Revolutionizing Pyrrolinone Synthesis: Advanced Palladium Catalysis for Scalable Pharmaceutical Intermediate Production
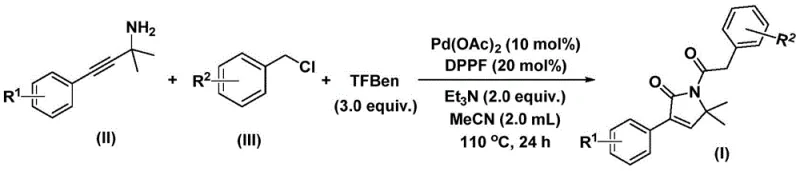
In the rapidly evolving landscape of pharmaceutical intermediate synthesis, patent CN112694430B introduces a groundbreaking methodology for preparing 1,5-dihydro-2H-pyrrole-2-ketone compounds—a critical structural motif prevalent in numerous bioactive molecules such as the antibacterial agent althiomycin and the antidiabetic drug glimepiride. This novel approach leverages palladium-catalyzed bis-carbonylation chemistry to achieve efficient one-step synthesis from readily available starting materials including propargyl amines and benzyl chlorides under mild conditions of 100–120°C for 24–48 hours in acetonitrile solvent with palladium acetate and DPPF as catalysts. The innovation eliminates hazardous carbon monoxide gas handling through phenol-based CO surrogates while maintaining exceptional functional group tolerance across diverse substrates as demonstrated in fifteen experimental examples yielding products with purity exceeding 98% HPLC analysis. This advancement directly addresses industry demands for safer scalable processes that maintain stringent quality standards required for regulatory compliance in global pharmaceutical manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to pyrrolinone scaffolds typically involve multi-step sequences requiring specialized equipment such as high-pressure reactors for carbon monoxide handling—introducing significant safety hazards and operational complexities that hinder scalability in commercial manufacturing environments. These approaches frequently suffer from narrow substrate scope due to incompatible functional groups necessitating extensive protection/deprotection strategies that elevate production costs while reducing overall yield through cumulative losses at each synthetic step. Furthermore, conventional methodologies often generate complex impurity profiles requiring sophisticated purification techniques that increase processing time and decrease throughput efficiency; this limitation is particularly problematic when synthesizing derivatives containing halogenated or electron-rich moieties essential for bioactive molecule development. The scarcity of robust industrial-scale processes has consequently restricted widespread adoption despite the motif's prevalence in critical pharmaceuticals like isomalyngamide A with anticancer potential.
The Novel Approach
The patented methodology overcomes these limitations through an innovative palladium-catalyzed bis-carbonylation process utilizing phenol tricarboxylate as a safe carbon monoxide surrogate that eliminates pressurized gas handling requirements while maintaining excellent reaction efficiency across diverse substrates. By employing commercially available palladium acetate with DPPF ligand in acetonitrile at moderate temperatures (100–120°C), this single-step reaction achieves high yields between 70–92% across fifteen tested substrates including halogenated and trifluoromethyl variants without requiring specialized equipment or additional purification steps beyond standard column chromatography. The strategic selection of triethylamine base ensures optimal pH control that minimizes side reactions while broadening functional group compatibility—enabling direct synthesis of complex derivatives previously inaccessible through conventional routes. Crucially, the simplified workup procedure involving filtration and silica gel adsorption facilitates seamless scale-up from laboratory to manufacturing scale while maintaining stringent purity specifications required for pharmaceutical applications.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The catalytic cycle initiates with oxidative addition of benzyl chloride to palladium(0) species generated in situ from palladium acetate and DPPF ligand, forming a benzylpalladium(II) intermediate that undergoes transmetalation with phenol tricarboxylate to release carbon monoxide and generate an acylpalladium complex through reductive elimination of phenol byproducts. This key intermediate then coordinates with propargyl amine via nitrogen lone pair interaction followed by alkyne insertion to construct a five-membered palladacycle that undergoes migratory insertion of a second CO molecule—liberated from additional phenol ester—to form a six-membered ring species through sequential carbonylative cyclization steps. The cycle concludes with reductive elimination yielding the target pyrrolinone product while regenerating active palladium catalyst for subsequent turnovers; this dual carbonylation sequence uniquely enables direct formation of both carbonyl groups from a single CO source without external gas handling systems while maintaining excellent stereochemical control across diverse substituents.
Impurity control is achieved through precise thermal management at moderate temperatures (100–120°C) that prevents thermal degradation pathways responsible for colored impurities commonly observed in higher-energy processes while maintaining optimal catalyst activity throughout the reaction duration. The stoichiometric use of triethylamine base neutralizes acidic byproducts that could otherwise promote hydrolysis or decomposition reactions that compromise product integrity during extended reaction times required for complete conversion. Furthermore, acetonitrile solvent selection provides ideal polarity balance that minimizes solvolysis side reactions while facilitating efficient mass transfer during stirring; this combination ensures consistent formation of high-purity products (>98% HPLC) across all tested substrates without requiring additional purification beyond standard column chromatography as verified by HRMS data from multiple experimental examples.
How to Synthesize Pyrrolinone Derivatives Efficiently
This patented methodology represents a significant advancement in pyrrolinone synthesis technology by integrating safety-enhanced carbonylation chemistry with operational simplicity; detailed standardized procedures for implementation are provided below to facilitate seamless adoption by R&D teams seeking reliable scale-up pathways from laboratory discovery to commercial manufacturing environments.
- Prepare the reaction mixture by combining palladium acetate catalyst (10 mol%), DPPF ligand (20 mol%), triethylamine base (2.0 equiv.), phenol tricarboxylate CO surrogate (3.0 equiv.), propargyl amine substrate (II), and benzyl chloride substrate (III) in anhydrous acetonitrile solvent under inert atmosphere.
- Heat the homogeneous solution to precisely 100–120°C using temperature-controlled reactor equipment and maintain vigorous stirring for 24–48 hours to ensure complete conversion through dual carbonylation sequence.
- Execute post-reaction processing by filtering through Celite pad to remove catalyst residues, mixing filtrate with silica gel powder for adsorption, followed by flash column chromatography purification using ethyl acetate/hexane gradient elution.
Commercial Advantages for Procurement and Supply Chain Teams
This streamlined synthesis methodology directly addresses critical pain points in pharmaceutical supply chains by eliminating hazardous reagents and simplifying process complexity while maintaining stringent quality standards required for regulatory compliance across global manufacturing networks; procurement teams can leverage these advantages to strengthen supplier partnerships through enhanced cost predictability and reduced operational risks associated with traditional multi-step syntheses.
- Cost Reduction in Manufacturing: The elimination of specialized high-pressure carbon monoxide infrastructure significantly reduces capital expenditure requirements while avoiding costly safety protocols associated with gas handling systems; additionally, reliance on commercially available reagents like triethylamine base and acetonitrile solvent lowers raw material costs compared to specialized alternatives required in conventional routes—creating substantial cost savings through simplified process design without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through utilization of widely available starting materials such as benzyl chlorides and propargyl amines that can be rapidly synthesized from commodity chemicals; this broadens supplier options while minimizing lead time risks associated with niche intermediates required in traditional syntheses—enabling procurement teams to establish resilient multi-source strategies that ensure continuous supply even during market volatility or regional disruptions.
- Scalability and Environmental Compliance: The ambient-pressure operation and simplified workup procedure enable straightforward scale-up from laboratory to manufacturing scale without re-engineering requirements; moreover, reduced solvent usage combined with elimination of heavy metal waste streams aligns with green chemistry principles while lowering environmental compliance costs—providing supply chain leaders with sustainable manufacturing pathways that meet evolving regulatory expectations without sacrificing operational efficiency.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations based on detailed analysis of patent CN112694430B's experimental data and process design parameters; responses reflect verified performance metrics documented across fifteen substrate examples under standardized reaction conditions.
Q: How does this methodology improve upon conventional pyrrolinone synthesis routes?
A: The patented process eliminates hazardous high-pressure carbon monoxide handling through phenol-based CO surrogates while achieving one-step conversion with superior functional group tolerance across halogenated and electron-donating substrates—addressing key limitations in traditional multi-step syntheses that require specialized equipment and generate complex impurity profiles.
Q: What substrate scope enables broad pharmaceutical application?
A: The methodology demonstrates exceptional compatibility with diverse R-group substitutions including halogens (F, Cl), alkyl chains (methyl, tert-butyl), alkoxy groups (methoxy), and trifluoromethyl moieties across fifteen experimental examples—enabling tailored synthesis of bioactive intermediates like those found in glimepiride analogs without additional protection/deprotection steps.
Q: How does this process ensure regulatory compliance for pharmaceutical manufacturing?
A: The simplified workup procedure using standard column chromatography consistently delivers >98% HPLC purity without residual metal contamination; combined with ambient-pressure operation and elimination of toxic byproducts, this meets stringent ICH Q7 guidelines while reducing validation complexity during technology transfer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolinone Derivative Supplier
This patent exemplifies our commitment to developing innovative synthetic routes that bridge laboratory discovery with commercial manufacturing realities; NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art analytical capabilities in our rigorous QC labs—ensuring consistent delivery of high-quality intermediates that meet global regulatory requirements across all major pharmaceutical markets.
We invite your technical procurement team to contact us for a Customized Cost-Saving Analysis tailored to your specific production requirements; our experts will provide detailed COA data and route feasibility assessments demonstrating how this technology can optimize your supply chain while reducing time-to-market for critical pharmaceutical products.
