Advanced Palladium-Catalyzed Synthesis of Beta-Halopyrrole Compounds for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Beta-Halopyrrole Compounds for Commercial Scale-Up
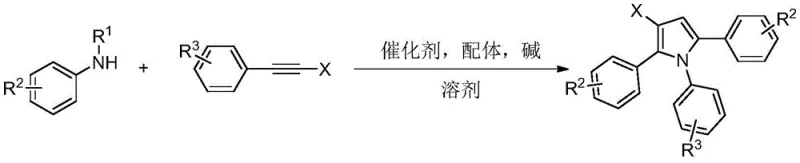
The landscape of organic synthesis for heterocyclic compounds is constantly evolving, driven by the demand for higher purity and more efficient manufacturing routes in the pharmaceutical and material science sectors. A significant breakthrough in this domain is detailed in patent CN114989063A, which discloses a robust synthesis method for beta-halogenated pyrrole compounds. This technology addresses long-standing challenges in constructing functionalized pyrrole skeletons by utilizing a novel three-component reaction strategy. By employing N-substituted aniline and two molecules of alkyne halide in an ordered serial cyclization, the method achieves high selectivity and operational simplicity. For R&D directors and procurement specialists, this represents a pivotal shift away from traditional, less selective halogenation techniques towards a more controlled, catalytic approach that ensures consistent quality and supply chain reliability for critical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of beta-halopyrroles has relied heavily on direct halogenation strategies, such as reacting pyrrole with N-bromosuccinimide (NBS). However, these classical methods suffer from inherent limitations regarding substrate range and selectivity. Due to the presence of multiple reactive sites on the pyrrole ring, direct halogenation frequently results in a mixture of poly-substituted products, making the isolation of a single beta-halopyrrole compound difficult and costly. Alternative approaches, such as intramolecular amine chlorination of allene compounds, often require specific substituents or additional oxidation steps using reagents like DDQ, which complicates the process and increases waste. Furthermore, existing oxidative cyclization reactions between alkyne moieties and N-allylaniline have shown improvements but still struggle with substrate diversity and the complexity of starting material preparation, limiting their utility in broad-scale industrial applications.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN114989063A introduces a highly efficient pathway utilizing palladium catalysis to drive an ordered serial cyclization. This approach leverages the unique reactivity of alkyne halides, which contain both a carbon-carbon triple bond and a carbon-halogen bond capable of participating in transition metal-catalyzed conversions. The reaction systematically combines one molecule of N-substituted aniline with two molecules of alkyne halide, effectively building the pyrrole ring with precise control over the halogen placement. This strategy not only simplifies the raw material sourcing, as N-substituted anilines are commercially abundant, but also drastically improves reaction selectivity. The ability to tolerate various functional groups while maintaining high efficiency makes this a superior choice for synthesizing complex heterocyclic intermediates required in modern drug discovery.
Mechanistic Insights into Pd-Catalyzed Ordered Serial Cyclization
The core of this synthetic innovation lies in its sophisticated catalytic mechanism, which ensures the high fidelity of the final product structure. Under the promotion of a mixed base system, the N-substituted aniline coordinates with the bivalent palladium catalyst and the diphenylphosphoric acid ligand to form a reactive nitrogen-palladium species. This intermediate then undergoes a migratory insertion with the carbon-carbon triple bond of the first alkyne halide molecule, generating an alkenyl palladium species. Subsequently, this species is oxidized and adds another molecule of alkyne halide, leading to the formation of an alkyne enamine through reduction and elimination processes. Finally, the beta-halopyrrole skeleton is constructed via cyclization driven by the interaction of bromide ions and palladium within the system. This stepwise assembly prevents random substitution and ensures that the halogen atom is positioned exactly at the beta-position of the pyrrole ring.
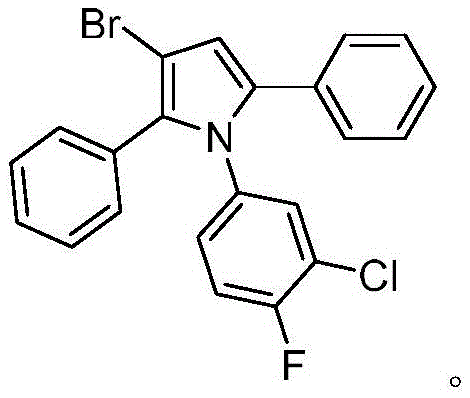
From an impurity control perspective, this mechanism offers distinct advantages over radical-based halogenation. Because the reaction proceeds through defined organometallic intermediates rather than free radical chains, the formation of regio-isomers and poly-halogenated byproducts is significantly suppressed. The use of a specific ligand and base combination further tunes the electronic environment of the catalyst, enhancing tolerance to sensitive functional groups such as aldehydes, fluorines, and chlorines on the aromatic rings. For instance, the successful synthesis of derivatives containing both fluoro and chloro substituents demonstrates the robustness of this catalytic cycle. This level of precision is critical for pharmaceutical applications where impurity profiles must be strictly managed to meet regulatory standards, ensuring that the final API intermediate possesses the necessary chemical integrity for downstream processing.
How to Synthesize Beta-Halopyrrole Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The process begins by charging a reactor with the substrate N-substituted aniline compound, the alkyne halide, a palladium salt catalyst, a ligand, alkali, and a solvent. The reaction is typically conducted in methyl tert-butyl ether (MTBE) at temperatures ranging from 100 to 110°C. Maintaining these conditions for a duration of 24 to 30 hours allows the ordered serial cyclization to proceed to completion. Following the reaction, the mixture is cooled to room temperature, and the product is isolated through standard extraction and purification techniques. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Charge reactor with N-substituted aniline, alkyne halide, PdCl2 catalyst, diphenylphosphoric acid ligand, mixed base (LiOH/NaOAc), and MTBE solvent.
- Stir the reaction mixture at 100-110°C for 24-30 hours to facilitate ordered serial cyclization.
- Cool to room temperature, extract with ethyl acetate, dry over MgSO4, and purify via column chromatography to isolate the target beta-halopyrrole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage stems from the accessibility and stability of the raw materials. N-substituted anilines and alkyne halides are commodity chemicals available from multiple global suppliers, reducing the risk of supply bottlenecks associated with exotic or custom-synthesized starting materials. Furthermore, the reaction conditions are relatively mild, operating at atmospheric pressure and moderate temperatures, which lowers the energy consumption and equipment stress compared to high-pressure hydrogenation or cryogenic processes. This translates directly into a more resilient supply chain capable of sustaining continuous production schedules without frequent interruptions for maintenance or specialized safety protocols.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the elimination of expensive and hazardous halogenating agents like NBS, which often require strict handling and generate significant succinimide waste. By utilizing a catalytic amount of palladium and common bases, the overall reagent cost is optimized. Additionally, the high selectivity of the reaction reduces the burden on downstream purification; fewer byproducts mean less solvent and silica gel are required for column chromatography, leading to substantial cost savings in waste disposal and material usage. The streamlined workflow minimizes the number of unit operations, thereby reducing labor costs and increasing overall plant throughput efficiency.
- Enhanced Supply Chain Reliability: The broad substrate scope of this reaction ensures that supply chains remain flexible even when specific substituted anilines are required. The method tolerates a wide variety of substituents, including methyl, phenoxy, fluoro, and chloro groups, allowing manufacturers to produce a diverse library of intermediates using the same core process infrastructure. This versatility means that production lines do not need extensive retooling to switch between different product grades. Moreover, the use of MTBE as a solvent, which is widely available and easy to recover, further stabilizes the supply chain against solvent shortages, ensuring consistent delivery timelines for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial production is facilitated by its homogeneous nature and lack of gaseous reagents. The absence of toxic gases or highly unstable intermediates simplifies the engineering controls required for large-scale reactors. From an environmental standpoint, the atom economy is improved by incorporating two molecules of alkyne halide into the final structure, minimizing waste generation. The simplified workup procedure, involving basic extraction and drying, reduces the volume of aqueous waste streams. This alignment with green chemistry principles not only lowers environmental compliance costs but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-halopyrrole synthesis technology. These insights are derived directly from the patent specifications and experimental data, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this method improve selectivity compared to traditional NBS halogenation?
A: Traditional NBS halogenation often yields multiple substituted products due to the multiple reactive sites on the pyrrole ring. This novel method utilizes an ordered serial cyclization of N-substituted aniline and two molecules of alkyne halide, which constructs the pyrrole skeleton with high regioselectivity, specifically targeting the beta-position without generating complex mixtures.
Q: What are the key catalyst and ligand systems used in this synthesis?
A: The process employs a common palladium salt, specifically palladium chloride (PdCl2), as the catalyst. It is paired with diphenylphosphoric acid as the ligand. This combination, along with a mixed base system of lithium hydroxide monohydrate and sodium acetate, facilitates the efficient migration and insertion of the alkyne triple bond.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It uses readily available raw materials like N-substituted anilines and operates under relatively mild conditions (100-110°C) in methyl tert-butyl ether (MTBE). The workup involves standard extraction and column chromatography, avoiding hazardous reagents or extreme pressures, making it viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Halopyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics and advanced materials. Our technical team has extensively analyzed the potential of the Pd-catalyzed serial cyclization route described in CN114989063A and is fully prepared to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from pilot studies to full-scale manufacturing. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of beta-halopyrrole meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss your project needs,索取 specific COA data, and obtain comprehensive route feasibility assessments. Let us help you secure a reliable supply of high-purity intermediates while driving efficiency and innovation in your production processes.
