Scalable Production of 2-Trifluoromethyl Benzimidazoles Using Recyclable Copper Catalysts
Scalable Production of 2-Trifluoromethyl Benzimidazoles Using Recyclable Copper Catalysts
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are critical for enhancing metabolic stability and bioavailability. Patent CN113429349A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted benzimidazole compounds utilizing a heterogeneous copper-doped carbon nitride catalyst. This innovation represents a significant leap forward from traditional homogeneous catalysis, addressing long-standing challenges regarding catalyst recovery and metal contamination in final active pharmaceutical ingredients. By leveraging a Cu/g-C3N4 system, the process achieves exceptional reaction efficiency under mild conditions, typically yielding products with purity profiles suitable for direct downstream processing. For R&D directors and procurement specialists, this technology offers a compelling value proposition by combining high atom economy with the operational simplicity of a recyclable solid catalyst, thereby streamlining the supply chain for these valuable intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted benzimidazoles has relied on several conventional pathways that present significant bottlenecks for large-scale manufacturing. Traditional methods often involve the condensation of o-amino aniline with trifluoroacetic acid or aldehyde, which frequently requires harsh reaction conditions and suffers from narrow substrate scope limitations. Alternatively, reductive cyclization of o-nitroaniline or direct trifluoromethylation of existing benzimidazole cores often necessitates the use of expensive reagents and generates substantial waste streams. Furthermore, earlier reports utilizing homogeneous copper-catalyzed cascade cyclizations, while effective, introduce severe complications in post-reaction processing. The removal of residual copper species from the final product is both costly and technically demanding, often requiring specialized scavengers or extensive chromatography, which drastically reduces overall process throughput and increases the cost of goods sold for the final API intermediate.
The Novel Approach
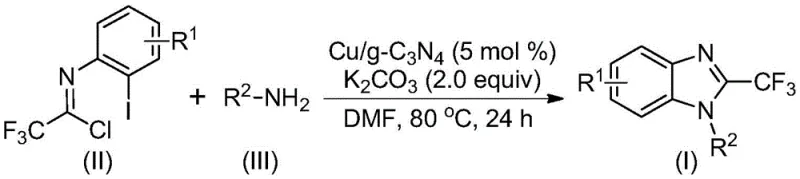
The novel approach detailed in the patent data fundamentally shifts the paradigm by employing a heterogeneous Cu/g-C3N4 catalyst that resolves the critical issue of catalyst separation. This method utilizes readily available trifluoroethylimidoyl chloride and various amines as starting materials, reacting them in the presence of potassium carbonate in a polar aprotic solvent like DMF. The key differentiator is the stability and recyclability of the catalyst; experimental data indicates that the catalytic efficiency remains high even after three recycling runs, with yields only partially decreasing. This translates to a drastic reduction in catalyst consumption costs and waste generation. Moreover, the reaction proceeds under relatively mild thermal conditions, typically around 80°C, which enhances energy efficiency and safety profiles compared to high-temperature alternatives. The broad tolerance for functional groups, including halogens and alkyl chains, ensures that this single platform technology can service a wide array of derivative syntheses without requiring extensive re-optimization.
Mechanistic Insights into Cu/g-C3N4 Catalyzed Cyclization
The mechanistic pathway for this transformation involves a sophisticated interplay between the heterogeneous catalyst surface and the organic substrates, facilitating a cascade cyclization that constructs the benzimidazole core with high precision. Initially, the trifluoroethylimidoyl chloride and the amine undergo a nucleophilic addition-elimination reaction to form an amidine intermediate. Subsequently, the copper species doped within the carbon nitride lattice inserts into the carbon-iodine bond of the aryl ring, forming a transient organocopper complex. This step is critical as it activates the aryl ring for the subsequent intramolecular attack. Under the basic conditions provided by potassium carbonate, a molecule of hydrogen iodide is eliminated, promoting the formation of a bivalent or trivalent cyclic copper intermediate. Finally, a reductive elimination step releases the desired 2-trifluoromethyl-substituted benzimidazole product and regenerates the active copper catalytic site on the support matrix.
This mechanism not only explains the high regioselectivity observed but also highlights the role of the graphitic carbon nitride support in stabilizing the copper species and preventing leaching. The heterogeneity of the system ensures that the active sites are accessible yet distinct from the bulk solution, allowing for the clean separation of the catalyst via simple filtration. For process chemists, understanding this mechanism is vital for troubleshooting and optimization, as it suggests that maintaining the integrity of the catalyst surface is key to long-term performance. The ability to tune the electronic properties of the carbon nitride support further offers a avenue for future catalyst optimization, potentially expanding the scope to even more challenging substrates. This deep mechanistic understanding provides a solid foundation for scaling the process from gram-scale laboratory experiments to multi-kilogram pilot plant operations with confidence.
How to Synthesize 2-Trifluoromethyl Benzimidazole Efficiently
Implementing this synthesis route in a production environment requires careful attention to the preparation of the catalyst and the control of reaction parameters to maximize yield and purity. The process begins with the synthesis of the Cu/g-C3N4 catalyst, which involves dispersing carbon nitride in DMF, treating it with isonicotinic acid chloride, and subsequently doping it with copper sulfate. Once the catalyst is prepared and characterized, the main reaction is conducted by mixing the trifluoroethylimidoyl chloride, the chosen amine, potassium carbonate, and the catalyst in DMF. The mixture is heated to approximately 80°C and stirred for 24 hours to ensure complete conversion. Following the reaction, the heterogeneous catalyst is removed by filtration, and the filtrate is concentrated and purified via standard column chromatography to isolate the high-purity product. Detailed standardized synthetic steps and specific stoichiometric ratios are outlined in the technical guide below.
- Prepare the heterogeneous catalyst by doping copper onto graphitic carbon nitride (g-C3N4) using copper sulfate pentahydrate and isonicotinic acid chloride.
- Mix trifluoroethylimidoyl chloride, amine, potassium carbonate, and the Cu/g-C3N4 catalyst in DMF solvent within a reaction vessel.
- Heat the mixture to 80°C for 24 hours, then filter to recover the catalyst and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this heterogeneous catalytic method offers profound advantages for procurement managers and supply chain heads focused on cost reduction in API manufacturing and operational efficiency. The primary economic driver is the elimination of expensive homogeneous catalysts and the associated downstream purification costs. Since the catalyst is solid and recyclable, the need for complex metal scavenging resins or extensive aqueous workups is removed, leading to significantly reduced solvent usage and waste disposal fees. Furthermore, the starting materials, such as trifluoroethylimidoyl chloride and various amines, are commodity chemicals that are readily available from multiple global suppliers, mitigating supply chain risks associated with proprietary or scarce reagents. This accessibility ensures a stable and continuous supply of raw materials, which is critical for maintaining production schedules and meeting delivery commitments to downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The implementation of a recyclable heterogeneous catalyst directly impacts the bottom line by lowering the cost per kilogram of the final intermediate. By avoiding the use of precious metal ligands and simplifying the purification train, manufacturers can achieve substantial cost savings without compromising on quality. The mild reaction conditions also contribute to lower energy consumption compared to high-pressure or high-temperature alternatives. Additionally, the high yields reported, often exceeding 90% for a wide range of substrates, minimize raw material waste and maximize the output from each batch, further enhancing the overall economic viability of the process for large-scale commercial production.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route translates into greater reliability for the supply chain. The use of stable, non-sensitive reagents and a catalyst that can be stored and reused reduces the complexity of inventory management. The broad substrate scope means that a single production line can be adapted to manufacture various derivatives of 2-trifluoromethyl benzimidazoles simply by changing the amine input, providing flexibility to respond to changing market demands. This versatility reduces the need for dedicated equipment for different products, optimizing capital expenditure and improving the agility of the manufacturing facility to handle diverse customer orders efficiently.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial scale is facilitated by the simplicity of the unit operations involved, primarily stirring, heating, and filtration. The heterogeneous nature of the catalyst aligns well with green chemistry principles by reducing metal contamination in waste streams, thereby easing the burden on environmental treatment facilities. The ability to recycle the catalyst multiple times significantly reduces the volume of solid waste generated. This environmental compliance is increasingly important for maintaining regulatory approvals and meeting the sustainability goals of modern pharmaceutical companies, making this method a preferred choice for long-term strategic partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this heterogeneous catalytic technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on the practical aspects of adopting this synthesis route. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios and assess the potential return on investment. The answers reflect the consensus on the method's capability to deliver high-purity intermediates while adhering to strict quality and safety standards required in the fine chemical industry.
Q: What are the advantages of using Cu/g-C3N4 over homogeneous copper catalysts?
A: The heterogeneous Cu/g-C3N4 catalyst allows for simple filtration and recycling, eliminating the need for complex and costly heavy metal removal processes required with homogeneous catalysts, while maintaining high catalytic efficiency over multiple cycles.
Q: What is the substrate scope for this benzimidazole synthesis method?
A: The method demonstrates excellent tolerance for various functional groups, successfully synthesizing derivatives with alkyl, halogen (Cl, Br), and trifluoromethyl substituents on the aromatic ring, as well as diverse aliphatic and aromatic amines.
Q: How does this process impact commercial scalability?
A: The use of cheap, commercially available starting materials, mild reaction conditions (80°C), and a recyclable catalyst significantly simplifies the post-treatment workflow, making the process highly suitable for large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and risk-mitigated. We are committed to delivering high-purity 2-trifluoromethyl benzimidazole derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in heterogeneous catalysis allows us to optimize this specific patent-protected route for maximum yield and minimal environmental impact, providing our partners with a competitive edge in the global market.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for custom synthesis projects. By partnering with us, you gain access to a reliable supply chain partner dedicated to innovation, quality, and sustainable growth in the pharmaceutical intermediate sector.
