Advanced Pd-Catalyzed Bis-Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrol-2-One Manufacturing
Introduction to Novel Pyrrol-2-One Synthesis Technology
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access privileged structural scaffolds efficiently. A significant breakthrough in this domain is detailed in Chinese Patent CN112694430B, which discloses a highly efficient preparation method for 1,5-dihydro-2H-pyrrol-2-one compounds. This structural motif is not merely an academic curiosity but a critical backbone found in numerous bioactive natural products and therapeutic agents. For instance, the antibiotic althiomycin, the hypoglycemic agent glimepiride, and the anticancer candidate isomalyngamide A all share this core architecture, highlighting its immense value in drug discovery and development. 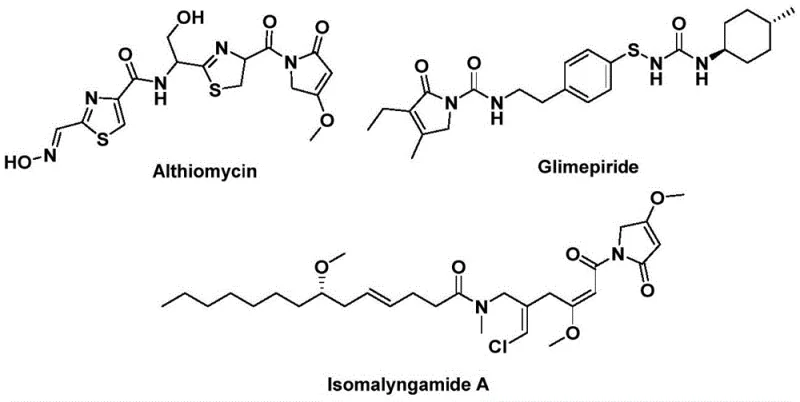 . The patented technology leverages a palladium-catalyzed bis-carbonylation strategy, utilizing propargyl amines and benzyl chlorides as readily available starting materials. By employing a solid carbon monoxide substitute, the process circumvents the logistical and safety nightmares associated with gaseous CO, offering a streamlined, one-step route that is ideally suited for reliable pharmaceutical intermediate supplier operations aiming for high purity and scalability.
. The patented technology leverages a palladium-catalyzed bis-carbonylation strategy, utilizing propargyl amines and benzyl chlorides as readily available starting materials. By employing a solid carbon monoxide substitute, the process circumvents the logistical and safety nightmares associated with gaseous CO, offering a streamlined, one-step route that is ideally suited for reliable pharmaceutical intermediate supplier operations aiming for high purity and scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of the 1,5-dihydro-2H-pyrrol-2-one ring system has often relied on multi-step synthetic sequences that are fraught with inefficiencies. Classical approaches might involve the use of hazardous carbon monoxide gas under high pressure, requiring specialized autoclaves and rigorous safety protocols that drive up capital expenditure and operational complexity. Furthermore, alternative cyclization strategies often suffer from poor atom economy, generating substantial stoichiometric waste, or they require harsh reaction conditions that limit the tolerance of sensitive functional groups. These limitations create bottlenecks in cost reduction in API manufacturing, as the need for extensive purification steps and the handling of dangerous reagents inflate the final cost of goods. Additionally, the reliance on gaseous reagents introduces supply chain volatility, where interruptions in gas supply can halt production lines, posing a significant risk to supply chain heads responsible for continuity.
The Novel Approach
In stark contrast, the methodology described in the patent introduces a transformative one-pot synthesis that elegantly resolves these historical challenges. By utilizing 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid, bench-stable carbon monoxide surrogate, the process allows for the safe and controlled release of CO in situ. This innovation enables the reaction to proceed in standard glassware or reactors without the need for high-pressure equipment, drastically simplifying the engineering requirements. The reaction couples propargyl amines and benzyl chlorides directly, forging two new carbon-carbon bonds and the lactam ring in a single operation. 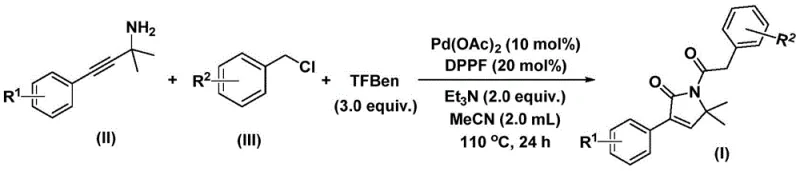 . This telescoped approach not only accelerates the timeline from raw material to product but also enhances the overall yield by minimizing intermediate isolation losses. The broad substrate scope, accommodating various electronic and steric environments, ensures that this method is versatile enough for the commercial scale-up of complex pharmaceutical intermediates, providing a distinct competitive advantage in the market.
. This telescoped approach not only accelerates the timeline from raw material to product but also enhances the overall yield by minimizing intermediate isolation losses. The broad substrate scope, accommodating various electronic and steric environments, ensures that this method is versatile enough for the commercial scale-up of complex pharmaceutical intermediates, providing a distinct competitive advantage in the market.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
Understanding the catalytic cycle is paramount for R&D directors focused on impurity control and process optimization. The reaction initiates with the oxidative addition of the palladium(0) catalyst into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium(II) intermediate. Subsequently, carbon monoxide, liberated thermally from the TFBen precursor, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes a nucleophilic attack by the internal alkyne of the propargyl amine, triggering a cyclization event that constructs the five-membered heterocyclic core. Following this cyclization, a second molecule of carbon monoxide inserts into the newly formed organopalladium bond, expanding the coordination sphere. Finally, reductive elimination releases the desired 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active palladium(0) catalyst, closing the loop. This intricate dance of insertion and elimination steps is highly sensitive to ligand choice; the patent specifies the use of DPPF (1,1'-bis(diphenylphosphino)ferrocene), which provides the optimal bite angle and electronic properties to facilitate these sequential transformations efficiently.
From an impurity profiling perspective, the mechanism suggests that side reactions could arise from incomplete carbonylation or homocoupling of the benzyl chloride. However, the use of a slight excess of the solid CO source (3.0 equivalents relative to the amine) drives the equilibrium towards the fully carbonylated product, suppressing partially reacted intermediates. The choice of acetonitrile as the solvent is also critical, as it stabilizes the polar transition states and ensures solubility of the ionic intermediates. By maintaining the reaction temperature at 110°C, the system provides sufficient thermal energy to overcome the activation barriers for the second CO insertion, which is often the rate-limiting step in bis-carbonylation reactions. This precise control over reaction parameters ensures that the final high-purity pharmaceutical intermediate meets stringent quality specifications, minimizing the burden on downstream purification teams.
How to Synthesize 1,5-Dihydro-2H-Pyrrol-2-One Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot plant production. The protocol involves charging a reaction vessel with the palladium catalyst, the bidentate phosphine ligand, the solid CO source, a base, and the two primary organic substrates in acetonitrile. The mixture is then heated to reflux conditions for a defined period to ensure complete conversion.
- Combine palladium acetate, DPPF ligand, triethylamine, solid CO source (TFBen), propargyl amine, and benzyl chloride in acetonitrile solvent.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to ensure complete conversion.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target pyrrol-2-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the economic implications of this patented process are profound. The shift from hazardous gaseous reagents to stable solids fundamentally alters the risk profile and cost structure of manufacturing these valuable intermediates. By eliminating the need for specialized high-pressure infrastructure and the associated safety audits, facilities can repurpose existing equipment, leading to immediate capital efficiency. Moreover, the use of commodity chemicals like benzyl chlorides and simple propargyl amines ensures a stable and diversified supply base, reducing the risk of single-source dependency. The high yields reported in the patent examples, often exceeding 80% and reaching up to 92%, translate directly into better material utilization and reduced waste disposal costs, aligning perfectly with green chemistry initiatives and sustainability goals.
- Cost Reduction in Manufacturing: The consolidation of multiple synthetic steps into a single pot operation significantly reduces labor hours, solvent consumption, and energy usage per kilogram of product. By avoiding the isolation of unstable intermediates, the process minimizes material loss and simplifies the workflow. The use of a solid CO source removes the expensive logistics of gas cylinder management and leak detection systems. Consequently, the overall cost of goods sold is substantially lowered, allowing for more competitive pricing strategies in the global marketplace without compromising on quality margins.
- Enhanced Supply Chain Reliability: The reliance on shelf-stable reagents means that raw materials can be stocked in bulk without degradation concerns, buffering the production schedule against short-term market fluctuations. The robustness of the reaction conditions, which tolerate a wide range of substituents, allows for flexible sourcing of starting materials; if one supplier of a specific substituted benzyl chloride faces issues, alternatives can often be sourced without re-validating the entire process. This flexibility is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API synthesis schedules are met consistently.
- Scalability and Environmental Compliance: The reaction generates minimal hazardous waste compared to traditional methods that might produce heavy metal sludge or acidic off-gases. The workup procedure, involving simple filtration and chromatography, is easily adaptable to continuous processing or large-batch operations. The absence of toxic gas emissions simplifies environmental permitting and monitoring, facilitating faster regulatory approval for new manufacturing sites. This scalability ensures that as demand for the final drug product grows, the supply of this critical intermediate can be ramped up seamlessly from kilograms to tons.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis platform, derived directly from the patent specifications and experimental data.
Q: What are the safety advantages of using TFBen over CO gas in this synthesis?
A: Using 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide substitute eliminates the need for handling toxic, high-pressure CO gas cylinders, significantly improving operational safety and simplifying reactor requirements for scale-up.
Q: What is the substrate compatibility of this Pd-catalyzed method?
A: The method demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as fluoro, chloro, bromo, and trifluoromethyl on both the amine and benzyl chloride substrates.
Q: How does this one-step approach impact production costs compared to traditional routes?
A: By consolidating the synthesis into a single pot with commercially available starting materials and avoiding complex multi-step sequences or hazardous gas handling, the process reduces unit operations, lowers labor costs, and minimizes waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrol-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for complex heterocyclic scaffolds. Our technical team has thoroughly analyzed the potential of this Pd-catalyzed bis-carbonylation technology and is prepared to implement it within our state-of-the-art facilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs equipped with advanced analytical instrumentation, guaranteeing that every batch of 1,5-dihydro-2H-pyrrol-2-one intermediate meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next drug development program. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you accelerate your pipeline with reliable, cost-effective, and high-quality chemical solutions.
