Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
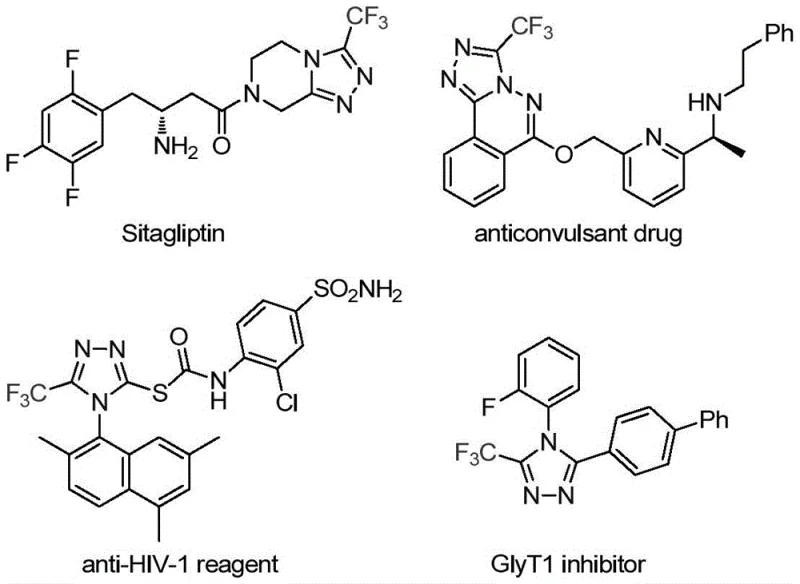
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which are pivotal for enhancing drug bioavailability and metabolic stability. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, utilizing a synergistic molybdenum and copper co-catalytic system. This innovation addresses critical bottlenecks in traditional synthesis by employing readily available trifluoroethylimidoyl chloride and functionalized isonitriles as starting materials. The process operates under mild thermal conditions, typically between 70°C and 90°C, and demonstrates exceptional functional group tolerance, making it an ideal candidate for the production of high-value pharmaceutical intermediates. As a leading manufacturer, we recognize the strategic importance of this technology in streamlining the supply chain for complex heterocyclic building blocks used in modern drug discovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on laborious multi-step sequences that often suffer from poor atom economy and harsh reaction conditions. Traditional routes frequently involve the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, which require stringent temperature controls and generate significant chemical waste. Furthermore, copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane, while effective, often involve hazardous reagents that pose safety risks during large-scale manufacturing. These legacy methods limit the structural diversity accessible to medicinal chemists and inflate the cost of goods due to low yields and complex purification requirements, creating a bottleneck for reliable pharmaceutical intermediate suppliers aiming to meet global demand.
The Novel Approach
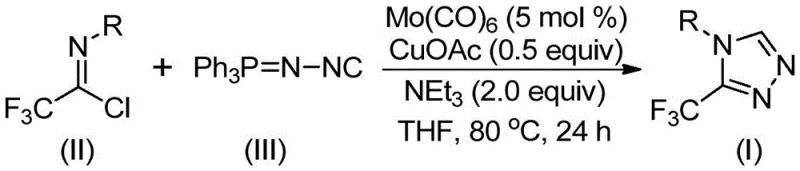
The methodology disclosed in CN113307778A represents a paradigm shift by leveraging a dual-metal catalytic system comprising molybdenum hexacarbonyl and cuprous acetate. This novel approach facilitates a direct [3+2] cycloaddition between trifluoroethylimidoyl chloride and functionalized isonitriles, bypassing the need for unstable diazo compounds or harsh hydrazine derivatives. The reaction proceeds efficiently in common aprotic solvents like THF, with triethylamine serving as a benign base to drive the transformation. By eliminating dangerous reagents and simplifying the operational workflow to a single pot, this method drastically reduces the environmental footprint and operational complexity. The ability to synthesize diverse 4-substituted 1,2,4-triazole compounds with high conversion rates underscores its superiority over prior art, offering a streamlined pathway for cost reduction in API manufacturing.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The mechanistic elegance of this transformation lies in the synergistic interaction between the molybdenum and copper species. Initially, molybdenum hexacarbonyl acts as a metal activator, coordinating with the functionalized isonitrile to form a reactive metal-isocyanide complex. This activation lowers the energy barrier for the subsequent nucleophilic attack. Concurrently, the cuprous acetate promotes the [3+2] cycloaddition of the activated isonitrile to the trifluoroethylimidoyl chloride, facilitating the formation of the five-membered triazole ring intermediate. The presence of water within the system, or generated during the process, aids in the elimination of triphenylphosphine oxide, driving the equilibrium towards the final 3-trifluoromethyl-substituted 1,2,4-triazole product. This intricate dance of metals ensures high regioselectivity and minimizes the formation of undesired byproducts, which is crucial for maintaining high purity specifications in pharmaceutical applications.
From an impurity control perspective, the choice of ligands and additives plays a vital role in suppressing side reactions. The use of molecular sieves in the reaction mixture helps to scavenge moisture that could otherwise hydrolyze the sensitive imidoyl chloride starting material, thereby preserving yield. Furthermore, the specific stoichiometry of triethylamine (typically 2.0 equivalents) ensures complete neutralization of the HCl byproduct without promoting decomposition of the triazole ring. The robustness of the catalytic cycle allows for a wide range of electronic variations on the aromatic ring of the starting imidoyl chloride, from electron-donating methyl groups to electron-withdrawing nitro groups, without significant loss in efficiency. This mechanistic stability translates directly to a cleaner crude reaction profile, simplifying downstream purification and ensuring consistent quality for high-purity OLED material or pharmaceutical intermediate batches.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential to maximize yield and minimize impurities. The protocol involves charging a reaction vessel with the catalyst system and substrates under an inert atmosphere to prevent oxidation of the sensitive metal centers. The standardized procedure outlined in the patent provides a robust framework for laboratory and pilot-scale operations, ensuring reproducibility across different batches. For detailed operational specifics regarding stoichiometry, solvent volumes, and workup procedures, please refer to the technical guide below which summarizes the optimized conditions derived from the patent data.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and functionalized isonitrile (Ph3P=N-NC) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C and stir for 18-30 hours, followed by filtration and column chromatography purification to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Mo/Cu co-catalyzed methodology offers tangible strategic benefits beyond mere chemical novelty. The shift towards this synthetic route addresses several pain points associated with the sourcing of complex heterocyclic intermediates, particularly regarding cost stability and lead time reliability. By utilizing commodity chemicals as catalysts and avoiding proprietary or hazardous reagents, manufacturers can secure a more resilient supply chain that is less susceptible to market volatility. The simplicity of the workup procedure, involving standard filtration and chromatography, further enhances throughput capabilities, allowing suppliers to respond more agilely to fluctuating demand from downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive and commercially available catalysts such as cuprous acetate and molybdenum hexacarbonyl, which eliminate the need for costly noble metals or specialized ligands. The high reaction efficiency and yields reported, often exceeding 90% for optimized substrates, mean that raw material consumption is minimized, directly lowering the cost per kilogram of the final active pharmaceutical ingredient intermediate. Additionally, the mild reaction temperatures reduce energy consumption compared to high-temperature reflux methods, contributing to substantial cost savings in utility expenses over long-term production runs.
- Enhanced Supply Chain Reliability: The starting materials, specifically trifluoroethylimidoyl chloride and functionalized isonitriles, are derived from abundant feedstocks and can be sourced from multiple global vendors, mitigating the risk of single-source dependency. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, ensuring that production timelines can be met even during periods of high demand. This reliability is critical for maintaining continuous manufacturing flows for key drugs like Sitagliptin, where interruptions in the supply of triazole intermediates could have cascading effects on the availability of finished dosage forms.
- Scalability and Environmental Compliance: The process has been validated at the gram level with clear pathways for scale-up to multi-kilogram and tonne scales, demonstrating excellent potential for commercial expansion. The use of THF as a primary solvent aligns with standard industrial practices for solvent recovery and recycling, reducing waste disposal costs. Furthermore, the avoidance of toxic diazo compounds and the generation of manageable byproducts like triphenylphosphine oxide simplify waste treatment protocols, ensuring compliance with increasingly stringent environmental regulations governing chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the method for industrial partners.
Q: What are the key advantages of the Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: The Mo/Cu co-catalyzed method described in patent CN113307778A offers significantly milder reaction conditions (70-90°C) compared to traditional harsh cyclization methods. It utilizes cheap and readily available starting materials like trifluoroethylimidoyl chloride and functionalized isonitriles, avoiding complex multi-step sequences and improving overall atom economy and reaction efficiency.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly demonstrates that the method can be expanded to gram-level reactions with high efficiency. The use of stable catalysts like molybdenum hexacarbonyl and cuprous acetate, along with common solvents like THF, suggests strong potential for commercial scale-up and continuous manufacturing processes required for API intermediate supply.
Q: What is the substrate scope for the R-group in this triazole synthesis?
A: The method exhibits broad substrate tolerance, accommodating various aryl and phenethyl groups. Successful examples include substituents such as methyl, tert-butyl, fluoro, chloro, methoxy, and nitro groups at ortho, meta, or para positions, allowing for the design of diverse 3-trifluoromethyl-1,2,4-triazole derivatives for medicinal chemistry applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising Mo/Cu co-catalyzed triazole synthesis can be seamlessly integrated into your supply chain. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole intermediate meets the exacting standards required for global pharmaceutical registration and drug development.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your product's competitiveness in the marketplace.
