Advanced Palladium-Catalyzed Synthesis of Beta-Halopyrrole Compounds for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Beta-Halopyrrole Compounds for Commercial Scale-Up
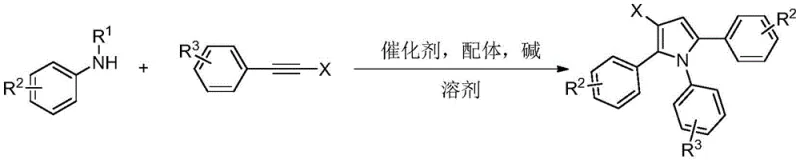
The landscape of organic synthesis for heterocyclic compounds is constantly evolving, driven by the need for more efficient and selective methodologies in the production of active pharmaceutical ingredients and advanced materials. A significant breakthrough in this domain is documented in patent CN114989063A, which discloses a robust synthesis method for beta-halogenated pyrrole compounds. This technology leverages a novel three-component reaction strategy involving N-substituted aniline and two molecules of alkyne halide under palladium catalysis. By utilizing an ordered serial cyclization mechanism, this approach overcomes many of the historical limitations associated with pyrrole functionalization, offering a pathway to highly functionalized scaffolds that are critical in medicinal chemistry and material science applications. The method stands out for its operational simplicity, utilizing common palladium salts and easily accessible ligands to achieve transformations that were previously difficult or required multi-step sequences.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of beta-halopyrrole skeletons has relied heavily on direct halogenation strategies, such as the reaction of pyrrole with N-bromosuccinimide (NBS). However, these classical methods are plagued by inherent selectivity issues due to the multiple reactive sites present on the pyrrole ring. Consequently, reactions often yield complex mixtures of mono-, di-, and poly-substituted products, making the isolation of a single beta-halopyrrole compound challenging and inefficient. Alternative approaches, such as intramolecular amine chlorination of allene compounds or oxidative cyclization of N-allylaniline, have been developed but often suffer from narrow substrate scope or require harsh oxidants like DDQ. Furthermore, many existing protocols necessitate the preparation of specialized starting materials through multi-step syntheses, which increases overall cost, waste generation, and process complexity, thereby hindering their applicability in large-scale commercial manufacturing environments.
The Novel Approach
In stark contrast to these traditional limitations, the methodology described in patent CN114989063A introduces a streamlined and highly selective route via palladium-catalyzed ordered serial cyclization. This innovative process directly couples commercially available N-substituted anilines with alkyne halides, bypassing the need for pre-functionalized pyrrole precursors. The reaction proceeds under relatively mild thermal conditions, typically between 100-110°C, using methyl tert-butyl ether as a solvent, which is favorable for industrial safety and environmental compliance. The core advantage lies in the ability to construct the pyrrole ring and introduce the halogen substituent simultaneously with high regiocontrol. This not only simplifies the synthetic workflow but also drastically reduces the formation of unwanted isomers, leading to cleaner reaction profiles and higher isolated yields of the target beta-halopyrrole compounds.
Mechanistic Insights into Pd-Catalyzed Ordered Serial Cyclization
The success of this synthesis relies on a sophisticated catalytic cycle initiated by the coordination of the N-substituted aniline with the bivalent palladium catalyst and the diphenyl phosphoric acid ligand. This coordination forms a reactive nitrogen-palladium species that serves as the entry point for the cyclization cascade. Subsequently, the carbon-carbon triple bond of the first alkyne halide molecule undergoes migratory insertion into the palladium-nitrogen bond, generating a key alkenyl palladium intermediate. This step is crucial as it sets the stage for the subsequent bond-forming events that define the pyrrole skeleton. The presence of the specific ligand and the mixed base system (lithium hydroxide monohydrate and sodium acetate) plays a vital role in stabilizing these intermediates and facilitating the necessary proton transfers without deactivating the catalyst.
Following the initial insertion, the alkenyl palladium species undergoes oxidative addition with a second molecule of the alkyne halide. This unique step distinguishes the process from standard coupling reactions, as it incorporates a second alkyne unit into the growing molecular framework. The resulting complex then undergoes reductive elimination to form an alkyne enamine intermediate. Finally, under the influence of bromide ions and the palladium species remaining in the system, an intramolecular cyclization occurs to close the ring, yielding the final beta-halopyrrole product. This mechanistic pathway ensures that the halogen atom is installed specifically at the beta-position, providing exceptional control over the impurity profile and eliminating the need for extensive chromatographic separation of regioisomers often seen in electrophilic aromatic substitution methods.
How to Synthesize Beta-Halopyrrole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to stoichiometry and reaction parameters to maximize yield and purity. The process begins by charging a reactor with the substrate N-substituted aniline, a significant excess of the alkyne halide (molar ratio of 6.0-7.0:1), and the palladium chloride catalyst. The addition of the diphenyl phosphoric acid ligand and the mixed alkali base is critical for driving the reaction to completion. The mixture is then heated to 100-110°C and stirred for a prolonged period, typically 24-30 hours, to ensure full conversion. Upon completion, the workup involves a standard aqueous extraction followed by drying and concentration. The crude product is then purified using column chromatography with petroleum ether or a petroleum ether/ethyl acetate mixture. For detailed standardized operating procedures and specific troubleshooting tips, please refer to the technical guide below.
- Charge reactor with N-substituted aniline, alkyne halide, PdCl2 catalyst, ligand, mixed base, and MTBE solvent.
- Stir the reaction mixture at 100-110°C for 24-30 hours to facilitate ordered serial cyclization.
- Cool, extract with ethyl acetate, dry over magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers substantial strategic benefits by addressing key pain points associated with the sourcing of complex heterocyclic intermediates. The reliance on readily available commodity chemicals, such as substituted anilines and phenyl bromoacetylene, mitigates the risk of raw material shortages and price volatility often associated with exotic reagents. Furthermore, the use of methyl tert-butyl ether as a solvent aligns with green chemistry principles, reducing the environmental burden and simplifying waste disposal protocols compared to processes requiring chlorinated solvents or heavy metal oxidants. The robustness of the reaction conditions also implies a lower risk of batch failure, ensuring a more reliable supply continuity for downstream manufacturing operations.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the elimination of multi-step precursor synthesis. By constructing the beta-halopyrrole core directly from simple building blocks, manufacturers can bypass several expensive and time-consuming synthetic stages. The high selectivity of the reaction minimizes the loss of valuable starting materials to side products, thereby improving the overall atom economy. Additionally, the use of a common palladium salt catalyst at relatively low loading levels helps to keep catalyst costs manageable, while the simplified purification process reduces the consumption of silica gel and solvents during the isolation phase.
- Enhanced Supply Chain Reliability: The broad substrate scope demonstrated in the patent examples indicates that this method is not limited to a single specific compound but is a platform technology applicable to a wide range of derivatives. This flexibility allows suppliers to rapidly adapt to changing customer demands for different substituted pyrroles without needing to develop entirely new processes for each variant. The operational safety of the procedure, which avoids high-pressure hydrogenation or cryogenic conditions, further enhances supply chain resilience by reducing the likelihood of unplanned shutdowns due to safety incidents or equipment failures.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or tonnage levels is facilitated by the homogeneous nature of the reaction mixture and the moderate temperature requirements. The absence of hazardous oxidants like DDQ or NBS in large quantities reduces the regulatory hurdles associated with handling dangerous goods. Moreover, the straightforward workup procedure involving liquid-liquid extraction and standard chromatography is easily transferable to industrial-scale separation units, ensuring that the high purity achieved in the lab can be maintained in commercial production without compromising environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing pipelines and for procurement specialists assessing the long-term viability of the supply source.
Q: What are the advantages of this Pd-catalyzed method over traditional NBS halogenation?
A: Unlike traditional NBS halogenation which often suffers from poor selectivity and multiple substituted byproducts, this novel method utilizes an ordered serial cyclization strategy. This ensures high regioselectivity for the beta-position and avoids the formation of complex mixtures, significantly simplifying downstream purification.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly suitable for scale-up. It employs readily available raw materials like N-substituted anilines and common solvents such as methyl tert-butyl ether. The reaction conditions are mild (100-110°C) and operationally safe, avoiding hazardous reagents or extreme pressures often found in alternative synthetic routes.
Q: What is the substrate scope regarding functional group tolerance?
A: The protocol demonstrates excellent functional group tolerance. It successfully accommodates various substituents on the aniline ring, including methyl, phenoxy, fluoro, chloro, and even sensitive groups like aldehydes. This versatility allows for the construction of a diverse library of highly functionalized beta-halopyrrole derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Halopyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation pharmaceuticals and functional materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering beta-halopyrrole compounds that meet stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical techniques to verify identity and assay. Our capability to handle complex palladium-catalyzed transformations allows us to offer this specific technology to clients seeking reliable alternatives to traditional halogenation methods.
We invite potential partners to engage with our technical procurement team to discuss how this synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this more efficient route. We encourage you to contact us today to obtain specific COA data for our available inventory and to receive comprehensive route feasibility assessments that will help accelerate your development timelines and secure your supply chain for the future.
