Advanced Palladium-Catalyzed Synthesis of Triazole-Ketone Intermediates for Scalable Pharmaceutical Manufacturing
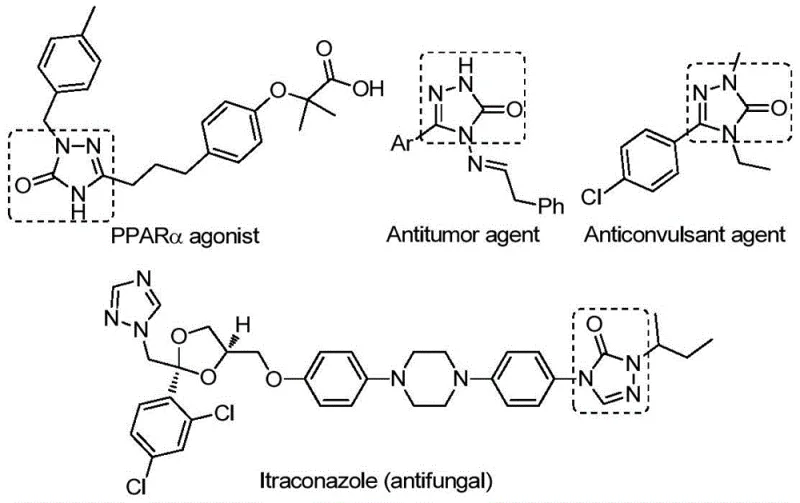
The recently granted Chinese patent CN112538054B introduces a groundbreaking methodology for synthesizing biologically active 1,2,4-triazole-3-one compounds through a novel palladium-catalyzed carbonylation tandem cyclization process that addresses critical limitations in traditional synthetic routes. This innovative approach utilizes readily available starting materials including chlorinated hydrazones and sodium azide under mild reaction conditions at precisely controlled temperatures of 100°C without requiring harsh reagents or complex pre-activation steps that have historically hindered industrial implementation. The patent demonstrates significant advancements in reaction efficiency with excellent substrate compatibility across diverse functional groups while maintaining operational simplicity essential for seamless integration into existing manufacturing workflows. This development holds substantial promise for pharmaceutical manufacturers seeking reliable sources of high-purity triazole intermediates with enhanced biological activity profiles including antifungal, antitumor, and anticonvulsant properties as evidenced by multiple pharmacological applications documented in peer-reviewed literature.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for producing triazole-ketone compounds suffer from multiple significant drawbacks that severely limit their industrial applicability and commercial viability within pharmaceutical manufacturing environments. Conventional methods typically require harsh reaction conditions such as elevated temperatures exceeding 150°C or strong bases like potassium hydroxide that increase operational complexity while creating substantial safety concerns during scale-up operations. Many established routes involve multi-step sequences with intermediate isolations that dramatically reduce overall yield through cumulative losses at each transformation stage while significantly increasing production costs due to additional processing requirements. The necessity for pre-activated substrates in several methodologies creates additional synthetic burdens that restrict structural diversity achievable through these pathways while complicating raw material sourcing strategies. Furthermore, narrow substrate scope in existing methods prevents efficient generation of diverse triazole derivatives needed for comprehensive structure-activity relationship studies in modern drug development programs where molecular diversity is critical for target optimization.
The Novel Approach
The patented methodology overcomes these historical challenges through an elegant palladium-catalyzed carbonylation tandem cyclization process that operates under significantly milder conditions while delivering superior reaction efficiency across diverse substrate classes. By utilizing readily available chlorinated hydrazones and sodium azide as starting materials with a precisely defined palladium catalyst system comprising Pd₂(dba)₃ and Xantphos ligand in anhydrous dioxane solvent at controlled temperature of 100°C, this approach eliminates the need for pre-activation steps and harsh reaction conditions that have plagued traditional methods. The process demonstrates remarkable substrate tolerance across alkyl groups including n-Bu and t-Bu substituents as well as diverse aryl groups containing halogen or methoxy functionalities while maintaining consistent high yields throughout the reaction scope without requiring specialized equipment modifications. This innovative strategy enables direct synthesis of structurally diverse triazole derivatives without intermediate isolations or complex purification procedures through a streamlined single-vessel operation that significantly reduces processing time while enhancing overall manufacturing efficiency.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Cyclization
The reaction mechanism begins with oxidative addition of the palladium catalyst into the carbon-chlorine bond of the chlorinated hydrazone substrate to form a key divalent palladium intermediate that serves as the foundation for subsequent transformations within this cascade process. Subsequently, TFBen decomposes under thermal conditions at precisely controlled temperature of 100°C to release carbon monoxide which inserts into the carbon-palladium bond forming an acyl palladium species that represents a critical intermediate in this synthetic pathway. This intermediate then reacts with sodium azide to generate an acyl azide compound that undergoes Curtius rearrangement to produce an isocyanate intermediate through a well-defined molecular rearrangement process without requiring additional reagents or catalysts. The final step involves intramolecular nucleophilic addition where the hydrazone nitrogen attacks the isocyanate carbon to form the triazole ring system with concomitant release of nitrogen gas as the driving force for ring closure completion.

Impurity control is achieved through precise management of multiple critical parameters including strict temperature control at exactly 100°C which prevents thermal decomposition pathways while maintaining optimal reaction kinetics throughout the transformation sequence. The carefully designed stoichiometric ratios between reactants prevent over-reaction or side product formation by ensuring complete consumption of starting materials without excess reagents that could generate impurities during workup procedures. The use of anhydrous dioxane as solvent eliminates potential hydrolysis side reactions while the specific catalyst system suppresses unwanted dimerization or oligomerization processes that could compromise product purity profiles required for pharmaceutical applications. Post-reaction processing through simple filtration followed by standard column chromatography effectively removes any residual catalyst or unreacted starting materials without requiring specialized purification techniques that would increase manufacturing complexity or cost while ensuring consistent production quality meeting stringent regulatory requirements.
How to Synthesize Triazole-Ketone Efficiently
This patented methodology provides a robust and scalable pathway for producing high-purity triazole-ketone intermediates essential for pharmaceutical applications requiring precise structural specifications across diverse therapeutic areas including antifungal and antitumor drug development programs. The process utilizes commercially available starting materials including chlorinated hydrazones and sodium azide with a well-defined palladium catalyst system that operates under controlled reaction conditions to ensure optimal yield and purity without requiring specialized equipment modifications during scale-up operations. By eliminating multiple synthetic steps required in conventional approaches such as pre-functionalization or intermediate isolations, this method significantly reduces processing time while maintaining excellent substrate flexibility across diverse functional groups including alkyl chains and substituted aryl moieties essential for medicinal chemistry optimization studies.
- Prepare reaction mixture by combining chlorohydrazone substrate (II), sodium azide (2.5 equiv), Pd₂(dba)₃ (2.5 mol%), Xantphos (5 mol%), and TFBen (5 equiv) in anhydrous dioxane under nitrogen atmosphere.
- Heat reaction mixture to precisely controlled temperature of 100°C with continuous stirring for optimal duration of 24 hours to ensure complete conversion through carbonylation tandem cyclization mechanism.
- Perform post-reaction processing by filtration through silica gel followed by standard column chromatography purification to isolate high-purity triazole-ketone product (I) meeting pharmaceutical quality specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial commercial benefits that directly address critical pain points in pharmaceutical intermediate procurement and supply chain management operations where reliability and consistency are paramount concerns for global sourcing strategies. By utilizing readily available starting materials from multiple qualified suppliers worldwide and eliminating complex multi-step sequences required in traditional approaches, this process significantly reduces raw material sourcing challenges while improving overall supply chain resilience against market fluctuations or regional supply disruptions common in specialty chemical markets.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions reduces energy consumption requirements while simplified purification procedures through standard column chromatography instead of complex isolation techniques contribute substantially to cost savings throughout manufacturing operations without requiring expensive metal recovery processes typically associated with transition metal catalysis.
- Enhanced Supply Chain Reliability: Reliance on widely available starting materials including sodium azide and chlorinated hydrazones sourced from multiple global suppliers significantly improves supply chain resilience while reducing dependency on single-source specialty chemicals that often create procurement bottlenecks during scale-up activities.
- Scalability and Environmental Compliance: The methodology demonstrates excellent scalability from laboratory to commercial production with consistent yields across different batch sizes while simplified waste streams through this streamlined process significantly reduce environmental impact compared to traditional multi-step synthetic routes requiring extensive solvent usage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented synthesis methodology for pharmaceutical intermediate production where quality assurance teams require detailed understanding before committing to new supply sources or manufacturing processes.
Q: How does this method improve upon conventional triazole synthesis approaches?
A: The patented methodology eliminates harsh reaction conditions and multi-step sequences required in traditional approaches by utilizing a single-step palladium-catalyzed carbonylation tandem cyclization that operates under milder conditions with broader substrate compatibility across diverse functional groups.
Q: What are the key supply chain advantages for pharmaceutical procurement teams?
A: This process utilizes readily available starting materials from multiple global suppliers while simplifying purification procedures through standard column chromatography, significantly enhancing supply chain reliability without requiring specialized equipment or infrastructure investments.
Q: Can this synthesis be reliably scaled for commercial production?
A: Yes, the methodology demonstrates excellent scalability from laboratory to commercial production as evidenced by consistent yields across different substrate scales with straightforward process parameters that facilitate smooth technology transfer between manufacturing sites.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole-Ketone Supplier
Our patented methodology represents a significant advancement in triazole intermediate synthesis that delivers exceptional value for pharmaceutical manufacturers seeking reliable sources of high-purity building blocks for drug development programs targeting multiple therapeutic areas including antifungal treatments where compounds like itraconazole demonstrate clinical efficacy. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production with stringent purity specifications consistently met through our rigorous QC labs equipped with state-of-the-art analytical instrumentation capable of detecting impurities at trace levels required by regulatory authorities worldwide.
For procurement teams seeking to optimize their supply chain strategy for critical pharmaceutical intermediates essential to drug development pipelines, we invite you to request a Customized Cost-Saving Analysis from our technical procurement team along with specific COA data and route feasibility assessments tailored to your manufacturing requirements and quality specifications.
