Scalable Synthesis of Beta-Halopyrrole Compounds via Ordered Serial Cyclization for Commercial Production
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and selective synthetic routes for bioactive scaffolds. A recent breakthrough detailed in patent CN114989063A introduces a sophisticated yet practical method for constructing beta-halopyrrole compounds, a structural motif prevalent in marine alkaloids and pharmaceutical agents. This innovation addresses long-standing challenges in regioselectivity and substrate availability by employing a palladium-catalyzed ordered serial cyclization strategy. By utilizing readily accessible N-substituted anilines and alkyne halides, this technology bypasses the limitations of classical halogenation methods, offering a streamlined pathway to highly functionalized intermediates. For industry leaders seeking a reliable pharmaceutical intermediate supplier, understanding this technological shift is crucial for optimizing supply chains and reducing development timelines in complex molecule synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of beta-halopyrrole skeletons has relied heavily on direct halogenation strategies using reagents like N-bromosuccinimide (NBS). While conceptually straightforward, these classical approaches suffer from significant drawbacks that hinder their utility in modern process chemistry. The inherent reactivity of the pyrrole ring often leads to poly-substitution, resulting in complex mixtures of regioisomers that are difficult and costly to separate. Furthermore, these methods frequently require the pyrrole core to be pre-synthesized with specific protecting groups or substituents to control selectivity, adding unnecessary steps to the overall synthesis. Such inefficiencies not only inflate the cost of goods but also generate substantial chemical waste, posing environmental compliance challenges for large-scale operations. Consequently, there is a pressing need for alternative strategies that offer superior control over bond formation without compromising on operational simplicity.
The Novel Approach
The methodology disclosed in CN114989063A represents a paradigm shift by constructing the pyrrole ring de novo through a three-component coupling reaction. Instead of modifying an existing pyrrole, this novel approach orchestrates the reaction between one molecule of N-substituted aniline and two molecules of alkyne halide under palladium catalysis. This ordered serial cyclization allows for the precise installation of the halogen atom at the beta-position during the ring-closing event itself. The reaction proceeds under mild conditions using methyl tert-butyl ether as a solvent, demonstrating remarkable tolerance to various functional groups on both the aniline and alkyne components. This level of precision eliminates the need for tedious purification of isomeric byproducts, thereby enhancing the overall efficiency and purity of the final product.
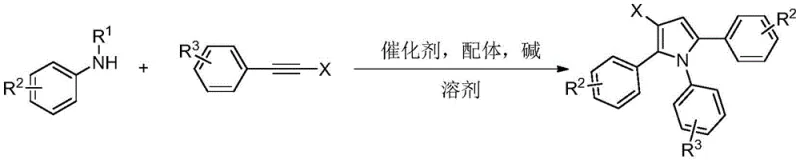
Mechanistic Insights into Pd-Catalyzed Ordered Serial Cyclization
At the heart of this synthetic advancement lies a sophisticated catalytic cycle driven by palladium species. The mechanism initiates with the coordination of the N-substituted aniline to the palladium center, facilitated by the diphenylphosphinic acid ligand, forming a reactive nitrogen-palladium species. This intermediate then undergoes migratory insertion with the carbon-carbon triple bond of the first alkyne halide molecule, generating an alkenyl-palladium complex. Subsequently, this species reacts with a second molecule of alkyne halide through an oxidative addition process. The sequence culminates in a reductive elimination and cyclization step, promoted by bromide ions within the system, which closes the pyrrole ring and releases the beta-halopyrrole product while regenerating the active catalyst. This intricate dance of bond formations ensures that the halogen atom is incorporated specifically at the desired position, showcasing the power of transition metal catalysis in controlling molecular architecture.
Beyond the primary catalytic cycle, the choice of reagents plays a pivotal role in managing impurity profiles and ensuring high purity specifications. The use of a mixed base system comprising lithium hydroxide monohydrate and sodium acetate is critical for neutralizing acidic byproducts and maintaining the optimal pH for the cyclization to proceed without side reactions. Furthermore, the specific molar ratios employed, such as a slight excess of alkyne halide (6.0-7.0 equivalents), drive the equilibrium towards the desired tri-substituted product, minimizing the formation of mono-alkynylated intermediates. This careful balancing of stoichiometry and reaction conditions results in a clean reaction profile, which is essential for meeting the stringent quality standards required for API intermediates and reduces the burden on downstream purification processes.
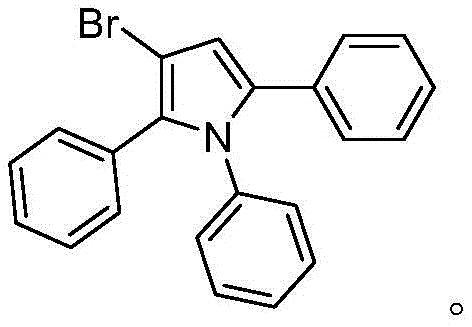
How to Synthesize Beta-Halopyrrole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the precise charging of substrates into a reactor equipped with efficient stirring capabilities, followed by the addition of the palladium catalyst and ligand under inert atmosphere conditions to prevent catalyst deactivation. Maintaining the reaction temperature within the narrow window of 100-110°C is vital for achieving complete conversion while avoiding thermal degradation of sensitive functional groups. Detailed standard operating procedures regarding the workup, including extraction with ethyl acetate and drying over anhydrous magnesium sulfate, are essential to isolate the crude product effectively before final purification via column chromatography.
- Charge a reactor with N-substituted aniline, alkyne halide (6.0-7.0 equiv), PdCl2 catalyst, diphenylphosphinic acid ligand, mixed base (LiOH/NaOAc), and MTBE solvent.
- Stir the reaction mixture at 100-110°C for 24-30 hours to facilitate the ordered serial cyclization.
- Cool to room temperature, extract with ethyl acetate, dry over magnesium sulfate, and purify via column chromatography to isolate the beta-halopyrrole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. By shifting from multi-step precursor synthesis to a direct one-pot cyclization, the overall process complexity is drastically simplified. This reduction in unit operations translates to lower capital expenditure on equipment and reduced labor costs associated with handling intermediate isolations. Moreover, the reliance on commodity chemicals like N-substituted anilines and alkyne halides, which are widely available from global chemical suppliers, mitigates the risk of raw material shortages. This accessibility ensures a stable supply chain, allowing manufacturers to plan production schedules with greater confidence and reliability.
- Cost Reduction in Manufacturing: The economic benefits of this method are derived primarily from the elimination of expensive and hazardous halogenating reagents like NBS, which often require specialized handling and disposal protocols. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, thereby improving the overall atom economy. Although palladium is a precious metal, the catalyst loading is kept relatively low, and the robust nature of the catalytic system suggests potential for recovery and recycling in continuous flow setups, further driving down the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: The use of methyl tert-butyl ether (MTBE) as the solvent is a strategic choice that enhances supply chain resilience. MTBE is a bulk industrial solvent with a well-established global supply network, unlike specialized fluorinated solvents that may face logistical bottlenecks. Furthermore, the reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors found in most contract manufacturing organizations, removing the need for exotic high-pressure or cryogenic equipment. This compatibility with existing infrastructure accelerates the technology transfer process from R&D to commercial production.
- Scalability and Environmental Compliance: Scalability is inherently supported by the homogeneous nature of the reaction mixture and the absence of gaseous reagents that complicate mass transfer in large vessels. The simplified workup procedure, involving standard liquid-liquid extraction, generates less aqueous waste compared to methods requiring extensive acid-base washes to remove inorganic salts. This reduction in waste volume aligns with green chemistry principles and lowers the costs associated with wastewater treatment and environmental compliance, making the process more sustainable for long-term manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-halopyrrole synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing alternatives. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline projects.
Q: What are the key advantages of this beta-halopyrrole synthesis method over traditional NBS halogenation?
A: Unlike traditional NBS halogenation which suffers from poor selectivity and multiple substitution issues, this patented method utilizes an ordered serial cyclization of N-substituted aniline and alkyne halides. This approach ensures high regioselectivity for the beta-position, avoids the need for pre-functionalized pyrrole precursors, and tolerates a wide range of functional groups, significantly simplifying the synthetic route.
Q: Is this palladium-catalyzed process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability. It employs commercially available raw materials like N-substituted anilines and alkyne halides, uses methyl tert-butyl ether (MTBE) as a common industrial solvent, and operates at moderate temperatures (100-110°C). The use of a robust palladium catalyst system with simple workup procedures makes it highly viable for commercial scale-up.
Q: What is the substrate scope for the alkyne halide component in this reaction?
A: The method demonstrates excellent substrate universality. It accommodates various alkyne halides including phenyl bromoacetylene and its derivatives with electron-donating groups (like methyl) or electron-withdrawing groups (like chloro, aldehyde). This flexibility allows for the construction of diverse beta-halopyrrole libraries essential for drug discovery and material science applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Halopyrrole Supplier
As the pharmaceutical and fine chemical industries continue to demand higher purity and more complex intermediates, partnering with an experienced CDMO is essential for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale manufacturing. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of beta-halopyrrole compound meets the exacting standards required for downstream drug synthesis. We understand the critical nature of timeline and quality in the competitive landscape of API development.
We invite you to leverage our technical expertise to optimize your supply chain and reduce your time to market. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your target molecules. Let us collaborate to bring your next generation of therapeutic agents to life with efficiency and precision.
