and reducing the carbon footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl source, simplifying the reagent list and improving atom economy compared to traditional methods requiring separate amine and carbonyl sources.
Q: What is the typical yield range for this palladium-catalyzed process?
A: According to patent CN113045489B, the reaction demonstrates high efficiency with isolated yields ranging from 68% to 97% across various substrates with different electronic properties.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available starting materials like benzyl chlorides and operates at a moderate temperature of 100°C without requiring high-pressure carbon monoxide gas, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN113045489B for accelerating drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-arylquinolin-2(1H)-one derivative meets the exacting standards required for clinical and commercial applications. Our commitment to quality ensures that the complex stereochemical and structural integrity of these heterocycles is preserved throughout the production lifecycle.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule's bill of materials. We encourage potential partners to contact us to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for novel derivatives. Let us help you optimize your supply chain and reduce time-to-market with our reliable 3-arylquinolin-2(1H)-one supplier capabilities.
