Advanced Palladium-Catalyzed Synthesis of Beta-Halopyrroles for Scalable Pharmaceutical Manufacturing
Introduction to Next-Generation Beta-Halopyrrole Synthesis
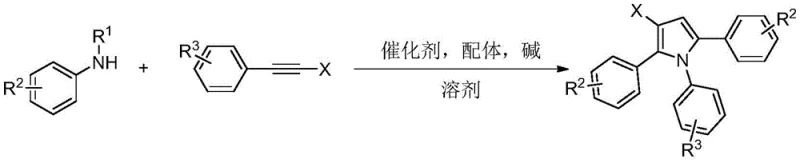
The landscape of heterocyclic chemistry is continuously evolving, driven by the demand for more efficient and selective synthetic routes for bioactive scaffolds. A significant breakthrough in this domain is detailed in patent CN114989063A, which discloses a robust synthesis method for beta-halogenated pyrrole compounds. This technology represents a paradigm shift from classical halogenation strategies, leveraging a novel palladium-catalyzed ordered serial cyclization reaction. By utilizing N-substituted aniline and two molecules of alkyne halide as the primary building blocks, this method constructs highly functionalized beta-halopyrrole cores with exceptional precision. For R&D directors and process chemists, this approach offers a compelling alternative to legacy methods, providing a pathway to complex intermediates that are critical in medicinal chemistry and material science applications. The ability to introduce halogen atoms selectively at the beta-position enhances the lipophilicity and electronic properties of the final molecules, making them invaluable precursors for further cross-coupling reactions.
The significance of this patent extends beyond mere academic interest; it addresses critical pain points in the supply chain of fine chemical intermediates. Traditional methods often rely on hazardous reagents or multi-step sequences that erode overall yield and increase waste generation. In contrast, the methodology described in CN114989063A operates under relatively mild conditions, typically between 100°C and 110°C, using methyl tert-butyl ether (MTBE) as a solvent. This operational simplicity translates directly into reduced energy consumption and safer handling protocols, key metrics for any modern manufacturing facility aiming for sustainability and cost-efficiency. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative technologies allows us to offer clients superior quality materials with a significantly reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of beta-halopyrrole skeletons has been fraught with challenges related to regioselectivity and substrate scope. The most classical approach involves the direct halogenation of pyrrole using N-bromosuccinimide (NBS). However, this method is inherently flawed due to the high reactivity of the pyrrole ring, which possesses multiple nucleophilic sites. Consequently, reactions often yield complex mixtures of alpha- and beta-substituted products, necessitating difficult and yield-reducing separation processes. Furthermore, achieving mono-halogenation specifically at the beta-position often requires pre-blocking the alpha-positions with protecting groups, adding unnecessary synthetic steps and cost. Other reported methods, such as intramolecular amine chlorination of allene compounds or oxidative cyclization of N-allylaniline, suffer from their own limitations, including the need for specialized, hard-to-source starting materials or harsh oxidizing agents that limit functional group tolerance.
The Novel Approach
The methodology presented in the patent data introduces a transformative three-component reaction strategy that circumvents these historical bottlenecks. By employing a palladium salt catalyst in conjunction with a diphenylphosphoric acid ligand, the reaction orchestrates an ordered serial cyclization between one molecule of N-substituted aniline and two molecules of alkyne halide. This approach is groundbreaking because it achieves selective conversion of two identical alkyne components within the same reaction system, a feat that is chemically non-trivial. The result is a direct, one-pot construction of the beta-halopyrrole core with high atom economy. The process tolerates a wide array of substituents on both the aniline and the alkyne, including electron-donating and electron-withdrawing groups, thereby enabling the rapid generation of diverse chemical libraries for drug discovery without the need for extensive route scouting.
Mechanistic Insights into Pd-Catalyzed Ordered Serial Cyclization
At the heart of this synthesis lies a sophisticated catalytic cycle driven by palladium chemistry. The reaction initiates with the coordination of the N-substituted aniline to the bivalent palladium species, facilitated by the diphenylphosphoric acid ligand and the basic environment provided by the lithium and sodium salt mixture. This coordination forms a reactive nitrogen-palladium intermediate, which is crucial for the subsequent activation of the alkyne triple bond. The first molecule of alkyne halide undergoes migratory insertion into the palladium-nitrogen bond, generating an alkenyl-palladium species. This step is pivotal as it sets the stereochemistry and connectivity for the final ring structure. The presence of the halogen on the alkyne is not merely a spectator; it plays an active role in the oxidative addition step that follows, allowing the system to incorporate the second molecule of alkyne halide efficiently.
Following the insertion and oxidative addition steps, the system undergoes a reduction and elimination sequence to form an alkyne enamine intermediate. The final cyclization to the beta-halopyrrole is then triggered by the interaction of bromide ions present in the system with the palladium center, closing the five-membered ring with high fidelity. This mechanistic pathway ensures that impurities arising from random polymerization or non-selective halogenation are minimized. For quality control teams, this means a cleaner crude reaction profile, which simplifies downstream purification. The use of a mixed base system, specifically lithium hydroxide monohydrate and sodium acetate, is optimized to maintain the necessary pH balance for catalyst turnover without degrading the sensitive alkyne functionalities. This level of mechanistic control is what allows the process to achieve yields ranging from 40% to over 60% across a broad spectrum of substrates, as evidenced by the experimental data in the patent.
How to Synthesize Beta-Halopyrrole Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and reaction parameters to maximize yield and purity. The process is designed to be operationally simple, utilizing standard laboratory or plant equipment. The key to success lies in the precise molar ratios of the catalyst, ligand, and base relative to the aniline substrate. Detailed standard operating procedures for this synthesis, including specific workup and purification protocols, are outlined below to ensure reproducibility at scale.
- Charge the reactor with N-substituted aniline, alkyne halide, palladium chloride catalyst, diphenylphosphoric acid ligand, mixed base (LiOH/NaOAc), and MTBE solvent.
- Heat the reaction mixture to 100-110°C and stir vigorously for 24-30 hours to facilitate the ordered serial cyclization.
- Cool to room temperature, extract with ethyl acetate, dry over magnesium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis method offers substantial strategic advantages for procurement managers and supply chain heads looking to optimize their sourcing of heterocyclic intermediates. The primary driver of cost reduction is the utilization of readily available, commodity-grade starting materials. N-substituted anilines and haloalkynes are widely produced chemicals with stable supply chains, eliminating the reliance on exotic or custom-synthesized precursors that often carry high price tags and long lead times. Furthermore, the reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment, significantly lowering capital expenditure requirements for manufacturing partners.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from its high step efficiency and simplified purification needs. By constructing the complex beta-halopyrrole scaffold in a single pot from simple precursors, the method eliminates the need for multiple isolation and protection/deprotection steps common in traditional routes. This consolidation of synthetic steps drastically reduces solvent consumption, labor hours, and waste disposal costs. Additionally, the use of a palladium catalyst, while a precious metal, is employed at low loading levels (approximately 10 mol%), and the robust nature of the reaction suggests that catalyst recovery and recycling strategies could be implemented to further drive down the cost of goods sold (COGS) for high-volume production campaigns.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the broad substrate scope of this chemistry. Because the reaction tolerates various functional groups such as halogens, methyls, and aldehydes, manufacturers are not locked into a single specific raw material specification. This flexibility allows for the substitution of alternative aniline or alkyne derivatives if supply disruptions occur, ensuring continuity of supply for critical API intermediates. Moreover, the reaction time of 24 to 30 hours fits well within standard batch processing schedules, allowing for predictable production planning and inventory management without the unpredictability associated with finicky, low-yielding chemistries.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage quantities is facilitated by the use of methyl tert-butyl ether (MTBE) as the solvent, which is a standard industrial solvent with well-understood handling and recovery protocols. The absence of highly toxic reagents or extreme reaction conditions simplifies the environmental health and safety (EHS) profile of the manufacturing process. Waste streams are primarily organic and can be treated using conventional methods, reducing the burden on wastewater treatment facilities. This alignment with green chemistry principles not only ensures regulatory compliance but also enhances the sustainability credentials of the final product, a factor increasingly weighted in vendor selection by major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of beta-halopyrrole compounds using this advanced palladium-catalyzed methodology. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering a transparent view into the capabilities and limitations of the process for potential partners and stakeholders.
Q: What are the key advantages of this Pd-catalyzed method over traditional NBS halogenation?
A: Unlike traditional NBS halogenation which suffers from poor regioselectivity and multiple substitution issues, this novel three-component cyclization utilizes ordered serial reactions to construct the beta-halopyrrole skeleton with high selectivity and functional group tolerance.
Q: Can this synthesis method tolerate diverse functional groups on the aniline substrate?
A: Yes, the method demonstrates excellent substrate universality, successfully accommodating various substituents such as methyl, fluoro, chloro, phenoxy, and even aldehyde groups on the aniline ring without compromising the cyclization efficiency.
Q: Is the process suitable for large-scale industrial production?
A: The process utilizes commercially available raw materials, mild reaction temperatures (100-110°C), and standard solvents like MTBE, making it highly scalable and safe for industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Halopyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics and advanced materials. Our technical team has thoroughly analyzed the synthetic route described in CN114989063A and validated its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of beta-halopyrrole delivered meets the exacting standards required for pharmaceutical applications.
We invite R&D directors and procurement specialists to collaborate with us to leverage this cutting-edge technology for your specific project needs. Whether you require custom synthesis of novel analogs or large-scale supply of established intermediates, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can accelerate your supply chain and reduce your overall manufacturing costs.
