Advanced Synthesis of Trifluoromethyl Quinazolinones: Commercial Scale-Up and Supply Chain Excellence
The patent CN112125856A introduces a groundbreaking synthetic methodology for producing 2-trifluoromethyl substituted quinazolinone derivatives, representing a significant advancement in the field of pharmaceutical intermediate manufacturing. This innovative approach addresses critical limitations in conventional synthesis routes by employing a solid carbon monoxide surrogate system that eliminates the need for handling toxic gaseous CO while maintaining high reaction efficiency and product purity. The methodology demonstrates exceptional versatility across diverse substrate combinations, enabling the production of structurally complex quinazolinone derivatives that serve as key building blocks for numerous therapeutic agents including anticonvulsants, antitumor compounds, and hypnotic medications. By leveraging readily available starting materials and simplified reaction protocols, this patented process offers pharmaceutical manufacturers a safer, more scalable pathway to access these valuable heterocyclic compounds without compromising on quality or yield parameters essential for drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for 2-trifluoromethyl quinazolinone derivatives suffer from multiple critical limitations that hinder their commercial viability and scalability in pharmaceutical manufacturing environments. Conventional methods typically require harsh reaction conditions involving high-pressure carbon monoxide gas handling systems, which present significant safety hazards and necessitate specialized equipment that increases capital expenditure and operational complexity. Many existing protocols rely on expensive or unstable reagents such as trifluoroacetic anhydride or unstable trifluoroacetamide intermediates that require pre-activation steps and generate substantial waste streams requiring complex disposal procedures. Furthermore, these methods often exhibit narrow substrate scope with limited functional group tolerance, resulting in low yields for structurally diverse derivatives and necessitating extensive optimization for each new compound variant. The requirement for multiple purification steps to remove transition metal catalysts and byproducts further complicates process economics and reduces overall manufacturing efficiency, making traditional approaches unsuitable for large-scale commercial production of pharmaceutical intermediates where consistency and cost-effectiveness are paramount.
The Novel Approach
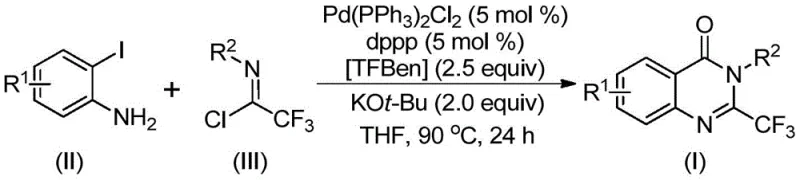
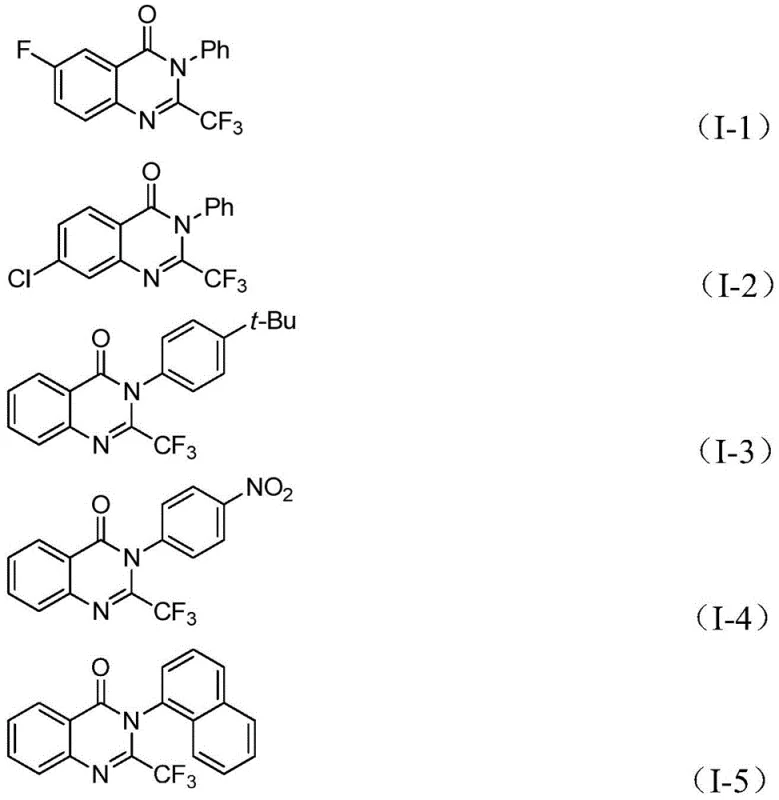
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation process utilizing TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide surrogate that safely releases CO in situ during the reaction sequence. This innovative approach eliminates the need for specialized high-pressure equipment while maintaining excellent reaction efficiency at a practical temperature of 90°C with reaction times ranging from 16 to 30 hours. The process demonstrates remarkable substrate versatility with various o-iodoaniline derivatives (R¹ = H, alkyl, halogen, CF₃) and trifluoroethylimide chloride analogs (R² = diverse aryl groups), enabling the synthesis of structurally complex quinazolinone derivatives without requiring significant protocol modifications. By employing readily available starting materials including commercially accessible palladium catalysts and ligands, the method achieves high yields while maintaining excellent purity profiles suitable for pharmaceutical applications. The simplified workup procedure involving standard filtration and column chromatography further enhances the process's commercial viability by reducing production complexity and associated costs compared to conventional approaches.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The reaction mechanism begins with potassium tert-butoxide-promoted intermolecular carbon-nitrogen bond coupling between o-iodoaniline and trifluoroethylimide chloride to form a trifluoroacetamidine intermediate. This key intermediate then undergoes oxidative addition with the palladium(0) catalyst generated in situ from Pd(PPh₃)₂Cl₂ and dppp ligand, forming a divalent palladium species that inserts into the carbon-iodine bond. Upon heating to 90°C, TFBen decomposes to release carbon monoxide which inserts into the carbon-palladium bond to form an acyl palladium intermediate. Subsequent base-promoted cyclization creates a seven-membered palladacycle intermediate that undergoes reductive elimination to yield the final 2-trifluoromethyl quinazolinone product while regenerating the active palladium catalyst for further catalytic cycles. This well-defined mechanistic pathway explains the high efficiency and selectivity observed across diverse substrate combinations while avoiding common side reactions that plague conventional methodologies.
The process inherently controls impurity formation through several key mechanistic features that ensure high product purity essential for pharmaceutical applications. The controlled release of carbon monoxide from TFBen prevents CO overpressure conditions that could lead to undesired side products, while the specific ligand system (dppp) stabilizes the palladium catalyst against decomposition pathways that typically generate metal-containing impurities. The reaction's tolerance for various functional groups without requiring protection/deprotection steps minimizes potential side reactions that could introduce impurities, and the mild basic conditions prevent acid-catalyzed degradation pathways common in alternative synthetic routes. The final column chromatography purification step effectively removes any residual catalyst or minor byproducts, consistently delivering products meeting stringent pharmaceutical purity requirements as evidenced by comprehensive analytical data including HRMS confirmation and multi-nuclear NMR characterization across all tested derivatives.
How to Synthesize Quinazolinone Derivatives Efficiently
This patented methodology provides a robust framework for manufacturing high-purity quinazolinone intermediates through a carefully optimized palladium-catalyzed carbonylation process that leverages solid CO surrogate technology to overcome traditional safety and scalability limitations. The synthesis pathway demonstrates exceptional versatility across diverse substrate combinations while maintaining consistent product quality and yield profiles essential for pharmaceutical manufacturing requirements. Detailed operational parameters including precise catalyst loading ratios, solvent selection criteria, and temperature control protocols have been validated through extensive experimental work to ensure reliable scale-up performance from laboratory to commercial production environments. The following standardized procedure outlines the critical implementation steps required to consistently achieve high-quality results while maintaining process efficiency and safety standards.
- Prepare reaction mixture with palladium catalyst, dppp ligand, TFBen as CO surrogate, and potassium tert-butoxide in THF solvent under inert atmosphere
- Add o-iodoaniline and trifluoroethylimide chloride substrates with precise molar ratios (1: 2:0.05) to initiate the carbonylation cascade
- Maintain reaction at 90°C for 24 hours with continuous monitoring, followed by standard workup and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial strategic advantages for procurement and supply chain decision-makers by addressing critical pain points in pharmaceutical intermediate sourcing while enhancing overall operational resilience and cost efficiency. The methodology's foundation in readily available starting materials and simplified process requirements creates significant opportunities for optimizing supply chain performance without compromising on product quality or regulatory compliance standards required in pharmaceutical manufacturing environments.
- Cost Reduction in Manufacturing: The elimination of hazardous carbon monoxide gas handling systems reduces capital expenditure requirements while avoiding costly safety infrastructure investments typically needed for high-pressure gas operations. The use of commercially available catalysts and ligands at optimized loadings minimizes raw material costs without sacrificing reaction efficiency, while the simplified workup procedure reduces solvent consumption and waste disposal expenses associated with complex purification protocols required by conventional methods.
- Enhanced Supply Chain Reliability: The reliance on widely accessible starting materials with established global supply chains significantly improves raw material security compared to specialized reagents required by alternative synthetic routes. The process's tolerance for minor variations in starting material quality ensures consistent production outcomes even when sourcing from multiple suppliers, reducing vulnerability to single-source dependencies while maintaining stringent quality specifications required for pharmaceutical intermediates.
- Scalability and Environmental Compliance: The atmospheric pressure operation eliminates complex engineering requirements for scale-up, enabling seamless transition from laboratory development to commercial production volumes without reoptimization. The reduced waste generation profile resulting from fewer processing steps and elimination of hazardous reagents supports environmental sustainability goals while simplifying regulatory compliance documentation required for modern pharmaceutical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patented methodology's implementation parameters and performance characteristics observed during extensive experimental validation across diverse substrate combinations.
Q: How does this method eliminate safety risks associated with traditional CO-based syntheses?
A: The patent replaces toxic gaseous carbon monoxide with solid TFBen (1,3,5-tricarboxylic acid phenol ester) as a carbon monoxide surrogate, eliminating high-pressure gas handling requirements while maintaining reaction efficiency through thermal decomposition at 90°C.
Q: What substrate flexibility does this methodology offer for pharmaceutical intermediate production?
A: The process demonstrates exceptional substrate scope with various R¹ groups (H, alkyl, halogen, CF₃) and R² aryl substituents (phenyl, naphthyl, nitrophenyl), enabling customized synthesis of diverse quinazolinone derivatives without modifying core reaction conditions.
Q: How does the elimination of transition metal removal steps impact commercial manufacturing costs?
A: By utilizing a palladium catalyst system that achieves high conversion without requiring extensive metal scavenging procedures, the method significantly reduces downstream purification complexity and associated costs while maintaining stringent purity specifications required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Derivatives Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation. As a specialized CDMO partner for complex heterocyclic compounds, we have successfully implemented this patented methodology across multiple client projects with consistent delivery of high-quality quinazolinone intermediates meeting exacting pharmaceutical standards required for global regulatory submissions.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis approach can optimize your specific supply chain requirements. Please contact us to obtain detailed COA data and route feasibility assessments tailored to your unique manufacturing needs.
