Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Intermediates for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access bioactive heterocyclic scaffolds, particularly those with proven therapeutic potential. Patent CN112538054B introduces a groundbreaking preparation method for 1,2,4-triazole-3-one compounds, a privileged structural motif found in numerous bioactive molecules ranging from antifungal agents like Itraconazole to potent antitumor and anticonvulsant drugs. This technology represents a significant leap forward in synthetic methodology, addressing long-standing challenges in constructing this five-membered nitrogen-containing heterocycle. By leveraging a transition metal palladium-catalyzed carbonylation tandem cyclization strategy, the invention provides a robust platform for generating diverse derivatives with high efficiency. For R&D directors and procurement specialists alike, this patent signals a shift towards more streamlined, cost-effective manufacturing processes that do not compromise on purity or structural complexity.
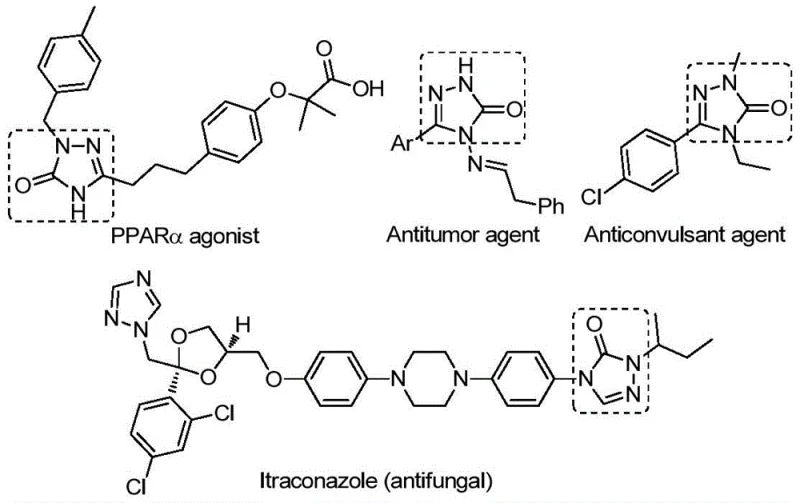
The strategic importance of this synthesis lies in its ability to access a wide chemical space. As illustrated in the provided data, the 1,2,4-triazole-3-one core is integral to various pharmacological classes, serving as a key building block for developing next-generation therapeutics. The versatility of this scaffold allows for extensive functionalization, making it a high-value target for contract development and manufacturing organizations (CDMOs) aiming to support drug discovery pipelines. The method described in CN112538054B not only simplifies the construction of the core ring but also ensures compatibility with a broad range of functional groups, thereby expanding the possibilities for medicinal chemistry optimization without the need for complex protecting group strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazole-3-one compounds has been fraught with significant operational and economic hurdles that hinder large-scale adoption. Traditional literature methods often rely on the cyclization of benzoyl hydrazides with urea under strongly basic conditions, or the tandem reaction of hydrazides with isocyanates. These conventional routes frequently suffer from harsh reaction conditions that require extreme temperatures or corrosive reagents, posing safety risks and increasing energy consumption. Furthermore, many existing protocols necessitate the pre-activation of substrates, adding extra synthetic steps that reduce overall atom economy and increase waste generation. The narrow substrate scope of these older methods is another critical bottleneck; they often fail to tolerate sensitive functional groups, limiting their utility in the synthesis of complex pharmaceutical intermediates. Additionally, low yields and difficult purification processes associated with these traditional routes result in higher production costs and longer lead times, which are unacceptable in the fast-paced environment of modern drug development.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in patent CN112538054B utilizes a palladium-catalyzed carbonylation tandem cyclization reaction that fundamentally reshapes the synthetic landscape for these compounds. This innovative strategy employs readily available chlorohydrazones and sodium azide as starting materials, bypassing the need for pre-activated or unstable precursors. A key feature of this methodology is the use of TFBen (phenyl 1,3,5-trimethylbenzene-2,4,6-tricarboxylate) as a solid carbon monoxide surrogate. This eliminates the logistical nightmares and safety hazards associated with handling toxic CO gas cylinders, making the process inherently safer and more suitable for standard laboratory and plant infrastructure. The reaction proceeds under relatively mild thermal conditions, typically around 100°C, in common aprotic solvents like 1,4-dioxane. This shift not only improves the safety profile but also enhances the reaction efficiency, allowing for the rapid assembly of the triazole ring with excellent yields across a diverse array of substrates.
Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams looking to optimize the process for specific applications. The reaction initiates with the oxidative addition of the palladium(0) catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, forming a reactive divalent palladium intermediate. This step is facilitated by the bulky and electron-rich Xantphos ligand, which stabilizes the metal center and promotes the subsequent insertion steps. Upon heating, the solid CO source, TFBen, decomposes to release carbon monoxide in situ. This generated CO then inserts into the carbon-palladium bond to form an acyl-palladium species, a critical intermediate that sets the stage for ring closure. The presence of sodium azide is pivotal; it reacts with the acyl-palladium complex to generate an acyl azide intermediate. This unstable species rapidly undergoes a Curtius rearrangement, losing nitrogen gas to form an isocyanate intermediate. Finally, an intramolecular nucleophilic attack by the hydrazine nitrogen onto the isocyanate carbon triggers the cyclization, yielding the final 1,2,4-triazole-3-one product and regenerating the active palladium catalyst.

Beyond the primary catalytic cycle, the control of impurities is a major advantage of this mechanistic pathway. The use of a solid CO source ensures a steady, controlled release of carbon monoxide, preventing the formation of side products that often arise from uncontrolled gas pressure or concentration spikes in traditional carbonylations. Furthermore, the specific choice of the Pd2(dba)3/Xantphos catalyst system minimizes homocoupling side reactions that are common in palladium chemistry. The reaction conditions are tuned to favor the desired intramolecular cyclization over intermolecular polymerization or decomposition of the azide. This high level of chemoselectivity translates directly to a cleaner crude reaction profile, reducing the burden on downstream purification processes. For quality control teams, this means a more consistent impurity profile and easier validation of the manufacturing process, which is essential for meeting stringent regulatory standards in pharmaceutical production.
How to Synthesize 1,2,4-Triazole-3-one Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory scale-up and industrial manufacturing. The protocol involves a straightforward one-pot procedure where all reagents are combined in a standard reaction vessel. The simplicity of the setup reduces the requirement for specialized equipment, lowering the barrier to entry for production facilities. Detailed standardized synthesis steps are provided below to guide process chemists in replicating these high-yielding results.
- Combine palladium catalyst (Pd2(dba)3), ligand (Xantphos), solid CO source (TFBen), chlorohydrazone substrate, and sodium azide in an aprotic organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to a temperature range of 100°C to 120°C and maintain stirring for a duration of 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, perform silica gel treatment, and purify the crude product via column chromatography to isolate the target 1,2,4-triazole-3-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The shift from hazardous gaseous reagents to stable solid surrogates drastically simplifies logistics and storage requirements. Sourcing chlorohydrazones and sodium azide is straightforward, as these are commodity chemicals available from multiple global suppliers, ensuring supply chain resilience and reducing the risk of single-source bottlenecks. The elimination of high-pressure gas handling equipment also reduces capital expenditure (CAPEX) for plant upgrades, allowing existing infrastructure to be utilized for this new chemistry. This flexibility enables faster time-to-market for new drug candidates relying on this scaffold, providing a competitive edge in the pharmaceutical marketplace.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the simplification of the raw material supply chain and the reduction of processing steps. By utilizing cheap and easily accessible starting materials like chlorohydrazones and avoiding the need for expensive, pre-activated substrates, the direct material costs are significantly lowered. Furthermore, the use of a solid CO source eliminates the need for specialized gas delivery systems and the associated safety monitoring infrastructure, leading to substantial operational cost savings. The high reaction efficiency and yield reported in the patent minimize waste generation and maximize the output per batch, which directly improves the overall cost of goods sold (COGS). The simplified workup procedure, involving basic filtration and chromatography, further reduces labor and solvent consumption costs compared to multi-step traditional syntheses.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness and generality of this synthetic method. Since the reaction tolerates a wide range of functional groups and substituents, manufacturers can produce a diverse library of intermediates using a unified platform technology. This standardization reduces the need for maintaining separate inventory lines for different synthetic routes. The reliance on commercially available catalysts and ligands, which are standard in the fine chemical industry, ensures that production is not held hostage by the scarcity of exotic reagents. Additionally, the mild reaction conditions reduce the risk of batch failures due to thermal runaways or equipment malfunctions, ensuring consistent delivery schedules to downstream clients. This reliability is critical for maintaining just-in-time manufacturing models in the pharmaceutical sector.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this methodology aligns perfectly with modern green chemistry principles. The avoidance of toxic carbon monoxide gas significantly reduces the environmental footprint and workplace exposure risks, simplifying compliance with increasingly stringent health and safety regulations. The reaction uses common organic solvents that can be readily recovered and recycled, minimizing hazardous waste disposal costs. The scalability of the process is evidenced by its successful demonstration on millimole scales with high conversion rates, indicating a clear path to ton-scale production. The robust nature of the catalytic system suggests that it can withstand the rigors of large-scale reactors without significant loss of activity, facilitating a smooth transition from pilot plant to full commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this synthesis route for their own portfolios.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis routes?
A: Unlike traditional methods that require harsh conditions, pre-activated substrates, or toxic gases, this novel approach utilizes cheap chlorohydrazones and a solid CO surrogate (TFBen). It offers broader substrate compatibility, milder reaction conditions (100°C), and eliminates the need for handling gaseous carbon monoxide directly, significantly improving operational safety and efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates the method's scalability. The use of stable, commercially available reagents and standard organic solvents like 1,4-dioxane facilitates easy scale-up from milligram laboratory scales to multi-kilogram commercial production without requiring specialized high-pressure equipment typically associated with carbonylation reactions.
Q: What is the role of the ligand and catalyst system in this reaction?
A: The system employs Tris(dibenzylideneacetone)dipalladium (Pd2(dba)3) as the catalyst precursor and Xantphos as the bidentate phosphine ligand. This specific combination is critical for facilitating the oxidative addition into the carbon-chlorine bond of the hydrazone and stabilizing the subsequent acyl-palladium intermediates, ensuring high turnover and selectivity for the triazole ring formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Pd-catalyzed carbonylation technology described in CN112538054B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory method into a robust commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 1,2,4-triazole-3-one intermediate meets the highest international standards, ready for immediate integration into your drug development pipeline.
We invite you to collaborate with us to leverage this advanced synthesis capability for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you optimize your supply chain with this cutting-edge chemistry.
