Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access nitrogen-containing heterocycles, particularly those enhanced with fluorine motifs for improved metabolic stability. Patent CN113045503B introduces a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in current synthetic workflows. This technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing cheap and readily available trifluoroethylimidoyl chloride and amines as primary starting materials. The significance of this innovation lies in its ability to construct the quinazolinone core while simultaneously installing the valuable trifluoromethyl group in a single operational sequence. For R&D directors and process chemists, this represents a paradigm shift from multi-step, low-yielding protocols to a streamlined, high-efficiency pathway that is immediately applicable to the synthesis of complex drug candidates and bioactive scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl-substituted quinazolinones has been plagued by significant operational and economic hurdles that hinder large-scale adoption. Traditional literature methods often rely on the cyclization of anthranilamide with ethyl trifluoroacetate, trifluoroacetic anhydride, or trifluoroacetic acid under varying conditions, which frequently require harsh reaction environments and extended processing times. Alternative routes involving anthranilates and unstable trifluoroacetamides suffer from substrate instability and difficult handling procedures, leading to inconsistent batch quality. Furthermore, methods utilizing isatoic anhydride or T3P-promoted cascade reactions are often limited by the high cost of pre-activated substrates and narrow substrate scope, failing to accommodate diverse functional groups without extensive protection-deprotection strategies. These conventional approaches typically result in lower overall yields and generate substantial chemical waste, making them economically unviable for the cost reduction in pharmaceutical intermediate manufacturing required by modern supply chains.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a palladium-catalyzed carbonylation strategy that fundamentally simplifies the synthetic architecture. By employing trifluoroethylimidoyl chloride and simple amines, the process bypasses the need for expensive pre-activated precursors. The reaction proceeds efficiently at 110°C in common organic solvents like 1,4-dioxane, utilizing TFBen as a safe, solid carbon monoxide surrogate instead of hazardous high-pressure CO gas. This approach not only enhances safety profiles but also dramatically improves reaction efficiency and substrate compatibility, allowing for the introduction of various substituents such as halogens, alkyl groups, and trifluoromethyl moieties without compromising yield. The versatility of this system is evidenced by its successful application in synthesizing a wide array of derivatives, positioning it as a superior choice for reliable pharmaceutical intermediate supplier networks seeking scalable solutions.
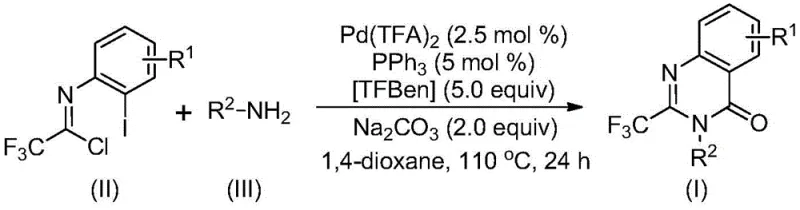
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation offers profound insights into its high efficiency and selectivity, which are crucial for R&D teams optimizing impurity profiles. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond coupling between the amine and the imidoyl chloride, generating a trifluoroacetamidine derivative in situ. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the aromatic ring, forming a reactive divalent palladium intermediate. A critical step involves the thermal decomposition of TFBen, which releases carbon monoxide directly into the reaction medium; this CO then inserts into the carbon-palladium bond to form an acyl-palladium species. Under the influence of the base, this intermediate undergoes intramolecular cyclization to form a seven-membered ring palladium complex, followed by reductive elimination to release the final 2-trifluoromethyl-substituted quinazolinone product. This elegant cascade minimizes the formation of side products typically associated with stepwise syntheses, ensuring a cleaner crude reaction mixture.
Furthermore, the successful application of this chemistry extends beyond simple model substrates to complex bioactive molecules, demonstrating its robustness in real-world scenarios. A prime example is the total synthesis of Rutaecarpine, a potent natural product with significant pharmacological potential. The patent details a streamlined three-step sequence where the key quinazolinone intermediate is constructed using this novel carbonylation method, followed by acid-mediated cyclization and base treatment. This sequence achieved an impressive overall yield of 77%, validating the method's utility for the commercial scale-up of complex pharmaceutical intermediates. The ability to tolerate sensitive functional groups during the carbonylation step ensures that the structural integrity of complex drug candidates is maintained, reducing the need for costly purification steps and enhancing the overall process mass intensity.
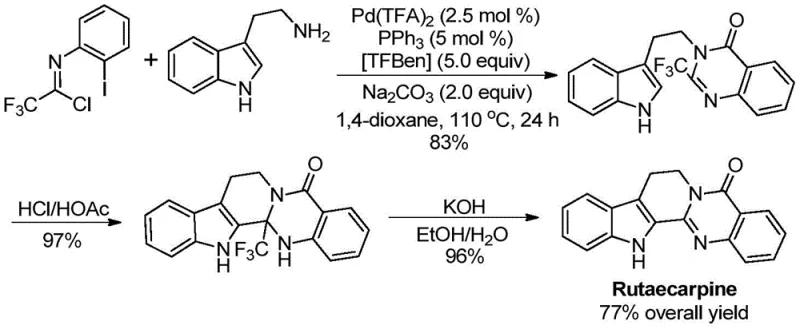
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Implementing this synthesis protocol requires precise control over reaction parameters to maximize yield and purity, yet the procedure remains remarkably straightforward for industrial adaptation. The process involves charging a reactor with the specific molar ratios of palladium trifluoroacetate, triphenylphosphine, and sodium carbonate, followed by the addition of the trifluoroethylimidoyl chloride and the desired amine substrate in 1,4-dioxane. The mixture is then heated to 110°C for a duration of 16 to 30 hours, allowing the carbonylation cascade to reach completion. Detailed standardized synthesis steps, including specific workup procedures like filtration and silica gel treatment, are outlined in the technical guide below to ensure reproducibility across different production scales.
- Combine palladium trifluoroacetate, triphenylphosphine, TFBen, sodium carbonate, trifluoroethylimidoyl chloride, and amine in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 110°C and stir for 16 to 30 hours to allow the carbonylation cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative benefits that directly address the pain points of cost volatility and material availability in the fine chemical sector. By shifting away from exotic, pre-activated substrates to commodity chemicals like simple amines and imidoyl chlorides, the raw material cost structure is significantly optimized. The use of a solid CO surrogate eliminates the logistical complexities and safety infrastructure costs associated with handling toxic carbon monoxide gas, thereby reducing the barrier to entry for manufacturing facilities. Moreover, the high substrate compatibility means that a single production line can be utilized to manufacture a diverse library of quinazolinone derivatives, enhancing asset utilization rates and providing greater flexibility in responding to market demands for various API intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive pre-activated substrates and the use of catalytic amounts of palladium with inexpensive ligands drastically lowers the bill of materials. Additionally, the simplified workup procedure, which avoids complex extraction sequences in favor of direct filtration and chromatography, reduces solvent consumption and labor hours. This streamlined workflow translates to substantial cost savings in API manufacturing, allowing for more competitive pricing strategies in the global market without sacrificing margin.
- Enhanced Supply Chain Reliability: The starting materials, including trifluoroethylimidoyl chloride and various amines, are commercially available and widely produced, mitigating the risk of supply disruptions common with specialized reagents. The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with pharmaceutical clients. This reliability strengthens the supply chain resilience, ensuring that downstream production schedules for critical medications are not compromised by raw material shortages or synthesis failures.
- Scalability and Environmental Compliance: The reaction operates under atmospheric pressure using standard heating equipment, making the transition from gram-scale laboratory optimization to multi-ton commercial production seamless and low-risk. The use of 1,4-dioxane, a common industrial solvent, facilitates easy recovery and recycling, aligning with increasingly stringent environmental regulations. The high atom economy of the carbonylation cascade minimizes waste generation, supporting sustainability goals and reducing the costs associated with waste disposal and environmental compliance auditing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent's experimental data and beneficial effects. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the specific advantages of the palladium-catalyzed system over traditional methods, focusing on yield, safety, and applicability to drug synthesis.
Q: What are the advantages of using TFBen over carbon monoxide gas?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a solid carbon monoxide surrogate, eliminating the need for high-pressure CO gas equipment and significantly improving operational safety and ease of handling in standard laboratory or plant settings.
Q: What is the substrate scope for this synthesis method?
A: The method demonstrates excellent compatibility with various functional groups, including alkyl, halogen (F, Cl, Br), and trifluoromethyl substituents on the aromatic ring, as well as diverse amine structures ranging from linear alkyl to bulky aryl groups.
Q: Can this method be applied to complex drug molecules?
A: Yes, the patent explicitly demonstrates the successful application of this methodology in the efficient total synthesis of the bioactive natural product Rutaecarpine, achieving a high overall yield through a streamlined three-step sequence.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN113045503B for accelerating drug development pipelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone intermediate meets the highest industry standards for potency and impurity control.
We invite you to collaborate with our technical team to explore how this efficient carbonylation route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this novel process. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your unique synthesis challenges, ensuring a partnership built on transparency, quality, and mutual growth.
