Advanced Palladium-Catalyzed Synthesis of Beta-Halopyrrole Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds with high precision and efficiency. A significant breakthrough in this domain is detailed in patent CN114989063A, which discloses a novel synthesis method for beta-halogenated pyrrole compounds. This technology represents a paradigm shift from traditional halogenation techniques by employing an ordered serial cyclization reaction between N-substituted aniline compounds and two molecules of alkyne halides. The process leverages a palladium salt catalyst system to drive the formation of the pyrrole ring, offering a streamlined pathway to access highly functionalized intermediates that are critical in the development of marine alkaloids, bioactive pharmaceutical ingredients, and advanced optoelectronic materials. By addressing the longstanding challenges of selectivity and substrate availability, this invention provides a powerful tool for chemists aiming to diversify their molecular libraries with beta-halopyrrole cores.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of beta-halopyrrole skeletons has relied heavily on direct halogenation strategies using reagents such as N-bromosuccinimide (NBS). While conceptually straightforward, these classical approaches suffer from inherent limitations regarding regioselectivity and substrate scope. The pyrrole ring possesses multiple reactive sites, often leading to the formation of complex mixtures of mono-, di-, and poly-halogenated byproducts, which complicates downstream purification and significantly reduces overall yield. Furthermore, alternative methods involving intramolecular amine chlorination of allene compounds or oxidative cyclization of N-allylaniline often require harsh oxidants like DDQ or specific, hard-to-synthesize starting materials. These constraints not only increase the cost of goods but also limit the structural diversity achievable, making it difficult for procurement teams to source specialized beta-halopyrrole derivatives reliably for drug discovery programs.
The Novel Approach
In stark contrast, the methodology described in the patent introduces a highly efficient three-component coupling strategy that circumvents these traditional bottlenecks. By utilizing simple and commercially available N-substituted anilines and alkyne halides, the reaction constructs the beta-halopyrrole framework through a sophisticated yet operationally simple catalytic cycle. The use of a palladium catalyst in conjunction with a diphenylphosphoric acid ligand enables the selective activation of carbon-halogen and carbon-carbon triple bonds, facilitating an ordered assembly that minimizes side reactions. This approach not only simplifies the synthetic route by eliminating the need for pre-functionalized pyrrole precursors but also enhances safety by operating under mild thermal conditions. The versatility of this system is visually represented in the general reaction scheme below, highlighting its capacity to accommodate diverse substituents.
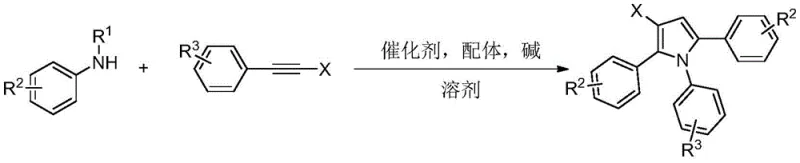
Mechanistic Insights into Pd-Catalyzed Serial Cyclization
The underlying mechanism of this transformation is a testament to the elegance of modern transition metal catalysis. The reaction initiates with the coordination of the N-substituted aniline to the bivalent palladium species, forming a reactive nitrogen-palladium intermediate. This species then undergoes migratory insertion with the carbon-carbon triple bond of the first alkyne halide molecule, generating an alkenyl-palladium complex. Subsequently, this intermediate engages in an oxidative addition with a second molecule of the alkyne halide, followed by reductive elimination to form an alkyne enamine species. The final cyclization step is driven by the presence of bromide ions and the palladium catalyst within the system, closing the ring to yield the target beta-halopyrrole. This multi-step cascade occurs seamlessly in a single pot, demonstrating remarkable atom economy and bond-forming efficiency that is rarely achieved in classical heterocyclic synthesis.
From an impurity control perspective, the ordered nature of this serial cyclization is paramount. Unlike radical halogenation processes that can attack various positions on the pyrrole ring indiscriminately, this palladium-mediated pathway directs the halogen atom specifically to the beta-position through well-defined organometallic intermediates. This intrinsic selectivity drastically reduces the formation of regioisomeric impurities, thereby simplifying the purification workflow. For R&D directors, this means that the resulting crude product typically requires less intensive chromatographic separation to achieve high purity standards. The ability to tolerate a wide array of functional groups—including methyl, fluoro, chloro, and even aldehyde moieties—without compromising the integrity of the catalytic cycle further underscores the robustness of this method for synthesizing complex pharmaceutical intermediates.
How to Synthesize Beta-Halopyrrole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting involves a straightforward protocol that balances reaction efficiency with operational safety. The process begins by charging a reactor with the requisite molar ratios of N-substituted aniline, alkyne halide, palladium chloride catalyst, and the specific ligand system. The reaction is conducted in methyl tert-butyl ether, a solvent chosen for its favorable boiling point and ability to dissolve the organic components effectively. Maintaining the reaction temperature between 100°C and 110°C for a period of 24 to 30 hours ensures complete conversion of the starting materials. Following the reaction, the workup procedure involves standard aqueous extraction and drying steps, followed by purification via column chromatography. For a detailed breakdown of the specific stoichiometric ratios and step-by-step operational parameters, please refer to the standardized guide below.
- Charge the reactor with N-substituted aniline, alkyne halide, palladium chloride catalyst, diphenylphosphoric acid ligand, mixed alkali base, and methyl tert-butyl ether solvent.
- Stir the reaction mixture at a controlled temperature between 100°C and 110°C for a duration of 24 to 30 hours to ensure complete conversion.
- Upon completion, cool to room temperature, extract with ethyl acetate, dry the organic phase, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The reliance on readily available commodity chemicals such as anilines and phenylacetylenes as starting materials significantly de-risks the supply chain, reducing dependency on exotic or custom-synthesized precursors that often suffer from long lead times and price volatility. Furthermore, the operational simplicity of the process—requiring no specialized high-pressure equipment or cryogenic conditions—translates directly into lower capital expenditure and operational costs for manufacturing partners. The use of a common palladium catalyst system, while requiring recovery protocols, is well-understood in the industry, allowing for established recycling workflows that mitigate the impact of precious metal costs on the final price per kilogram.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by its high atom efficiency and the elimination of multi-step precursor synthesis. By constructing the pyrrole ring and installing the halogen functionality in a single concerted operation, the method removes the need for separate halogenation steps that typically generate substantial salt waste and require additional purification resources. Although specific commercial savings percentages depend on volume, the qualitative reduction in unit operations and solvent consumption inherently drives down the cost of goods sold, making high-purity beta-halopyrrole derivatives more accessible for budget-constrained research projects.
- Enhanced Supply Chain Reliability: The broad substrate scope of this reaction acts as a buffer against supply disruptions. If a specific substituted aniline becomes unavailable, the chemistry is robust enough to accommodate alternative analogs with minimal re-optimization, ensuring continuity of supply for downstream API synthesis. Additionally, the mild reaction conditions and the use of stable solvents like methyl tert-butyl ether simplify logistics and storage requirements, reducing the risk of hazardous material incidents that could otherwise halt production lines and delay deliveries to global clients.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage levels is facilitated by its homogeneous nature and the absence of gaseous reagents or extreme pressures. The workup procedure, which utilizes standard liquid-liquid extraction and chromatography, is easily adaptable to continuous processing or large-batch reactors. From an environmental standpoint, the high selectivity of the reaction minimizes the generation of hazardous halogenated byproducts, aligning with increasingly stringent global regulations on waste disposal and green chemistry principles, thereby reducing the environmental compliance burden for manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-halopyrrole synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters. Understanding these nuances is essential for R&D teams evaluating this route for their specific pipeline candidates and for procurement specialists assessing the feasibility of long-term supply agreements.
Q: What are the primary advantages of this synthesis method over traditional NBS halogenation?
A: Unlike traditional NBS halogenation which often suffers from poor selectivity and multiple substituted byproducts, this novel palladium-catalyzed method utilizes an ordered serial cyclization strategy. This ensures high regioselectivity for the beta-position and allows for the construction of highly functionalized pyrrole skeletons from simple, commercially available starting materials without the need for complex pre-functionalization.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed with scalability in mind. It employs mild reaction conditions (100-110°C), uses common solvents like methyl tert-butyl ether, and relies on readily accessible raw materials such as N-substituted anilines and alkyne halides. The operational safety and simple workup procedures involving standard extraction and chromatography make it highly viable for commercial scale-up.
Q: What is the substrate scope for the alkyne halide component?
A: The method exhibits excellent substrate universality. It tolerates a wide range of substituents on the alkyne halide, including electron-donating groups like methyl and electron-withdrawing groups like chloro, fluoro, and aldehyde functionalities. This versatility allows for the efficient synthesis of a diverse library of beta-halopyrrole derivatives tailored for specific medicinal or material science applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Halopyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in accelerating drug discovery and development. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering beta-halopyrrole compounds that meet stringent purity specifications, supported by our rigorous QC labs which employ state-of-the-art analytical instrumentation to verify identity and assay. Our capability to implement advanced palladium-catalyzed processes allows us to offer customized manufacturing solutions that align with your specific project timelines and quality requirements.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be leveraged for your upcoming projects. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this streamlined route for your supply chain. We encourage you to contact us today to obtain specific COA data for our available beta-halopyrrole inventory and to receive comprehensive route feasibility assessments tailored to your unique molecular targets.
