Advanced Palladium-Catalyzed Synthesis of 4-Acyl-2(5H)-Furanones for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 4-Acyl-2(5H)-Furanones for Commercial Scale-Up
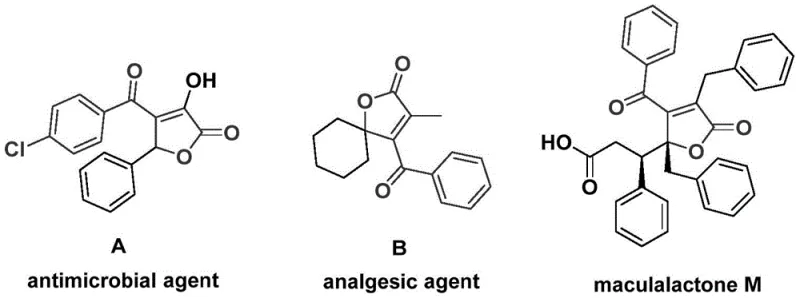
The landscape of organic synthesis for bioactive heterocycles is constantly evolving, driven by the need for more efficient and scalable methodologies. A significant breakthrough in this domain is detailed in patent CN112239436A, which discloses a robust preparation method for 4-acyl-2(5H)-furanone compounds. These structural motifs are not merely academic curiosities; they serve as critical backbones in numerous natural products and pharmaceutical agents. As illustrated in the provided data, compounds featuring this core exhibit potent biological activities, ranging from antimicrobial properties seen in Agent A to analgesic effects in Agent B, and even antifouling capabilities in Maculalactone M. For R&D directors and procurement specialists alike, mastering the synthesis of these scaffolds represents a strategic advantage in developing next-generation therapeutics and agrochemicals.
The traditional approaches to constructing the 4-acyl-2(5H)-furanone ring system often suffer from significant limitations that hinder industrial application. Conventional methods frequently rely on multi-step sequences involving harsh reagents, unstable intermediates, or the direct use of toxic carbon monoxide gas under high pressure. These factors introduce substantial safety risks and complicate the supply chain, making cost reduction in fine chemical manufacturing difficult to achieve. Furthermore, older methodologies often struggle with substrate tolerance, failing to accommodate diverse functional groups without extensive protection-deprotection strategies. This lack of versatility limits the chemical space available for drug discovery and increases the lead time for high-purity pharmaceutical intermediates.
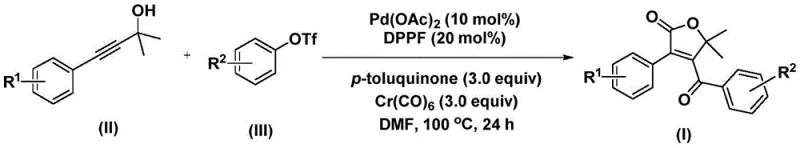
In stark contrast, the novel approach outlined in the patent utilizes a palladium-catalyzed bis-carbonylation strategy that elegantly bypasses these hurdles. By employing propiolic alcohols and aryl trifluoromethanesulfonates as readily available starting materials, the method constructs the target scaffold in a single operational step. The use of chromium hexacarbonyl as a solid carbon monoxide surrogate is particularly ingenious, as it allows for the controlled release of CO in situ, thereby avoiding the logistical nightmares associated with gaseous CO handling. This transformation proceeds under relatively mild conditions (100 °C in DMF) and demonstrates exceptional functional group compatibility, enabling the rapid generation of diverse libraries of furanone derivatives for biological screening.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
Understanding the catalytic cycle is paramount for R&D teams aiming to optimize this process for commercial scale-up. The reaction initiates with the oxidative addition of the aryl trifluoromethanesulfonate to the active palladium(0) species, generated in situ from palladium acetate and the DPPP ligand. This forms a crucial aryl-palladium intermediate. Subsequently, carbon monoxide, liberated from the chromium hexacarbonyl additive, inserts into the palladium-carbon bond to generate an acyl-palladium species. This first carbonylation event sets the stage for the subsequent cyclization.
The mechanism then proceeds through a fascinating cascade where the acyl-palladium intermediate interacts with the propiolic alcohol substrate. This interaction facilitates the formation of a five-membered ring palladium intermediate, followed by a second carbon monoxide insertion to yield a six-membered ring species. The cycle concludes with a reductive elimination step that releases the final 4-acyl-2(5H)-furanone product and regenerates the palladium catalyst. This intricate dance of insertion and elimination ensures high atom economy. Moreover, the presence of p-toluquinone as an additive likely plays a role in re-oxidizing any reduced palladium species or stabilizing the catalytic cycle, ensuring consistent turnover numbers and minimizing the formation of palladium black, which is a common cause of catalyst deactivation in such transformations.
How to Synthesize 4-Acyl-2(5H)-Furanone Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The protocol dictates the use of anhydrous DMF as the solvent, which is critical for solubilizing the polar transition states and maintaining catalyst stability. The stoichiometry is equally important, with a molar ratio of palladium acetate to ligand to chromium carbonyl to quinone optimized at 0.1:0.2:3:3. Deviating from these ratios can lead to incomplete conversion or the accumulation of side products. The reaction is typically run at 100 °C for 24 hours, a timeframe that balances reaction completeness with throughput efficiency. Post-reaction workup is straightforward, involving filtration to remove metal residues and silica gel treatment followed by column chromatography.
- Combine palladium acetate, DPPP ligand, p-toluquinone, chromium hexacarbonyl, propiolic alcohol, and aryl triflate in DMF solvent.
- Heat the reaction mixture to 100 °C and stir for 24 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 4-acyl-2(5H)-furanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this technology are profound. The shift from multi-step syntheses to a one-pot procedure drastically simplifies the manufacturing workflow. By eliminating intermediate isolation steps, the process reduces solvent consumption, labor costs, and waste generation, leading to substantial cost savings in pharmaceutical intermediate manufacturing. The reliance on commercially available starting materials, such as substituted phenols (precursors to triflates) and terminal alkynols, ensures a stable and reliable supply chain. These raw materials are produced on a massive industrial scale, mitigating the risk of shortages that often plague specialty chemical sectors.
- Cost Reduction in Manufacturing: The elimination of high-pressure gas equipment and the use of solid CO surrogates significantly lowers capital expenditure (CAPEX) for reactor setup. Furthermore, the high yields reported (often exceeding 80-90% for optimized substrates) mean less raw material is wasted per kilogram of product. The simplified purification process, which avoids complex crystallizations or distillations of unstable intermediates, further drives down the operational expenditure (OPEX), making the final API intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against various functional groups means that a single production line can be adapted to produce a wide array of furanone derivatives simply by swapping the aryl triflate input. This flexibility allows manufacturers to respond rapidly to changing market demands without retooling entire facilities. Additionally, the use of standard organic solvents like DMF, which are widely stocked by chemical distributors, ensures that production schedules are not disrupted by solvent logistics.
- Scalability and Environmental Compliance: From an environmental standpoint, the use of chromium hexacarbonyl, while requiring careful handling, avoids the venting of CO gas, aligning with stricter emissions regulations. The high atom efficiency of the bis-carbonylation reaction minimizes the E-factor (mass of waste per mass of product). As the industry moves towards greener chemistry, processes that offer high selectivity and reduced waste streams are increasingly favored by regulatory bodies and corporate sustainability goals, facilitating smoother approval processes for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scalability and specificity of this palladium-catalyzed methodology. Understanding these nuances is essential for technology transfer teams evaluating the feasibility of adopting this route for large-scale production. The answers are derived directly from the experimental data and mechanistic understanding provided in the patent literature.
Q: What are the key advantages of using Chromium Hexacarbonyl in this synthesis?
A: Chromium hexacarbonyl serves as a safe, solid carbon monoxide substitute, eliminating the need for handling hazardous high-pressure CO gas while ensuring efficient CO insertion into the palladium intermediate.
Q: What is the substrate scope for the aryl triflate component?
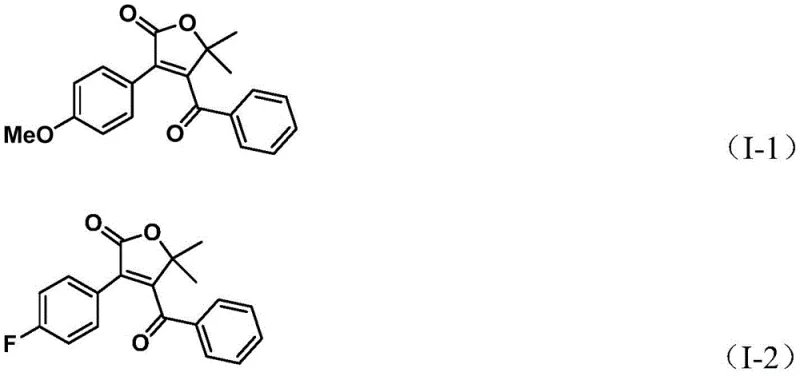
A: The method demonstrates excellent compatibility with various substituents including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens (F, Cl, Br) and trifluoromethyl.
Q: How does this method improve upon traditional furanone synthesis?
A: Unlike traditional multi-step routes, this protocol achieves the complex 4-acyl-2(5H)-furanone skeleton in a single operational step with high atom economy and simplified post-processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2(5H)-Furanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN112239436A. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 4-acyl-2(5H)-furanone meets the highest international standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your pipeline projects. Whether you require a Customized Cost-Saving Analysis for your existing supply chain or need specific COA data and route feasibility assessments for new analogs, our technical procurement team is ready to assist. Contact us today to discuss how we can support your R&D goals with reliable, high-quality chemical intermediates tailored to your specific needs.
