Scalable Palladium-Catalyzed Bis-Carbonylation for High-Purity 4-Acyl-2(5H)-Furanone Intermediates
Introduction to Advanced Furanone Scaffold Synthesis
The structural motif of 4-acyl-2(5H)-furanones represents a privileged scaffold in medicinal chemistry, frequently appearing in natural products and bioactive molecules with significant therapeutic potential. As highlighted in recent literature, derivatives of this core structure exhibit potent antibacterial, analgesic, and antifouling activities, making them highly sought-after targets for drug discovery programs. However, traditional synthetic routes to access these complex heterocycles often suffer from multi-step sequences, harsh reaction conditions, or poor atom economy, creating bottlenecks in the supply chain for research and development teams. Addressing these critical challenges, the invention disclosed in patent CN112239436A introduces a robust and efficient preparation method that leverages palladium-catalyzed bis-carbonylation to construct the furanone core in a single operational step.
This novel methodology utilizes readily available propiolic alcohols and aryl trifluoromethanesulfonates as starting materials, reacting them in the presence of a palladium catalyst and a solid carbon monoxide source. The significance of this approach lies not only in its chemical elegance but also in its practical applicability for industrial manufacturing. By streamlining the synthesis into a concise one-pot procedure, the technology offers a reliable pathway for producing high-purity pharmaceutical intermediates with enhanced substrate tolerance. For global procurement and supply chain leaders, this represents a strategic opportunity to secure a stable supply of key building blocks while mitigating the risks associated with complex, low-yield synthetic routes.
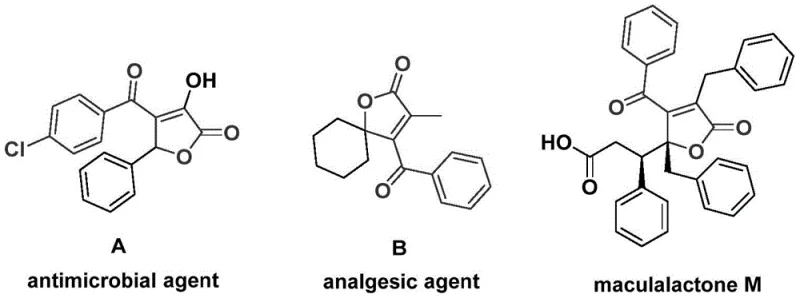
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 4-acyl-2(5H)-furanone skeleton has relied on classical condensation reactions or multi-step functional group manipulations that are inherently inefficient for large-scale production. Conventional strategies often require the pre-functionalization of substrates, leading to increased waste generation and higher raw material costs due to the necessity of protecting group chemistry. Furthermore, many existing protocols utilize gaseous carbon monoxide under high pressure, which imposes severe safety constraints and requires specialized autoclave equipment that is not universally available in standard pilot plants. These operational hazards, combined with the difficulty in controlling regioselectivity during ring closure, frequently result in complex impurity profiles that demand extensive and costly purification efforts.
The Novel Approach
In stark contrast to these legacy methods, the patented process described in CN112239436A employs a sophisticated palladium-catalyzed cascade reaction that achieves bis-carbonylation and cyclization simultaneously under mild conditions. The core innovation involves the use of chromium hexacarbonyl as a safe, solid surrogate for carbon monoxide, which decomposes in situ to provide the necessary CO equivalents without the logistical nightmare of handling toxic gases. This reaction proceeds efficiently in polar aprotic solvents like DMF at moderate temperatures, typically around 100 °C, ensuring high conversion rates while minimizing thermal degradation of sensitive functional groups. The result is a direct, one-step transformation that significantly reduces the number of unit operations, thereby lowering the overall cost of goods sold (COGS) and accelerating the timeline from benchtop discovery to commercial manufacturing.
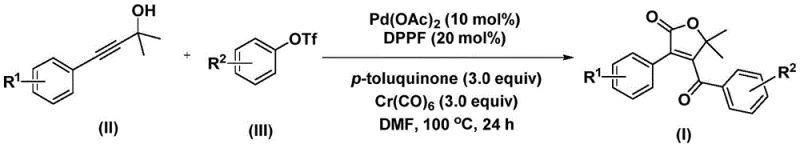
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
The mechanistic pathway of this transformation is a testament to the precision of modern organometallic catalysis, initiating with the oxidative addition of the palladium(0) species into the carbon-oxygen bond of the aryl trifluoromethanesulfonate. This critical step generates an aryl-palladium intermediate, which subsequently undergoes migratory insertion of carbon monoxide liberated from the chromium hexacarbonyl additive to form an acyl-palladium complex. The presence of the bidentate ligand DPPF (1,1'-bis(diphenylphosphino)ferrocene) is crucial here, as its wide bite angle stabilizes the palladium center and facilitates the subsequent coordination and insertion of the propiolic alcohol triple bond. This sequence creates a five-membered ring palladium intermediate, setting the stage for the second carbonylation event.
Following the initial cyclization, a second molecule of carbon monoxide inserts into the palladium-carbon bond, expanding the coordination sphere to form a six-membered ring palladium intermediate prior to the final ring contraction. The cycle concludes with a reductive elimination step that releases the desired 4-acyl-2(5H)-furanone product and regenerates the active palladium(0) catalyst. From an impurity control perspective, this mechanism is highly advantageous because the intramolecular nature of the cyclization steps inherently favors the formation of the five-membered lactone ring over intermolecular oligomerization. The use of p-toluquinone as an additive further assists in maintaining the catalytic cycle efficiency, likely by re-oxidizing any inactive palladium black back into the active catalytic species, thus ensuring consistent reaction performance and high product purity throughout the batch.
How to Synthesize 4-Acyl-2(5H)-Furanone Efficiently
The execution of this synthesis protocol is designed for operational simplicity, requiring standard laboratory glassware or stainless steel reactors capable of maintaining temperatures up to 120 °C. The process begins by charging the reactor with the palladium catalyst, ligand, chromium carbonyl, and oxidant, followed by the addition of the propiolic alcohol and aryl triflate substrates in DMF solvent. The mixture is then heated to reflux conditions, typically maintained at 100 °C for a duration of 24 hours, allowing sufficient time for the slow release of CO and the completion of the catalytic cycle. Upon completion, the workup is remarkably straightforward, involving simple filtration to remove insoluble metal residues and chromium byproducts, followed by adsorption onto silica gel and purification via standard column chromatography to yield the target compound.
- Combine palladium acetate, DPPF ligand, chromium hexacarbonyl, p-toluquinone, propiolic alcohol, and aryl triflate in DMF solvent.
- Heat the reaction mixture to 100 °C and stir continuously for 24 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Filter the reaction mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 4-acyl-2(5H)-furanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials; both propiolic alcohols and aryl triflates are commodity chemicals that can be sourced from multiple global vendors, reducing dependency on single-source suppliers and mitigating geopolitical supply risks. Furthermore, the replacement of gaseous carbon monoxide with a solid chromium carbonyl source eliminates the need for expensive high-pressure reactor infrastructure and the associated regulatory compliance costs for handling toxic gases. This shift not only lowers capital expenditure (CAPEX) for new production lines but also significantly reduces the operational overhead related to safety monitoring and emergency response planning.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the convergence of high atom economy and reduced processing time. By consolidating what would traditionally be a multi-step synthesis into a single pot, manufacturers eliminate the solvent usage, energy consumption, and labor costs associated with intermediate isolation and purification steps. Although palladium is a precious metal, the catalytic nature of the reaction means that the metal loading is minimal relative to the mass of the product produced, and the ability to recover and recycle the metal from the filtration cake further enhances the cost-efficiency profile. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove side products, reducing the burden on downstream purification units and increasing the overall throughput of the facility.
- Enhanced Supply Chain Reliability: The robustness of this chemical platform ensures a consistent and reliable supply of critical intermediates, which is paramount for maintaining uninterrupted API production schedules. The tolerance of the reaction to a wide variety of functional groups—including halogens, alkoxy groups, and trifluoromethyl moieties—means that a single standardized protocol can be adapted to produce a diverse library of furanone derivatives without requiring extensive process re-optimization. This flexibility allows supply chain planners to respond rapidly to changing market demands or clinical trial requirements, as the lead time for scaling up new analogs is significantly compressed compared to traditional bespoke synthetic routes.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns well with modern green chemistry principles by utilizing a closed system that prevents the release of volatile organic compounds and toxic gases. The use of DMF as a solvent, while requiring careful management, is a well-understood industrial solvent with established recovery and recycling protocols, facilitating waste minimization. The scalability of the reaction is supported by the homogeneous nature of the catalytic system, which ensures efficient heat and mass transfer even in large-volume reactors, thereby reducing the risk of thermal runaways and ensuring batch-to-batch consistency essential for regulatory compliance in pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, aimed at clarifying the operational parameters for potential licensees or manufacturing partners. Understanding these nuances is critical for R&D teams evaluating the feasibility of integrating this technology into their existing production portfolios.
Q: What is the role of Chromium Hexacarbonyl in this synthesis?
A: Chromium hexacarbonyl [Cr(CO)6] serves as a solid carbon monoxide substitute, eliminating the need for hazardous high-pressure CO gas cylinders while providing a controlled release of CO for the carbonylation steps.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating electron-donating groups like methoxy and alkyl chains, as well as electron-withdrawing groups such as halogens and trifluoromethyl moieties.
Q: What represents the primary cost driver in this catalytic system?
A: While palladium is a precious metal, the use of commercially available ligands like DPPF and the avoidance of specialized high-pressure reactor infrastructure significantly offset capital expenditures, making the overall process economically viable for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2(5H)-Furanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced heterocyclic intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity 4-acyl-2(5H)-furanone derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in palladium catalysis and carbonylation chemistry positions us as a strategic partner capable of navigating the complexities of this specific synthesis to deliver value to your organization.
We invite you to engage with our technical procurement team to discuss how this innovative synthetic route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages this method offers over your current supply chain. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to initiate a dialogue regarding route feasibility assessments for your proprietary targets, ensuring your pipeline remains robust and competitive in the global market.
