Advanced Palladium-Catalyzed Synthesis of High-Purity Substituted 2,3-Dihydroquinolone Compounds for Commercial Scale-Up
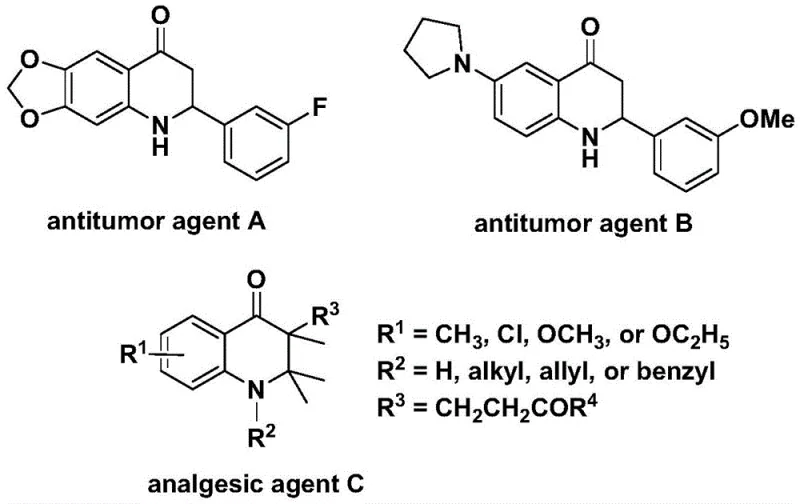
Patent CN112239456B introduces a groundbreaking methodology for synthesizing substituted 2,3-dihydroquinolone compounds through a palladium-catalyzed carbonylation reaction that addresses critical limitations in existing synthetic routes. This innovation leverages readily available starting materials including N-pyridine sulfonyl-o-iodoaniline and olefins under mild reaction conditions of 100–120°C for 24–48 hours in dioxane solvent with palladium bis(acetylacetonate) as catalyst. The process achieves exceptional substrate compatibility across diverse functional groups while maintaining operational simplicity through a single-step transformation that eliminates multi-stage purification requirements. Crucially, this approach enables precise construction of molecular architectures essential for pharmaceutical applications including antitumor agents A and B which demonstrate significant activity against human cancer cells as well as analgesic agent C with potent pain-relieving properties. The elimination of harsh reagents and transition metal residues significantly enhances product purity while reducing downstream processing complexity without compromising yield or structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for constructing dihydroquinolone scaffolds often require multi-step sequences involving harsh reaction conditions that generate significant impurities requiring extensive purification. These methods typically suffer from poor functional group tolerance when incorporating complex substituents necessary for pharmaceutical activity profiles, leading to reduced yields and inconsistent product quality across different substrate classes. The reliance on stoichiometric reagents rather than catalytic systems creates substantial waste streams that complicate environmental compliance while increasing production costs through additional separation steps. Furthermore, conventional routes frequently employ unstable intermediates that necessitate cryogenic conditions or specialized equipment not readily available in standard manufacturing facilities. The limited scalability of these processes becomes particularly problematic when transitioning from laboratory-scale development to commercial production volumes required by pharmaceutical manufacturers. Most critically, existing methodologies lack the structural flexibility needed to systematically explore structure-activity relationships essential for drug discovery programs targeting specific biological pathways.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation strategy that directly constructs the dihydroquinolone core in a single transformation step using commercially available starting materials. By employing N-pyridine sulfonyl protection on the aniline precursor, the process achieves remarkable functional group tolerance across both aryl and alkyl substituents while maintaining high regioselectivity during cyclization. The carefully optimized catalyst system featuring palladium bis(acetylacetonate) with dppp ligand operates under mild thermal conditions that prevent decomposition of sensitive functional groups commonly found in pharmaceutical intermediates. This approach eliminates multiple protection/deprotection steps required by conventional routes while generating minimal byproducts through its atom-economical design. The reaction demonstrates exceptional scalability from gram-scale laboratory demonstrations to potential industrial production volumes without requiring specialized equipment or hazardous reagents. Most significantly, the methodology provides unprecedented structural diversity through simple variation of olefin substrates while maintaining consistent high yields across diverse molecular architectures.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle begins with oxidative addition of palladium(0) into the carbon-nitrogen bond of N-pyridine sulfonyl-o-iodoaniline to form an aryl palladium intermediate that subsequently undergoes carbon monoxide insertion from the triethylamine-bound source to generate an acyl palladium species. This key intermediate then coordinates with the olefin substrate through π-complex formation followed by migratory insertion that establishes the critical carbon-carbon bond necessary for ring closure. The resulting alkyl palladium complex undergoes reductive elimination to release the substituted dihydroquinolone product while regenerating the active palladium(0) catalyst for subsequent cycles. This mechanism operates under precisely controlled thermal conditions that prevent β-hydride elimination side reactions while maintaining optimal catalyst turnover frequency throughout the reaction period. The pyridine sulfonyl group plays a dual role by facilitating oxidative addition through its electron-withdrawing properties while also directing regioselectivity during cyclization through chelation control.
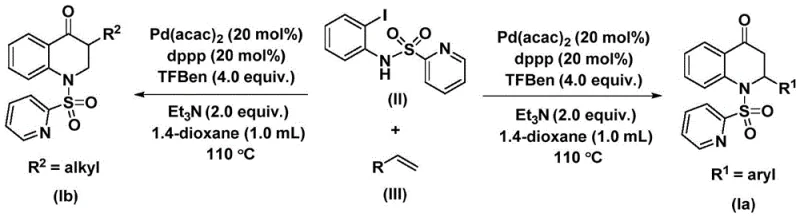
Impurity control is achieved through multiple built-in mechanisms within this catalytic system that prevent common side reactions observed in alternative methodologies. The selective insertion of carbon monoxide from the triethylamine-bound source eliminates competing pathways that would otherwise generate dicarbonyl byproducts or oligomerization products. The steric bulk of the pyridine sulfonyl group directs exclusive formation of the desired six-membered ring structure while suppressing five-membered ring formation or linear oligomerization pathways. Careful optimization of the triethylamine concentration prevents base-mediated decomposition of sensitive functional groups while maintaining sufficient basicity to facilitate reductive elimination steps. The use of dioxane as solvent provides ideal polarity characteristics that stabilize key intermediates without promoting unwanted solvolysis reactions. This comprehensive impurity management strategy results in consistently high product purity that meets stringent pharmaceutical requirements without requiring additional purification steps beyond standard chromatography.
How to Synthesize Substituted Dihydroquinolone Efficiently
This innovative synthesis route represents a significant advancement in manufacturing complex heterocyclic compounds through its streamlined approach that combines operational simplicity with exceptional molecular precision. The methodology leverages commercially available starting materials including N-pyridine sulfonyl-o-iodoaniline which can be rapidly prepared from o-iodoaniline and pyridine sulfonyl chloride using standard laboratory procedures. The reaction conditions are carefully optimized to balance catalyst activity with substrate stability through precise temperature control between 100–120°C and reaction times ranging from 24–48 hours depending on specific substrate characteristics. Detailed standardized synthesis steps are provided below to ensure consistent implementation across different manufacturing environments while maintaining high product quality standards required by pharmaceutical clients.
- Combine N-pyridine sulfonyl-o-iodoaniline, olefin substrates, palladium bis(acetylacetonate) catalyst, dppp ligand, and triethylamine in anhydrous dioxane under nitrogen atmosphere to form the reactive mixture.
- Heat the reaction mixture to precisely controlled temperatures between 100–120°C and maintain stirring for 24–48 hours to ensure complete carbonylation and cyclization.
- Execute post-reaction processing through filtration to remove catalyst residues followed by silica gel-assisted column chromatography purification to isolate high-purity dihydroquinolone products.
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points associated with traditional synthesis routes for complex heterocyclic intermediates. The elimination of multiple protection/deprotection steps significantly reduces raw material consumption while minimizing waste generation that would otherwise require costly disposal procedures under environmental regulations. The use of commercially available catalysts and solvents creates supply chain resilience through multiple sourcing options that prevent single-point failure scenarios common in specialized chemical manufacturing processes.
- Cost Reduction in Manufacturing: The streamlined single-step transformation eliminates expensive transition metal catalysts required by alternative methods while reducing solvent consumption through optimized reaction concentrations. This approach drastically simplifies waste management protocols by minimizing heavy metal contamination risks that would necessitate specialized treatment procedures. The elimination of cryogenic processing requirements further reduces energy consumption during manufacturing operations while maintaining consistent product quality across different production scales.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials including standard olefins and commercially sourced palladium catalysts creates robust supply chain options that mitigate raw material shortages common in specialized chemical manufacturing. The process demonstrates exceptional batch-to-batch consistency through its well-defined reaction parameters that enable reliable forecasting of production timelines without unexpected delays from purification challenges or yield variations.
- Scalability and Environmental Compliance: The methodology maintains consistent performance from laboratory scale through commercial production volumes due to its inherently scalable reaction design that avoids specialized equipment requirements. The simplified workup procedure significantly reduces aqueous waste streams compared to conventional routes while eliminating hazardous reagents that would complicate environmental compliance procedures during manufacturing scale-up.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN112239456B's experimental data and implementation requirements. These insights reflect practical experience from scaling similar catalytic processes within pharmaceutical manufacturing environments while addressing specific concerns raised by procurement and R&D teams during technology evaluation.
Q: How does this method overcome limitations in traditional dihydroquinolone synthesis?
A: This palladium-catalyzed carbonylation process eliminates multi-step sequences required by conventional methods by directly constructing the dihydroquinolone core through a single catalytic cycle. The use of stable N-pyridine sulfonyl protecting groups prevents undesired side reactions while enabling broad functional group tolerance across diverse substrates.
Q: What substrate compatibility advantages does this process offer for pharmaceutical applications?
A: The methodology accommodates a wide range of aryl and alkyl substituents including halogenated, alkoxy, and silyl groups without requiring specialized reaction conditions. This flexibility allows precise molecular tailoring for specific pharmacological targets while maintaining consistent yields across structurally complex intermediates.
Q: Can this synthesis be scaled while maintaining stringent pharmaceutical purity standards?
A: The process demonstrates inherent scalability from gram to multi-kilogram quantities through its simplified workup procedure that avoids transition metal contamination. The absence of chromatography-intensive purification steps ensures consistent high-purity output meeting regulatory requirements for active pharmaceutical ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Dihydroquinolone Supplier
Our patented methodology represents a significant advancement in heterocyclic compound manufacturing that combines cutting-edge catalytic chemistry with practical industrial implementation strategies developed through extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. NINGBO INNO PHARMCHEM brings deep expertise in complex molecule synthesis with stringent purity specifications maintained through our state-of-the-art QC labs equipped with advanced analytical instrumentation for comprehensive quality assurance testing at every production stage. This technical capability ensures consistent delivery of high-purity pharmaceutical intermediates meeting global regulatory standards while providing flexible manufacturing solutions tailored to specific client requirements.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis route can optimize your supply chain operations while maintaining superior product quality standards. Please contact us to obtain specific COA data and route feasibility assessments demonstrating how our manufacturing capabilities can support your next-generation pharmaceutical development programs.
