Advanced Catalytic Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Scalable Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries continuously seek robust methodologies for constructing nitrogen-containing heterocycles, particularly the 1,2,4-triazole scaffold, which serves as a critical pharmacophore in numerous bioactive molecules. As highlighted in recent intellectual property developments, specifically patent CN113307778A, a significant breakthrough has been achieved in the preparation of 3-trifluoromethyl substituted 1,2,4-triazole compounds. This innovation addresses long-standing challenges in synthetic efficiency and substrate versatility, offering a streamlined pathway to access high-value intermediates used in drugs like Sitagliptin and various agrochemical agents. The introduction of the trifluoromethyl group is strategically vital, as it enhances metabolic stability, lipophilicity, and bioavailability of the parent molecule, making these compounds highly desirable for modern drug discovery pipelines. For R&D directors and procurement specialists, understanding this novel catalytic system is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering complex fluorinated heterocycles with consistent quality.
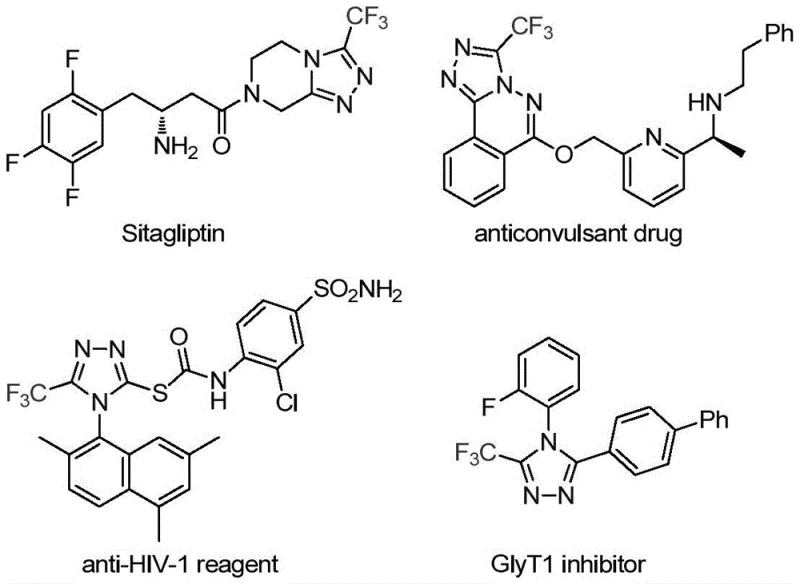
The limitations of conventional methods for synthesizing trifluoromethyl substituted 1,2,4-triazoles have historically hindered efficient large-scale production. Traditional approaches often rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl substituted 1,2,4-oxazolinone, processes that frequently require harsh conditions and generate significant waste. Furthermore, copper-catalyzed multi-component reactions involving diazonium salts and trifluorodiazoethane, while effective, often suffer from safety concerns regarding the handling of unstable diazo species and limited substrate scope. These legacy methods typically struggle to produce fully substituted 1,2,4-triazoles efficiently, and reports on synthesizing 3,4-disubstituted variants are notably scarce, creating a bottleneck for medicinal chemists designing next-generation therapeutics. The reliance on expensive or hazardous reagents in these older protocols also complicates the supply chain, increasing the cost reduction in API manufacturing efforts and posing environmental compliance risks.
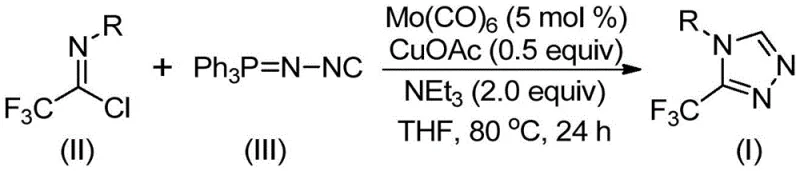
In stark contrast, the novel approach detailed in the patent utilizes a synergistic molybdenum and copper co-catalytic system to drive a [3+2] cycloaddition reaction between trifluoroethylimidoyl chloride and functionalized isonitriles. This method operates under remarkably mild conditions, typically between 70°C and 90°C, utilizing common organic solvents like THF, which drastically simplifies the operational complexity. The reaction mechanism involves the activation of the functionalized isonitrile by molybdenum hexacarbonyl to form a metal complex, followed by a copper-promoted cycloaddition to the imidoyl chloride, ultimately yielding the target triazole after the elimination of triphenylphosphine oxide. This pathway not only avoids the use of dangerous diazonium salts but also exhibits exceptional functional group tolerance, accommodating electron-donating and electron-withdrawing groups alike. By leveraging cheap and easily obtainable starting materials, this technology represents a paradigm shift towards greener and more economical chemical synthesis.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The mechanistic elegance of this transformation lies in the dual-metal activation strategy that facilitates the construction of the five-membered triazole ring with high regioselectivity. Molybdenum hexacarbonyl acts as a crucial metal activator, coordinating with the functionalized isonitrile to enhance its nucleophilicity and stabilize reactive intermediates during the cycle. Simultaneously, cuprous acetate serves as an efficient catalyst for the cycloaddition step, promoting the attack of the activated isonitrile species onto the electrophilic carbon of the trifluoroethylimidoyl chloride. This cooperative catalysis ensures that the reaction proceeds smoothly to form the five-membered ring intermediate, which subsequently undergoes hydrolysis or elimination to release the final 3-trifluoromethyl-substituted 1,2,4-triazole product. The presence of triethylamine as a base further aids in scavenging protons and driving the equilibrium towards product formation, ensuring high conversion rates even with sterically hindered substrates.
From an impurity control perspective, this mechanism offers distinct advantages over traditional acid-mediated cyclizations. The mild basic conditions and the specific coordination chemistry involved minimize side reactions such as polymerization of the isonitrile or hydrolysis of the sensitive imidoyl chloride prior to cyclization. The patent data indicates that the reaction tolerates a wide array of substituents on the aromatic ring, including halogens, alkyl groups, and nitro groups, without significant degradation in yield. This robustness implies a cleaner crude reaction profile, reducing the burden on downstream purification processes like column chromatography. For quality control teams, this means a more predictable impurity profile and easier validation of the manufacturing process, which is critical for meeting the stringent purity specifications required for GMP-grade active pharmaceutical ingredients.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this synthesis effectively, operators should follow a standardized protocol that maximizes catalyst turnover and minimizes exposure to moisture, which can deactivate the metal carbonyl species. The process begins by charging a reaction vessel with the catalytic system comprising molybdenum hexacarbonyl and cuprous acetate in an anhydrous aprotic solvent such as THF. Subsequently, the trifluoroethylimidoyl chloride and the functionalized isonitrile are introduced, typically with a slight excess of the isonitrile to ensure complete consumption of the halide. The detailed standardized synthesis steps see the guide below.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Mo/Cu-catalyzed methodology presents substantial opportunities for optimizing the sourcing of high-purity OLED material precursors and pharmaceutical intermediates. The primary economic driver is the utilization of commercially available and inexpensive starting materials, such as aromatic amines and trifluoroacetic acid derivatives, which are produced on a massive global scale. Unlike legacy methods that require specialized, high-cost reagents like trifluorodiazoethane, this route relies on stable solids and liquids that are easy to transport and store, thereby enhancing supply chain reliability and reducing the risk of production stoppages due to raw material shortages. The simplicity of the workup procedure, involving basic filtration and chromatography, further contributes to operational efficiency.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts beyond the catalytic amounts of Mo and Cu, combined with the use of cheap bases like triethylamine, leads to significant cost savings in reagent procurement. Furthermore, the high reaction efficiency and yields reported (often exceeding 90% for optimized substrates) mean that less raw material is wasted, directly lowering the cost of goods sold (COGS). The avoidance of cryogenic conditions or high-pressure equipment also reduces energy consumption and capital expenditure on specialized reactor infrastructure, making this a highly attractive option for cost-sensitive bulk manufacturing.
- Enhanced Supply Chain Reliability: Since the key reagents trifluoroethylimidoyl chloride and functionalized isonitriles can be synthesized from widely available commodity chemicals, the supply chain is less vulnerable to geopolitical disruptions or single-source supplier failures. The robustness of the reaction conditions allows for flexible scheduling and batch processing, ensuring consistent delivery timelines for downstream customers. This reliability is paramount for maintaining continuous production lines in the fast-paced pharmaceutical sector, where delays in intermediate supply can halt entire drug development programs.
- Scalability and Environmental Compliance: The patent explicitly notes that the method can be expanded to gram-level and potentially larger scales without loss of efficiency, indicating strong potential for commercial scale-up of complex polymer additives or drug intermediates. The use of THF as a solvent, which is readily recyclable, and the generation of benign byproducts like triphenylphosphine oxide, align with modern green chemistry principles. This facilitates easier waste management and regulatory compliance, reducing the environmental footprint of the manufacturing process and avoiding costly fines or remediation efforts associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for their own production needs. Understanding these nuances is critical for assessing the feasibility of integrating this chemistry into existing manufacturing workflows.
Q: What are the key advantages of the Mo/Cu co-catalyzed method for triazole synthesis?
A: The method described in patent CN113307778A offers mild reaction conditions (70-90°C), utilizes cheap and readily available starting materials like trifluoroethylimidoyl chloride, and achieves high reaction efficiency with yields up to 99% for certain substrates, significantly simplifying the purification process compared to traditional harsh cyclization methods.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent explicitly states that the preparation method can be easily expanded to gram-level reactions and provides the possibility for subsequent scale production. The use of common organic solvents like THF and standard catalysts supports robust commercial scale-up of complex pharmaceutical intermediates.
Q: What types of substituents are tolerated in this triazole synthesis?
A: The method demonstrates a wide tolerance range for substrate functional groups. It successfully synthesizes derivatives where the R group is phenethyl or substituted/unsubstituted aryl, including substituents like methyl, methoxy, fluoro, chloro, nitro, and tert-butyl groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Mo/Cu-catalyzed synthesis for producing high-value fluorinated heterocycles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of organometallic catalysis, including strict moisture control and efficient solvent recovery systems, guaranteeing stringent purity specifications for every batch. With our rigorous QC labs and dedicated process development team, we are committed to delivering 3-trifluoromethyl-1,2,4-triazole derivatives that meet the highest international standards for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can accelerate your time-to-market while maximizing value. Let us be your strategic partner in navigating the complexities of fine chemical synthesis.
