Scalable Glucose-Mediated Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Manufacturing
Scalable Glucose-Mediated Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Manufacturing
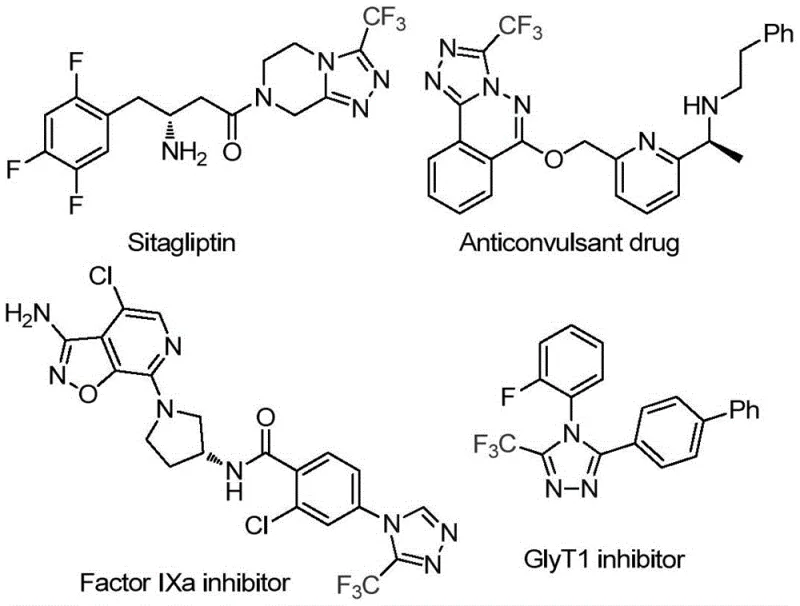
The pharmaceutical industry continuously seeks robust and sustainable pathways for constructing nitrogen-containing heterocycles, particularly the 1,2,4-triazole scaffold which serves as a critical pharmacophore in numerous bioactive molecules. As highlighted in patent CN113880781A, a groundbreaking methodology has emerged that leverages glucose, a ubiquitous biomass resource, as the primary carbon source for generating 3-trifluoromethyl-substituted 1,2,4-triazole compounds. This innovation represents a paradigm shift from traditional petrochemical-derived precursors to renewable feedstocks, addressing both economic and environmental imperatives in modern chemical manufacturing. The significance of this structural motif cannot be overstated, as evidenced by its presence in high-profile therapeutic agents such as Sitagliptin and various anticonvulsant and kinase inhibitors shown in recent literature. By utilizing a trifluoromethanesulfonic acid-catalyzed cascade reaction, this process achieves high efficiency without the need for exotic reagents, positioning it as a highly attractive route for the production of reliable pharmaceutical intermediate supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted triazoles has relied heavily on the condensation of specific aldehydes or nitriles with hydrazine derivatives, often necessitating harsh reaction conditions that pose significant safety and scalability challenges. Traditional protocols frequently demand strictly anhydrous and anaerobic environments to prevent side reactions or catalyst deactivation, which drastically increases operational complexity and infrastructure costs for large-scale production facilities. Furthermore, the reliance on pre-functionalized aldehyde starting materials introduces supply chain vulnerabilities, as these intermediates can be expensive, unstable, or subject to significant price volatility in the global market. The use of heavy metal catalysts or stoichiometric oxidants in older methods also generates substantial toxic waste streams, complicating downstream purification and environmental compliance efforts. These cumulative factors often result in prolonged lead times for high-purity pharmaceutical intermediates and inflated manufacturing costs that ultimately impact the affordability of final drug products.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes glucose, a cheap and abundant carbohydrate, which undergoes acid-catalyzed cleavage to generate the necessary aldehyde species in situ, thereby bypassing the need for isolated aldehyde reagents. This cascade transformation proceeds under remarkably mild conditions, typically requiring temperatures between 70°C and 90°C, and notably tolerates the presence of water, which is actually beneficial for the reaction efficiency rather than detrimental. The elimination of strict moisture control requirements simplifies the engineering controls needed for reactor operations, allowing for more flexible and cost-effective process design. Additionally, the use of tert-butyl hydroperoxide (TBHP) as a terminal oxidant ensures a clean aromatization step with minimal hazardous byproducts, aligning with green chemistry principles. This streamlined workflow not only enhances the overall atom economy but also significantly reduces the number of unit operations required, facilitating a more agile response to market demands for cost reduction in API manufacturing.
Mechanistic Insights into TfOH-Catalyzed Cascade Cyclization
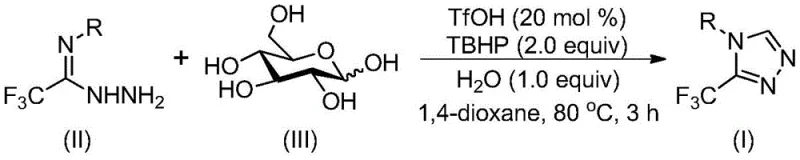
The mechanistic pathway of this transformation is a sophisticated example of acid-promoted biomass valorization, where trifluoromethanesulfonic acid (TfOH) plays a dual role in activating the glucose substrate and promoting subsequent cyclization events. Initially, the strong Brønsted acidity of TfOH facilitates the dehydration and cleavage of the glucose ring structure to yield reactive aldehyde intermediates, which immediately engage in condensation with the trifluoroethylimide hydrazide to form a hydrazone species. This in situ generation of the electrophile prevents the accumulation of unstable aldehydes and drives the equilibrium forward towards the desired intermediate. Following hydrazone formation, an intramolecular nucleophilic addition occurs, closing the five-membered triazole ring system through a concerted cyclization process that is kinetically favored under the reaction conditions. The final step involves oxidative aromatization mediated by TBHP, which removes two hydrogen atoms to establish the fully aromatic 1,2,4-triazole core, locking in the thermodynamic stability of the product. Understanding this cascade sequence is vital for R&D teams aiming to optimize reaction parameters for specific substrate variants.
From an impurity control perspective, the mechanism inherently minimizes the formation of common side products associated with traditional triazole syntheses, such as over-oxidized species or polymerization byproducts often seen with free aldehydes. Because the aldehyde is generated and consumed rapidly within the reaction matrix, its concentration remains low, suppressing competing aldol condensation pathways that typically plague carbohydrate-based syntheses. The specificity of the TfOH catalyst ensures that the cyclization occurs regioselectively to favor the 1,2,4-isomer over potential 1,2,3-triazole contaminants, which is crucial for meeting the stringent purity specifications required for pharmaceutical applications. Moreover, the aqueous compatibility of the system allows for simple workup procedures where water-soluble byproducts and catalyst residues can be easily separated from the organic phase. This inherent selectivity and cleanliness of the reaction profile translate directly into higher crude purity, reducing the burden on downstream purification units and ensuring consistent quality across different production batches.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot-plant validation without requiring specialized equipment. The protocol involves combining the hydrazide precursor, glucose, catalytic amounts of TfOH, and TBHP in a solvent such as 1,4-dioxane, followed by heating to the specified temperature range. The robustness of the reaction allows for a relatively wide operating window, providing process engineers with flexibility in managing heat transfer and mixing dynamics during scale-up. Detailed standardized synthetic steps see the guide below for precise molar ratios and timing.
- Combine trifluoroethylimide hydrazide, glucose, trifluoromethanesulfonic acid catalyst, and tert-butyl hydroperoxide oxidant in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to a temperature range of 70-90°C and maintain stirring for a duration of 2 to 4 hours to ensure complete conversion.
- Upon completion, perform post-processing via filtration and silica gel column chromatography to isolate the pure 3-trifluoromethyl-substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this glucose-mediated technology offers transformative potential in terms of cost structure and supply security. By shifting the carbon source from specialized petrochemical aldehydes to commodity-grade glucose, manufacturers can decouple their production costs from the volatility of the oil market and leverage the stable pricing of agricultural biomass. This fundamental change in raw material sourcing creates a buffer against supply shocks, ensuring continuous availability of critical intermediates even during periods of geopolitical instability or logistics disruptions. Furthermore, the simplified reaction conditions eliminate the need for expensive drying agents and inert gas blanketing systems, resulting in substantial cost savings in utility consumption and capital expenditure for facility upgrades. The ability to operate under ambient atmospheric conditions also reduces the training burden on operational staff and lowers the risk of safety incidents related to pyrophoric reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of high-cost aldehyde starting materials with inexpensive glucose, which is produced on a massive global scale for the food and fermentation industries. Additionally, the high atom efficiency of the cascade reaction means that less raw material is wasted as byproduct, further improving the overall cost-per-kilogram metric for the final active ingredient. The reduction in purification steps due to the clean reaction profile also lowers the consumption of silica gel and solvents, which are significant cost drivers in fine chemical production. These cumulative efficiencies allow for a more competitive pricing strategy without compromising on the quality or purity of the delivered intermediates.
- Enhanced Supply Chain Reliability: Sourcing glucose is inherently more stable than sourcing complex fluorinated aldehydes, as it is a staple commodity with a diversified global supply base that is less susceptible to single-point failures. The robustness of the reaction to water and oxygen means that transportation and storage requirements for the reagents are less stringent, reducing the risk of degradation during transit and expanding the pool of qualified suppliers. This resilience is critical for maintaining just-in-time inventory levels and avoiding production stoppages that can ripple through the entire pharmaceutical value chain. Consequently, partners adopting this technology can offer more reliable delivery schedules and shorter lead times to their downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively at the gram level with clear pathways to kilogram and tonne-scale production using standard stainless steel reactors. The use of water as a beneficial additive and the generation of benign byproducts align perfectly with increasingly rigorous environmental regulations regarding waste discharge and solvent emissions. This eco-friendly profile simplifies the permitting process for new manufacturing lines and enhances the corporate sustainability credentials of the producing organization. The ability to scale up complex pharmaceutical intermediates using such a green methodology positions companies as leaders in sustainable chemistry, appealing to environmentally conscious investors and consumers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this glucose-based triazole synthesis platform. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on substrate compatibility and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of using glucose in this triazole synthesis?
A: Using glucose as a carbon source offers significant sustainability benefits as it is a widely available biomass feedstock. It eliminates the need for complex aldehyde precursors, simplifying the supply chain and reducing raw material costs while maintaining high reaction efficiency under mild conditions.
Q: Does this method require strict anhydrous or anaerobic conditions?
A: No, one of the key operational advantages of this patented process is that it does not require stringent anhydrous or anaerobic environments. The reaction tolerates the presence of water, which is actually added as an additive to improve efficiency, making it highly suitable for standard industrial reactor setups.
Q: What is the substrate scope for the R group in the final triazole product?
A: The method demonstrates broad substrate tolerance, successfully accommodating various substituted aryl groups including those with methyl, methoxy, methylthio, halogen, or trifluoromethyl substituents at ortho, meta, or para positions, as well as phenethyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative synthetic methodologies like the glucose-mediated triazole formation in driving the next generation of pharmaceutical development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are seamlessly translated into robust industrial processes. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging our expertise in process optimization and impurity profiling, we help our clients navigate the complexities of regulatory filings and accelerate their time-to-market for novel therapeutics.
We invite you to collaborate with us to explore how this sustainable synthesis route can enhance your project economics and supply chain resilience. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for efficient and responsible chemical manufacturing.
