Scalable One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Applications
Scalable One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Applications
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activity. Quinazolinone derivatives, characterized by their fused bicyclic structure, are ubiquitous in pharmacologically active molecules, serving as key motifs in drugs with antifungal, antibacterial, antiviral, and anticancer properties. Notable examples include Methaqualone and Afloqualone, which highlight the therapeutic potential of this chemical class. 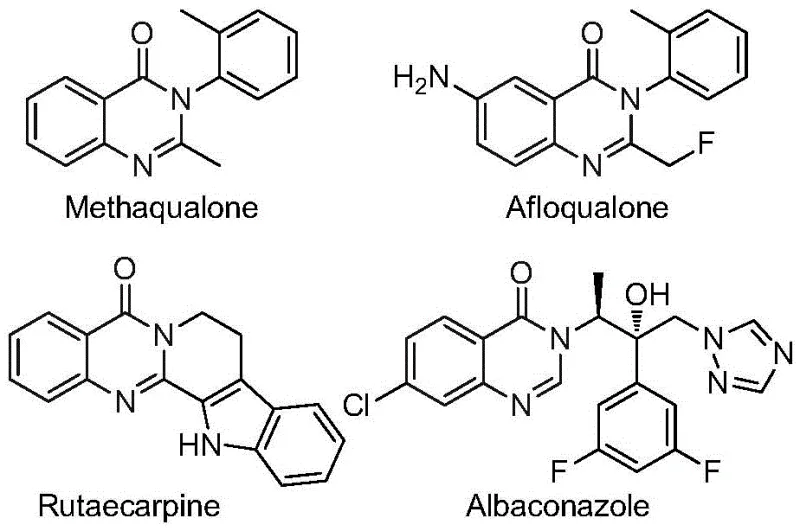 . Recent advancements disclosed in patent CN112480015B introduce a transformative multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. This technology leverages a palladium-catalyzed carbonylation cascade, utilizing inexpensive nitro compounds and trifluoroethylimidoyl chloride as starting materials. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this methodology represents a significant leap forward in process efficiency, offering a robust pathway to high-purity fluorinated heterocycles essential for next-generation drug discovery.
. Recent advancements disclosed in patent CN112480015B introduce a transformative multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. This technology leverages a palladium-catalyzed carbonylation cascade, utilizing inexpensive nitro compounds and trifluoroethylimidoyl chloride as starting materials. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this methodology represents a significant leap forward in process efficiency, offering a robust pathway to high-purity fluorinated heterocycles essential for next-generation drug discovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied on methodologies that present substantial logistical and economic challenges for large-scale manufacturing. Traditional synthetic strategies often necessitate the use of high-pressure carbon monoxide gas, which requires specialized autoclave equipment and rigorous safety protocols due to the extreme toxicity and flammability of CO. Furthermore, many established routes depend on pre-activated substrates, such as 2-bromoformylaniline or acid anhydrides, which are significantly more expensive and less atom-economical than simple nitroarenes. Alternative methods involving iron or ruthenium catalysis frequently suffer from narrow substrate scope, harsh reaction conditions, or low yields, limiting their utility in the commercial scale-up of complex pharmaceutical intermediates. These factors collectively contribute to elevated production costs and extended lead times, creating bottlenecks in the supply chain for critical API precursors.
The Novel Approach
The innovative protocol described in the patent data overcomes these historical barriers through a cleverly designed tandem reaction sequence. By employing Molybdenum Hexacarbonyl (Mo(CO)6) as a solid carbon monoxide surrogate, the process eliminates the need for handling hazardous high-pressure CO gas, thereby drastically simplifying the reactor setup and enhancing operational safety. The reaction utilizes readily available nitro compounds and trifluoroethylimidoyl chloride, which undergo a palladium-catalyzed cascade to form the target quinazolinone structure in a single pot. 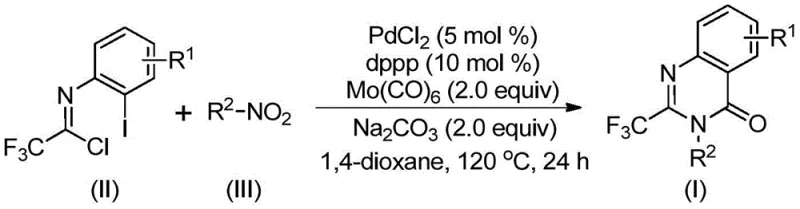 . This approach not only reduces the number of synthetic steps but also improves overall atom economy. The use of a PdCl2/dppp catalytic system ensures high turnover and excellent functional group tolerance, allowing for the synthesis of diverse derivatives without the need for protecting group strategies. This streamlined workflow translates directly into cost reduction in pharmaceutical intermediate manufacturing, making it an attractive option for industrial adoption.
. This approach not only reduces the number of synthetic steps but also improves overall atom economy. The use of a PdCl2/dppp catalytic system ensures high turnover and excellent functional group tolerance, allowing for the synthesis of diverse derivatives without the need for protecting group strategies. This streamlined workflow translates directly into cost reduction in pharmaceutical intermediate manufacturing, making it an attractive option for industrial adoption.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
A deep understanding of the reaction mechanism is crucial for R&D teams aiming to optimize this process for specific analogs. The transformation initiates with the reduction of the nitro group in the starting material to an amine species, mediated by the Mo(CO)6 complex under thermal conditions. This generated amine subsequently undergoes a base-promoted intermolecular coupling with the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate in situ. Following this, the palladium catalyst inserts into the carbon-iodine bond of the imidoyl chloride moiety, generating a divalent palladium species. As the temperature is maintained at 120°C, the Mo(CO)6 releases carbon monoxide, which inserts into the carbon-palladium bond to form an acyl-palladium intermediate. This key step is followed by intramolecular cyclization, where the nitrogen atom attacks the acyl center, facilitated by the base, to form a seven-membered palladacycle. Finally, reductive elimination releases the 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium catalyst, completing the cycle.
The exceptional substrate compatibility of this method is evidenced by the successful synthesis of various derivatives bearing different electronic and steric properties. 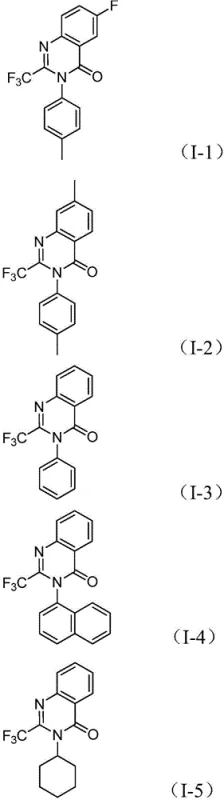 . The system tolerates electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens and trifluoromethyl groups on the aromatic rings. This broad scope is vital for medicinal chemists exploring structure-activity relationships (SAR), as it allows for the rapid generation of diverse libraries. Furthermore, the mechanism inherently controls impurity profiles by favoring the desired cyclization pathway over potential side reactions, ensuring high purity of the final product. The ability to fine-tune the reaction by adjusting the ligand environment (using dppp) and the base (sodium carbonate) provides additional levers for process optimization, ensuring consistent quality across different batches.
. The system tolerates electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens and trifluoromethyl groups on the aromatic rings. This broad scope is vital for medicinal chemists exploring structure-activity relationships (SAR), as it allows for the rapid generation of diverse libraries. Furthermore, the mechanism inherently controls impurity profiles by favoring the desired cyclization pathway over potential side reactions, ensuring high purity of the final product. The ability to fine-tune the reaction by adjusting the ligand environment (using dppp) and the base (sodium carbonate) provides additional levers for process optimization, ensuring consistent quality across different batches.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise control over reaction parameters to maximize yield and purity. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO source, and substrates in an aprotic solvent such as 1,4-dioxane. The mixture is then heated to facilitate the cascade reaction. Detailed standard operating procedures regarding stoichiometry, addition order, and workup techniques are critical for reproducibility. For a comprehensive guide on executing this transformation, please refer to the standardized synthesis steps outlined below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 16 to 30 hours to allow the carbonylation cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers compelling advantages that address key pain points in the chemical supply chain. The shift from high-pressure gas systems to solid reagents significantly lowers the barrier to entry for manufacturing facilities, reducing capital expenditure on specialized equipment. Moreover, the reliance on commodity chemicals like nitro compounds and simple imidoyl chlorides ensures a stable and cost-effective raw material supply, mitigating risks associated with volatile pricing of exotic precursors.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide infrastructure results in substantial operational savings. By using Mo(CO)6 as a safe CO source, manufacturers avoid the costs associated with gas cylinder logistics, leak detection systems, and high-specification reactors. Additionally, the one-pot nature of the reaction minimizes solvent usage and waste generation, leading to a greener and more economical process. The high yields reported across various substrates further enhance the cost-efficiency by maximizing the output per unit of raw material input.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, particularly nitro compounds, are widely available from global chemical suppliers, ensuring a robust supply chain. Unlike methods requiring custom-synthesized or unstable intermediates, this route relies on shelf-stable reagents that can be sourced in bulk quantities. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream pharmaceutical clients. The simplicity of the post-treatment process, involving filtration and standard chromatography, also accelerates the turnaround time from reaction completion to product shipment.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be scalable, with the potential for expansion from gram to kilogram scales without significant loss in efficiency. The use of 1,4-dioxane as a solvent, while requiring proper handling, is a well-established industrial solvent with known recovery and recycling protocols. The avoidance of toxic heavy metal waste streams associated with some alternative reduction methods simplifies environmental compliance and waste disposal. This alignment with green chemistry principles makes the process attractive for companies aiming to reduce their environmental footprint while maintaining high production standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, offering clarity for technical teams evaluating this route for their specific projects.
Q: What are the advantages of using nitro compounds over amines in this synthesis?
A: Nitro compounds are generally cheaper and more readily available than their corresponding amines. This method utilizes the nitro group as both a precursor for the amine (via reduction) and a directing group, streamlining the synthetic route and reducing raw material costs.
Q: Is high-pressure carbon monoxide gas required for this reaction?
A: No, high-pressure CO gas is not required. The protocol uses Molybdenum Hexacarbonyl (Mo(CO)6) as a solid carbon monoxide surrogate, which releases CO in situ under heating. This significantly enhances operational safety and simplifies equipment requirements compared to traditional carbonylation methods.
Q: What is the substrate scope for the R2 group in the nitro compound?
A: The method demonstrates excellent compatibility with various substituents. R2 can be C1-C10 alkyl, cycloalkyl, or substituted/unsubstituted aryl groups. Electron-donating and electron-withdrawing groups on the aryl ring, such as methyl, methoxy, halogens, and trifluoromethyl, are well-tolerated.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic methodologies in driving pharmaceutical innovation. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned to industrial reality. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement advanced catalytic processes, such as the palladium-catalyzed carbonylation described herein, positions us as a strategic partner for your complex synthesis needs.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your project timelines while optimizing your manufacturing costs.
