Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrole-2-One Scaffolds for Commercial Production
Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrole-2-One Scaffolds for Commercial Production
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and scalable synthetic routes. A significant breakthrough in this domain is detailed in patent CN112694430B, which discloses a novel preparation method for 1,5-dihydro-2H-pyrrole-2-one compounds. This structural backbone is of paramount importance in medicinal chemistry, serving as the core scaffold for potent bioactive molecules such as the antibiotic althiomycin, the hypoglycemic agent glimepiride, and the anticancer candidate isomalyngamide A. The ability to construct this heterocyclic system efficiently opens new avenues for drug discovery and process development. The patented methodology leverages a palladium-catalyzed bis-carbonylation strategy, transforming simple starting materials into complex heterocycles with remarkable precision.
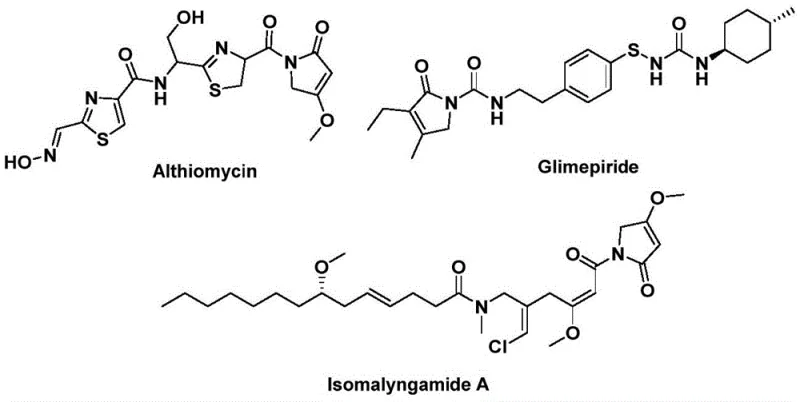
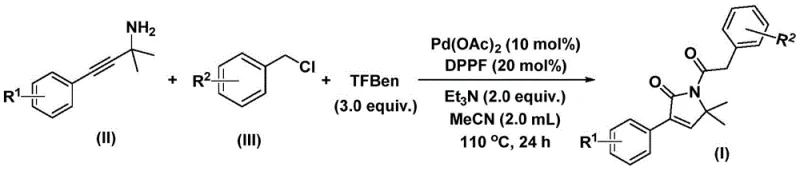
For R&D directors and process chemists, the significance of this technology lies in its operational simplicity and robust substrate compatibility. Unlike traditional methods that may require harsh conditions or multiple protection-deprotection steps, this approach utilizes a direct coupling of propargyl amines and benzyl chlorides. The reaction proceeds smoothly in common organic solvents like acetonitrile, utilizing a solid carbon monoxide substitute (1,3,5-tricarboxylic acid phenol ester) rather than toxic CO gas. This not only enhances safety profiles but also simplifies the engineering requirements for commercial scale-up, positioning this technology as a highly attractive option for reliable pharmaceutical intermediate suppliers seeking to optimize their production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,5-dihydro-2H-pyrrol-2-one skeletons has been fraught with challenges that hinder efficient commercial production. Conventional routes often rely on the cyclization of gamma-amino acids or the condensation of beta-keto esters with amines, processes that frequently suffer from poor atom economy and the generation of substantial chemical waste. Furthermore, many existing methodologies necessitate the use of gaseous carbon monoxide under high pressure, which introduces significant safety hazards and requires specialized, expensive reactor infrastructure that is not universally available in standard fine chemical plants. Additionally, traditional pathways often exhibit limited substrate scope, struggling to accommodate diverse functional groups on the aromatic rings without compromising yield or selectivity. These limitations result in higher manufacturing costs, extended lead times, and a restricted ability to rapidly generate analog libraries for structure-activity relationship (SAR) studies, creating a bottleneck in the early stages of drug development.
The Novel Approach
In stark contrast, the novel approach described in the patent data revolutionizes the synthesis of these valuable intermediates through a streamlined, one-pot palladium-catalyzed bis-carbonylation. By employing benzyl chlorides and propargyl amines as the primary building blocks, the method bypasses the need for pre-functionalized precursors, thereby reducing the overall step count and material costs. The use of 1,3,5-tricarboxylic acid phenol ester (TFBen) as a safe and convenient solid source of carbon monoxide eliminates the logistical and safety burdens associated with handling CO gas. This innovative strategy ensures high reaction efficiency and excellent yields across a broad range of substrates, including those with electron-donating and electron-withdrawing substituents. The operational simplicity, combined with the use of standard laboratory reagents and mild reaction conditions, makes this method exceptionally suitable for cost reduction in pharmaceutical intermediate manufacturing, allowing producers to deliver high-purity compounds with greater speed and reliability.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation Cyclization
The mechanistic pathway of this transformation is a sophisticated orchestration of organometallic steps that ensures the precise construction of the five-membered lactam ring. The cycle initiates with the oxidative addition of the palladium(0) catalyst into the carbon-chlorine bond of the benzyl chloride, generating a reactive benzyl-palladium intermediate. Subsequently, carbon monoxide, which is liberated in situ from the thermal decomposition of the phenol 1,3,5-tricarboxylate additive, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the amino group of the propargyl amine, followed by coordination of the alkyne moiety to the metal center. This sequence sets the stage for the crucial cyclization event, where the alkyne inserts into the palladium-nitrogen or palladium-carbon bond, forming a five-membered ring palladium intermediate. A second insertion of carbon monoxide expands the coordination sphere, eventually leading to reductive elimination that releases the final 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active palladium catalyst for the next turnover.
Understanding this mechanism is critical for impurity control and process optimization. The choice of ligand, specifically bidentate phosphines like 1,1'-bis(diphenylphosphino)ferrocene (DPPF), plays a pivotal role in stabilizing the palladium intermediates and facilitating the sequential CO insertions. The presence of a base, such as triethylamine, is essential to neutralize the hydrochloric acid byproduct generated during the oxidative addition step, preventing catalyst deactivation and ensuring the reaction proceeds to completion. Furthermore, the controlled release of CO from the solid surrogate helps maintain an optimal concentration of the gas in the solution, minimizing side reactions such as homocoupling of the benzyl chloride or polymerization of the alkyne. This precise control over the catalytic cycle results in a clean reaction profile with minimal byproduct formation, which is a key requirement for producing high-purity pharmaceutical intermediates that meet stringent regulatory standards.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
The execution of this synthesis protocol is designed for reproducibility and ease of handling, making it accessible for both laboratory-scale optimization and pilot plant operations. The procedure involves charging a reaction vessel with the palladium catalyst, ligand, base, CO source, and the two primary substrates in acetonitrile. The mixture is then heated to a temperature range of 100-120°C, typically around 110°C, and maintained for a period of 24 to 48 hours to ensure full conversion. Following the reaction, the workup is straightforward, involving filtration to remove insoluble palladium residues and silica gel treatment, followed by standard column chromatography purification. For detailed operational parameters and specific stoichiometric ratios optimized for various substrates, please refer to the standardized synthesis guide below.
- Combine palladium acetate, DPPF ligand, triethylamine, TFBen, propargyl amine, and benzyl chloride in acetonitrile solvent.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 1,5-dihydro-2H-pyrrole-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers transformative benefits that directly impact the bottom line and operational resilience. The shift towards this methodology addresses several critical pain points associated with the sourcing and production of complex heterocyclic intermediates. By simplifying the synthetic route and utilizing commodity chemicals, manufacturers can achieve significant improvements in cost efficiency and supply security. The robustness of the reaction conditions ensures consistent quality output, reducing the risk of batch failures and delivery delays that often plague complex chemical syntheses. This reliability is paramount for maintaining uninterrupted production schedules in the fast-paced pharmaceutical industry.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the utilization of inexpensive and widely available starting materials such as benzyl chlorides and propargyl amines. Unlike traditional routes that may require exotic reagents or multi-step sequences, this one-pot approach drastically reduces the consumption of solvents, energy, and labor hours. The elimination of high-pressure gas equipment further lowers capital expenditure (CAPEX) and operational expenditure (OPEX) related to safety compliance and maintenance. Consequently, the overall cost of goods sold (COGS) for the final intermediate is substantially lowered, providing a competitive pricing advantage in the global market without compromising on quality or purity specifications.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of stable, shelf-stable reagents that are easily sourced from multiple global vendors. The reliance on a solid CO surrogate instead of gaseous carbon monoxide removes a major logistical bottleneck, as there is no need for specialized gas cylinders or on-site generation units. This flexibility allows for manufacturing in a wider range of facilities, diversifying the supply base and mitigating the risk of regional disruptions. Furthermore, the high yield and conversion rates reported in the patent data imply that less raw material is required to produce a given amount of product, optimizing inventory management and reducing the strain on upstream supply chains for precious starting materials.
- Scalability and Environmental Compliance: The environmental footprint of this process is significantly reduced compared to conventional methods, aligning with modern green chemistry principles and increasingly strict regulatory requirements. The use of acetonitrile, a solvent that can be readily recovered and recycled, minimizes waste generation. Additionally, the avoidance of toxic CO gas enhances workplace safety and simplifies environmental permitting processes. The reaction's scalability is evidenced by its tolerance to various functional groups and its operation at moderate temperatures, which facilitates a smooth transition from gram-scale laboratory synthesis to multi-kilogram or ton-scale commercial production. This scalability ensures that the supply chain can rapidly respond to increased demand as drug candidates progress through clinical trials to market launch.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled a set of frequently asked questions based on the patent specifications and practical application scenarios. These insights address common concerns regarding reaction scope, safety, and scalability, providing a comprehensive overview for stakeholders evaluating this technology for their supply chains. Understanding these details is crucial for making informed decisions about adopting this novel route for the production of key pharmaceutical building blocks.
Q: What are the key advantages of this palladium-catalyzed method over traditional synthesis?
A: This method utilizes readily available benzyl chlorides and propargyl amines in a one-pot reaction, eliminating the need for pre-functionalized substrates and reducing step count significantly compared to classical multi-step routes.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
A: Yes, the protocol uses standard organic solvents like acetonitrile and operates at moderate temperatures (100-120°C), making it highly amenable to scale-up in standard stainless steel reactors without requiring specialized high-pressure carbonylation equipment.
Q: What is the substrate scope for R1 and R2 groups in this reaction?
A: The reaction demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens (F, Cl, Br) and trifluoromethyl groups on both aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the palladium-catalyzed bis-carbonylation described in CN112694430B. As a leading CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped to handle complex organometallic reactions safely and efficiently, adhering to stringent purity specifications and rigorous QC labs protocols. We are committed to leveraging such innovative technologies to deliver high-purity 1,5-dihydro-2H-pyrrole-2-one derivatives that meet the exacting standards of the global pharmaceutical industry.
We invite potential partners to engage with our technical team to explore how this cost-effective and scalable route can be integrated into your specific project requirements. Whether you are in the early stages of process development or looking to secure a long-term supply for commercial manufacturing, our experts are ready to provide a Customized Cost-Saving Analysis tailored to your needs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and quotations that demonstrate the tangible benefits of partnering with a supplier dedicated to innovation and excellence.
