Advanced Palladium-Catalyzed Synthesis of 4-Acyl-2(5H)-furanone Intermediates for Pharmaceutical Manufacturing
Introduction to Next-Generation Furanone Synthesis
The landscape of organic synthesis for bioactive scaffolds is constantly evolving, driven by the need for more efficient and safer manufacturing processes. Patent CN112239436A introduces a groundbreaking preparation method for 4-acyl-2(5H)-furanone compounds, a structural backbone prevalent in numerous natural products and pharmacologically active molecules. These compounds are not merely academic curiosities; they serve as critical motifs in antimicrobial agents, analgesics, and antifouling agents used in marine ecosystems. The significance of this technology lies in its ability to construct these complex heterocyclic systems through a streamlined, one-pot palladium-catalyzed bis-carbonylation reaction. By leveraging aryl trifluoromethanesulfonates and propiolic alcohols as starting materials, this method bypasses the limitations of traditional multi-step syntheses. The versatility of this approach is underscored by the broad range of bioactivities associated with the furanone core, ranging from significant antibacterial properties to potent analgesic effects, as illustrated by known bioactive examples in the field.
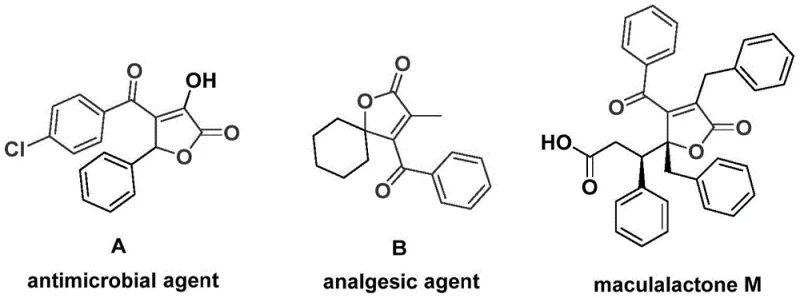
For research and development teams focused on novel drug discovery, access to diverse libraries of functionalized furanones is essential. The methodology described in this patent provides a robust platform for generating such libraries with high efficiency. Unlike older methods that might struggle with regioselectivity or require harsh conditions, this new protocol operates under controlled thermal parameters using accessible reagents. The integration of a palladium catalyst system with a solid carbon monoxide substitute represents a significant leap forward in process safety and operational simplicity. This technological advancement positions manufacturers to supply high-purity pharmaceutical intermediates with greater reliability, addressing the growing demand for complex heterocyclic building blocks in the global medicinal chemistry market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles like 4-acyl-2(5H)-furanones has been fraught with challenges that hinder large-scale adoption. Traditional carbonylation reactions often rely on the direct use of carbon monoxide gas, which poses severe safety hazards due to its toxicity and flammability, requiring specialized high-pressure equipment and rigorous safety protocols that inflate capital expenditure. Furthermore, existing literature reports on the synthesis of this specific furanone skeleton via carbonylation are scarce, indicating a gap in reliable, generalizable methodologies. Many conventional routes suffer from poor substrate compatibility, failing to tolerate diverse functional groups on the aromatic ring, which limits the chemical space available for drug optimization. Additionally, multi-step sequences often required to install the acyl group can lead to cumulative yield losses and increased waste generation, making the process economically unviable for commercial scale-up. The lack of efficient, one-step transformations has long been a bottleneck for procurement teams seeking cost-effective sources of these valuable intermediates.
The Novel Approach
The innovative method disclosed in patent CN112239436A effectively dismantles these barriers by introducing a palladium-catalyzed bis-carbonylation strategy that is both operationally simple and chemically robust. Instead of handling dangerous CO gas, the process utilizes chromium hexacarbonyl as a solid, manageable carbon monoxide substitute, which releases CO in situ under the reaction conditions. This substitution dramatically enhances workplace safety and simplifies reactor design requirements. The reaction proceeds by coupling propiolic alcohols with aryl trifluoromethanesulfonates in the presence of a palladium catalyst and a specific ligand system, typically involving palladium acetate and 1,1'-bis(diphenylphosphino)ferrocene (DPPP). This catalytic system facilitates the formation of two carbon-carbon bonds and the incorporation of two carbonyl units in a single operational step. The result is a direct, high-yielding pathway to the target 4-acyl-2(5H)-furanone structure, significantly reducing the number of unit operations and processing time compared to legacy methods.
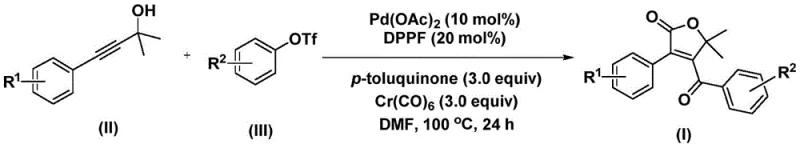
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific substrates. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-oxygen bond of the aryl trifluoromethanesulfonate, generating an aryl-palladium intermediate. This step is critical and is facilitated by the electron-rich nature of the DPPP ligand, which stabilizes the metal center. Subsequently, carbon monoxide, liberated from the decomposition of chromium hexacarbonyl, inserts into the aryl-palladium bond to form an acyl-palladium species. This acyl intermediate then undergoes a migratory insertion with the triple bond of the propiolic alcohol substrate. This unique sequence constructs the five-membered ring framework characteristic of the furanone core. The precision of this insertion step dictates the regioselectivity of the final product, ensuring that the acyl group is positioned correctly at the 4-position of the furanone ring.
Following the initial ring closure, the mechanism proceeds with a second carbon monoxide insertion event. This step transforms the five-membered ring palladium intermediate into a six-membered acyl-palladium species, setting the stage for the final bond formation. The cycle concludes with a reductive elimination step, which releases the desired 4-acyl-2(5H)-furanone product and regenerates the active palladium(0) catalyst to continue the cycle. This dual-carbonylation mechanism is highly efficient, minimizing the formation of side products that typically plague complex cascade reactions. The use of p-toluquinone as an additive further aids in maintaining the catalytic activity and preventing catalyst deactivation. The robustness of this mechanism allows for a wide range of substituents on both the alkyne and the aryl triflate, as evidenced by the successful synthesis of various derivatives bearing electron-donating and electron-withdrawing groups, ensuring high purity and consistent quality in the final intermediate.
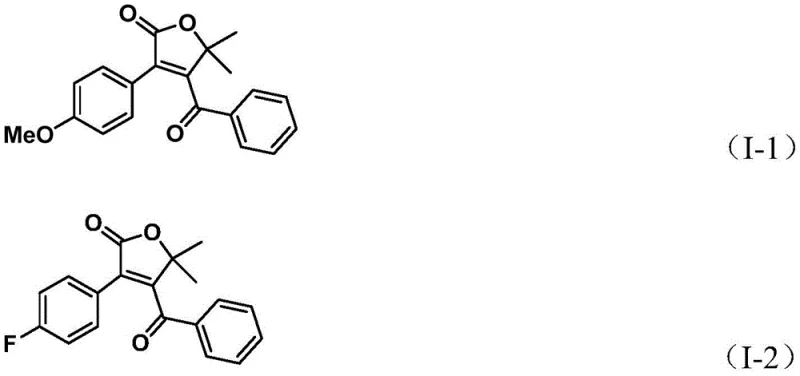
How to Synthesize 4-Acyl-2(5H)-furanone Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to maximize yield and reproducibility. The process begins with the precise weighing of reagents, including the palladium catalyst, ligand, chromium carbonyl source, and the organic substrates, which are then dissolved in a polar aprotic solvent such as N,N-dimethylformamide (DMF). The reaction mixture is heated to a temperature range of 100-120°C and maintained under stirring for a period of 24 to 48 hours to ensure complete conversion. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below to assist technical teams in replicating this high-efficiency protocol.
- Combine palladium acetate, DPPP ligand, chromium carbonyl, p-toluquinone, propiolic alcohol, and aryl triflate in DMF solvent.
- Heat the reaction mixture to 100-120°C and stir for 24-48 hours under inert atmosphere to facilitate bis-carbonylation.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 4-acyl-2(5H)-furanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis method offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant reduction of operational complexity and associated costs. By replacing hazardous gaseous carbon monoxide with a solid surrogate, the process eliminates the need for expensive high-pressure reactors and specialized gas handling infrastructure, leading to substantial capital expenditure savings. Furthermore, the use of readily available starting materials like aryl triflates and propiolic alcohols ensures a stable and resilient supply chain, mitigating the risk of raw material shortages that can disrupt production schedules. The simplicity of the post-treatment process, which involves basic filtration and standard chromatography, reduces the demand for specialized purification equipment and lowers utility consumption.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the high atom economy and the elimination of costly safety measures associated with toxic gases. The use of common solvents like DMF and commercially sourced catalysts keeps reagent costs low, while the high yields reported minimize waste disposal fees. This streamlined approach translates directly into a lower cost of goods sold (COGS) for the final pharmaceutical intermediate, providing a competitive pricing advantage in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions allows for consistent batch-to-batch reproducibility, a critical factor for maintaining long-term supply contracts with major pharmaceutical clients. The tolerance of the method to various functional groups means that a single manufacturing platform can produce a diverse array of furanone derivatives, increasing supply chain flexibility. This adaptability ensures that manufacturers can quickly respond to changing market demands for specific analogues, reducing lead times and enhancing overall service levels for downstream customers seeking reliable pharma intermediate suppliers.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with green chemistry principles by avoiding the use of pressurized toxic gases and utilizing efficient catalytic cycles. The simplified workup reduces the volume of organic waste generated, easing the burden on waste treatment facilities and ensuring compliance with increasingly stringent environmental regulations. The scalability of the reaction from gram to kilogram scales has been demonstrated, confirming its suitability for commercial production. This scalability ensures that supply chain heads can plan for long-term volume requirements with confidence, knowing that the technology can support growth from pilot batches to multi-ton annual production capacities.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled answers to common questions regarding the synthesis and application of these furanone compounds. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions regarding the integration of this intermediate into their own development pipelines.
Q: What are the key advantages of this carbonylation method over traditional routes?
A: This method utilizes solid chromium carbonyl as a safe CO surrogate, eliminating the need for high-pressure carbon monoxide gas equipment. It offers excellent substrate compatibility and operates under relatively mild thermal conditions (100-120°C).
Q: What is the typical purity profile of the synthesized furanone intermediates?
A: The process demonstrates high reaction efficiency and selectivity, yielding products that can be purified to high specifications using standard silica gel column chromatography, suitable for downstream pharmaceutical applications.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the protocol uses commercially available catalysts and reagents with simple post-treatment steps (filtration and chromatography), making it highly amenable to scale-up from laboratory to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2(5H)-furanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team of expert chemists has thoroughly analyzed the potential of this palladium-catalyzed bis-carbonylation route and is prepared to leverage it for your specific needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from the laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 4-acyl-2(5H)-furanone delivered meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term strategic goals in the competitive pharmaceutical marketplace.
