Advanced Primary Amine-Directed Synthesis of 2-Alkynyl Indoles for Commercial API Production
Advanced Primary Amine-Directed Synthesis of 2-Alkynyl Indoles for Commercial API Production
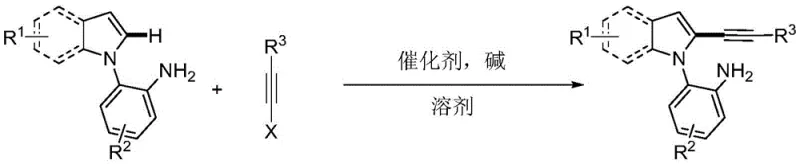
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign methodologies for constructing complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN108864164B, which discloses a novel synthesis method for primary amine-guided 2-alkynyl indole compounds. This technology addresses long-standing challenges in regioselective C-H functionalization by leveraging a simple yet effective palladium-catalyzed cross-coupling strategy. By utilizing 2-(1H-indol-1-yl)aniline derivatives and alkyne halides in an aqueous medium, this process achieves high atom economy and exceptional selectivity. For R&D directors and procurement specialists, this patent represents a pivotal shift towards greener, more cost-efficient manufacturing of high-value pharmaceutical intermediates, eliminating the need for hazardous halogenation pre-steps and toxic organic solvents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of functionalized indole derivatives, particularly at the C2 position, has been fraught with synthetic inefficiencies and selectivity issues. The classical approach typically involves a multi-step sequence starting with C-H halogenation of the indole core, followed by a transition-metal catalyzed cross-coupling reaction. This conventional pathway is inherently flawed due to the higher electron cloud density at the C3 position of the indole ring compared to the C2 position, making C3-functionalization kinetically favored and difficult to suppress without elaborate protecting group strategies. Furthermore, these traditional methods often rely on stoichiometric amounts of hazardous halogenating agents and require anhydrous, toxic organic solvents, which escalate both the environmental footprint and the operational costs associated with solvent recovery and waste disposal. The necessity for pre-functionalization also reduces the overall atom economy, generating significant chemical waste before the final coupling even occurs.
The Novel Approach
In stark contrast, the methodology described in patent CN108864164B introduces a direct, one-pot alkynylation strategy that bypasses the need for pre-halogenation. By employing a primary amine moiety on the aniline ring as an intrinsic directing group, the reaction achieves precise coordination with the palladium catalyst, effectively overriding the natural electronic bias of the indole ring to favor C2-selectivity. This innovative approach not only simplifies the synthetic route by merging multiple steps into a single transformation but also dramatically improves safety profiles by operating under mild conditions. The use of water as a solvent, or mixed systems with toluene, aligns perfectly with the principles of green chemistry, offering a sustainable alternative to volatile organic compounds. This shift allows for the direct construction of highly functionalized 2-alkynyl indoles from readily available starting materials, streamlining the supply chain for complex heterocyclic intermediates.
Mechanistic Insights into Pd-Catalyzed C-H Alkynylation
The success of this synthesis hinges on a sophisticated mechanistic pathway driven by the coordination chemistry of palladium. Under the promotion of a suitable base, such as cesium pivalate or potassium acetate, the primary amine group on the 2-(1H-indol-1-yl)aniline substrate acts as a powerful ligand. It coordinates with the palladium salt catalyst to form a thermodynamically stable six-membered palladacycle intermediate. This cyclometallation step is the critical determinant of regioselectivity, as it positions the metal center in immediate proximity to the C2-H bond of the indole ring, facilitating selective activation. Once the C-H bond is activated, the alkyne halide undergoes oxidative addition to the palladium center. Subsequent reductive elimination releases the desired 2-alkynyl indole product and regenerates the active catalytic species, completing the cycle. This mechanism ensures that even with diverse substituents on the indole or aniline rings, the reaction proceeds with high fidelity and minimal formation of C3-isomers or homocoupling byproducts.
From an impurity control perspective, this mechanism offers distinct advantages for industrial scale-up. The reliance on a specific directing group interaction minimizes non-specific background reactions that often plague radical-based or un-directed C-H activation processes. The use of water as a solvent further aids in impurity management, as many organic byproducts and unreacted starting materials can be easily separated via extraction, while inorganic salts remain in the aqueous phase. The tolerance for various functional groups, including halogens (fluoro, chloro), electron-donating groups (methyl, methoxy), and electron-withdrawing groups (cyano), demonstrates the robustness of the catalytic system. This broad substrate scope implies that a single optimized platform can be adapted to synthesize a wide library of analogues, reducing the R&D burden when exploring structure-activity relationships (SAR) for new drug candidates.
How to Synthesize 2-Alkynyl Indole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and purity. The process begins with the precise charging of substrates, specifically the 2-(1H-indol-1-yl)aniline derivative and the triisopropylsilyl-protected alkyne halide, into a reactor equipped with efficient stirring. The choice of palladium catalyst, such as palladium tetranitrate tetrafluoroborate or palladium acetate, along with the appropriate cesium or potassium base, is critical for initiating the catalytic cycle. The reaction is typically conducted at temperatures ranging from 80°C to 110°C, allowing sufficient thermal energy to overcome the activation barrier for C-H bond cleavage while maintaining the stability of the sensitive alkyne functionality. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are essential for reproducibility.
- Charge a reactor with 2-(1H-indol-1-yl)aniline substrate, alkyne halide, palladium salt catalyst, alkali base, and water solvent.
- Stir the reaction mixture at elevated temperatures between 80°C and 110°C for 12 to 24 hours to facilitate cross-coupling.
- Cool the reaction, extract with ethyl acetate, dry the organic phase, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible strategic advantages beyond mere chemical novelty. The primary benefit lies in the drastic simplification of the manufacturing workflow. By eliminating the pre-halogenation step and utilizing water as the primary solvent, the process significantly reduces the consumption of raw materials and the volume of hazardous waste generated. This reduction in process complexity directly correlates to lower operational expenditures (OPEX), as fewer unit operations are required, and the costs associated with solvent purchase, recovery, and disposal are minimized. Furthermore, the use of commercially available and inexpensive starting materials, such as o-iodoaniline-derived precursors and simple alkyne halides, ensures a stable and cost-effective supply chain, mitigating risks associated with sourcing exotic or proprietary reagents.
- Cost Reduction in Manufacturing: The economic impact of switching to this aqueous, direct-alkynylation method is profound. Traditional methods often incur high costs due to the use of expensive, anhydrous organic solvents and the need for rigorous moisture exclusion protocols. By contrast, this method's compatibility with water eliminates the need for costly drying agents and inert atmosphere equipment for the bulk solvent. Additionally, the high atom economy means that a greater proportion of the input mass ends up in the final product, reducing the cost per kilogram of the active pharmaceutical ingredient (API) intermediate. The removal of transition metal scavenging steps, often required to meet strict residual metal limits in pharma, is also facilitated by the clean reaction profile, further driving down purification costs.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the broad substrate tolerance and the use of commodity chemicals. Since the reaction works effectively with a wide range of substituted indoles and anilines, manufacturers are not locked into a single, fragile supply line for highly specific precursors. The ability to source generic building blocks from multiple vendors reduces the risk of supply disruptions. Moreover, the mild reaction conditions (80-110°C) and the use of non-corrosive aqueous media extend the lifespan of standard stainless steel reactors, reducing capital expenditure on specialized corrosion-resistant equipment and minimizing downtime for maintenance, thereby ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces safety and environmental hurdles, but this methodology is inherently designed for scalability. The use of water as a solvent provides excellent heat transfer properties, which is crucial for managing exothermic reactions on a large scale, thus enhancing process safety. From a regulatory standpoint, the alignment with green chemistry principles facilitates easier approval from environmental agencies. The reduction in volatile organic compound (VOC) emissions simplifies compliance with increasingly stringent environmental regulations, avoiding potential fines and production stoppages. This eco-friendly profile also enhances the brand value of the final pharmaceutical product, appealing to stakeholders who prioritize sustainability in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this primary amine-directed synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the primary amine directing group significant in this synthesis?
A: The primary amine group coordinates with the palladium catalyst to form a stable six-membered ring intermediate, ensuring high regioselectivity for C2-alkynylation over the naturally more reactive C3 position of the indole ring.
Q: What are the environmental benefits of this patented method?
A: Unlike traditional methods requiring toxic organic solvents, this process utilizes water as the primary reaction medium, significantly reducing volatile organic compound (VOC) emissions and simplifying waste treatment protocols.
Q: Can this method tolerate diverse functional groups on the indole scaffold?
A: Yes, the method demonstrates broad substrate universality, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as fluoro, chloro, and cyano substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkynyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies like the one described in CN108864164B for the next generation of pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial realities. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, including rigorous QC labs capable of detecting trace metal residues to meet stringent purity specifications. We are committed to delivering high-purity 2-alkynyl indole compounds that adhere to the highest quality standards required by global regulatory bodies.
We invite R&D directors and procurement leaders to collaborate with us to leverage this efficient synthesis route for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this green chemistry approach can optimize your budget. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary candidates, ensuring a reliable and competitive supply chain for your critical drug development programs.
