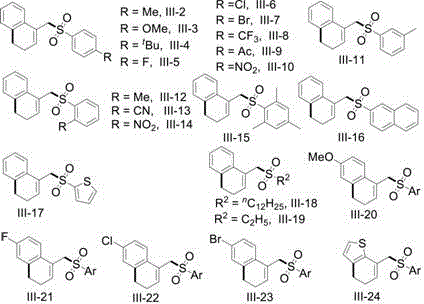
Advanced Visible Light Mediated Synthesis of 1-Sulfonylmethyl-3,4-Dihydronaphthalene Derivatives for Commercial Scale Production
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for constructing complex molecular scaffolds. Patent CN110540516B introduces a groundbreaking preparation method for 1-sulfonylmethyl-substituted 3,4-dihydronaphthalene compounds, utilizing a visible light mediated strategy that fundamentally shifts the paradigm from traditional thermal activation to photochemical precision. This innovation addresses critical pain points in organic synthesis by enabling the direct reaction of vinylcyclopropane compounds with sulfonyl chlorides under remarkably mild conditions. The significance of this technology lies not only in its ability to construct valuable dihydronaphthalene structural units, which are pervasive in bioactive molecules, but also in its operational simplicity and high efficiency. By leveraging visible light as a clean energy source, this method eliminates the need for hazardous oxidants and extreme thermal inputs, thereby aligning perfectly with modern green chemistry principles while ensuring high target product yields. For R&D directors and process chemists, this represents a viable pathway to access novel chemical space with reduced experimental risk.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of sulfonyl-containing dihydronaphthalene frameworks has relied heavily on conventional thermal methodologies that often suffer from significant drawbacks regarding safety, selectivity, and environmental impact. Traditional routes frequently necessitate the use of strong oxidants, harsh acidic or basic conditions, and elevated temperatures that can lead to the decomposition of sensitive functional groups and the formation of complex impurity profiles. These aggressive conditions not only compromise the overall yield of the desired intermediate but also impose severe constraints on the scope of substrates that can be tolerated, limiting the structural diversity accessible to medicinal chemists. Furthermore, the reliance on stoichiometric oxidants generates substantial amounts of chemical waste, creating burdensome downstream processing requirements and increasing the overall cost of goods sold. From a supply chain perspective, the instability of reagents under these harsh conditions can lead to batch-to-batch variability, posing risks to production continuity and quality control standards required by regulatory bodies.
The Novel Approach
In stark contrast, the novel approach disclosed in CN110540516B utilizes a visible light mediated C-C sigma-bond sulfonylation and arylation reaction that circumvents the limitations of thermal activation. By employing a photocatalytic system, the reaction proceeds through a radical mechanism that is initiated by low-energy visible light, allowing for the gentle activation of sulfonyl chlorides and vinylcyclopropanes without degrading sensitive moieties. This methodology offers exceptional substrate adaptability, accommodating a wide range of electronic and steric environments on both reaction partners, which is crucial for the late-stage functionalization of complex drug candidates. The mildness of the reaction conditions ensures that the integrity of the molecular scaffold is preserved, leading to cleaner reaction profiles and simplified purification processes. Additionally, the use of readily available and inexpensive sulfonyl chlorides as sulfur sources enhances the economic feasibility of the process, making it an attractive option for large-scale manufacturing where cost efficiency is paramount.

Mechanistic Insights into Visible Light Mediated C-C Sigma-Bond Sulfonylation
The core of this technological breakthrough lies in the intricate photocatalytic cycle that facilitates the cleavage of the strained C-C bond in the vinylcyclopropane and the subsequent formation of new C-S and C-C bonds. Under visible light irradiation, the photocatalyst, typically a ruthenium or iridium complex, absorbs photons to reach an excited state capable of engaging in single-electron transfer (SET) processes. This excited species interacts with the sulfonyl chloride to generate a sulfonyl radical, a highly reactive intermediate that initiates the cascade by attacking the electron-rich double bond of the vinylcyclopropane. The resulting radical intermediate undergoes a rapid ring-opening event driven by the relief of ring strain, generating a distal carbon-centered radical that is perfectly positioned for intramolecular cyclization onto the aromatic ring. This sequence of radical addition, ring opening, and cyclization is meticulously controlled by the reaction conditions, ensuring high regioselectivity and stereoselectivity in the formation of the dihydronaphthalene core.
Impurity control is inherently built into this mechanistic pathway due to the specificity of the radical interactions and the mild nature of the propagation steps. Unlike thermal radical reactions that often suffer from non-selective hydrogen abstraction or polymerization side reactions, the photocatalytic cycle maintains a steady concentration of active radicals, minimizing bimolecular termination events that lead to byproducts. The choice of base, such as 2,6-lutidine, plays a critical role in scavenging the hydrochloric acid byproduct generated during the sulfonyl radical formation, preventing acid-catalyzed degradation of the product or starting materials. Furthermore, the inert atmosphere protects the radical intermediates from quenching by oxygen, ensuring that the reaction proceeds efficiently to completion. This high level of mechanistic control translates directly to a cleaner crude reaction mixture, reducing the burden on downstream purification and enhancing the overall process mass intensity, a key metric for sustainable manufacturing.
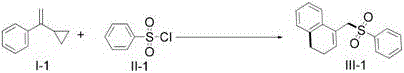
How to Synthesize 1-Sulfonylmethyl-3,4-Dihydronaphthalene Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the optimization of reaction parameters to maximize yield and reproducibility. The standard protocol involves the sequential addition of the vinylcyclopropane substrate, sulfonyl chloride coupling partner, photocatalyst, base, and organic solvent into a sealed reactor equipped for inert gas handling. The reaction mixture is then subjected to visible light irradiation, typically using blue LEDs, while being heated moderately to accelerate the kinetics without compromising the stability of the photocatalyst. Detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios, solvent choices, and workup procedures necessary to achieve the high yields reported in the patent data. Adhering to these optimized conditions ensures that the full potential of this visible light mediated transformation is realized, providing a reliable route for the production of high-purity intermediates.
- Charge a Schlenk tube with vinylcyclopropane compound, sulfonyl chloride, Ru(bpy)3Cl2 photocatalyst, 2,6-lutidine base, and 1,4-dioxane solvent under inert atmosphere.
- Place the reactor in an oil bath at 100°C and irradiate with a 5W blue LED lamp while stirring for 24 hours to ensure complete conversion.
- Filter the mixture through silica gel, wash with ethyl acetate and brine, dry over Na2SO4, and purify via column chromatography to isolate the target sulfone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this visible light mediated synthesis offers compelling strategic advantages that extend beyond mere technical novelty. The ability to utilize cheap and abundant sulfonyl chlorides as building blocks significantly reduces the raw material costs associated with producing these complex sulfone intermediates. Moreover, the mild reaction conditions eliminate the need for specialized high-pressure or high-temperature equipment, allowing for the utilization of standard glass-lined reactors or even flow chemistry setups, which lowers capital expenditure and simplifies facility requirements. The robustness of the reaction across a wide range of substrates means that a single manufacturing line can be adapted to produce a diverse portfolio of derivatives, enhancing asset utilization and flexibility in response to market demands. This versatility is crucial for maintaining supply continuity in the face of fluctuating demand for specific pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts often required in cross-coupling reactions, replaced by more economical photocatalysts that can be used in low loadings, drives down the direct material costs. Furthermore, the simplified workup procedure, which avoids complex extraction or neutralization steps associated with harsh reagents, reduces solvent consumption and waste disposal fees. The high yield and selectivity of the process minimize the loss of valuable starting materials, ensuring that the overall cost per kilogram of the final API intermediate is significantly optimized compared to legacy synthetic routes.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials, such as vinylcyclopropanes and sulfonyl chlorides, mitigates the risk of supply disruptions caused by scarce or custom-synthesized reagents. The mild conditions also reduce the safety hazards associated with exothermic runaway reactions, leading to fewer unplanned shutdowns and a more predictable production schedule. This reliability allows supply chain planners to maintain leaner inventory levels while confidently meeting delivery commitments to downstream pharmaceutical customers, thereby improving working capital efficiency.
- Scalability and Environmental Compliance: The photochemical nature of this reaction is inherently scalable, particularly with the advent of modern flow photoreactors that provide uniform light penetration and efficient heat transfer. This scalability ensures that the process can be seamlessly transferred from gram-scale R&D to multi-ton commercial production without significant re-optimization. Additionally, the reduced generation of hazardous waste and the use of greener reaction conditions align with increasingly stringent environmental regulations, reducing the regulatory burden and enhancing the corporate sustainability profile of the manufacturing operation.
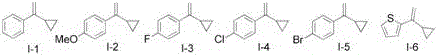
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light mediated synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this method for industrial applications. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing workflows.
Q: What are the primary advantages of this visible light mediated method over traditional thermal synthesis?
A: The visible light mediated approach operates under significantly milder conditions compared to traditional thermal methods, eliminating the need for extreme temperatures or harsh oxidants. This results in higher target product yields, broader substrate adaptability, and improved safety profiles for large-scale manufacturing operations.
Q: Is the photocatalyst used in this process recyclable and cost-effective?
A: Yes, the patent highlights the use of robust photocatalysts like Ru(bpy)3Cl2 which offer high efficiency. The methodology is designed to minimize catalyst loading while maintaining high conversion rates, contributing to substantial cost savings in raw material consumption for commercial production.
Q: Can this synthetic route accommodate diverse functional groups on the aromatic rings?
A: Absolutely. The method demonstrates wide substrate applicability, tolerating various substituents such as halogens, alkyl groups, alkoxy groups, and nitro groups on both the vinylcyclopropane and sulfonyl chloride components, making it highly versatile for synthesizing complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Sulfonylmethyl-3,4-Dihydronaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic technologies in the synthesis of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN110540516B can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with specialized photoreactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We are committed to delivering consistent quality and supply security for complex molecules, leveraging our deep technical expertise to overcome scale-up challenges associated with photochemistry.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full capabilities of this synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient method can lower your overall production costs. Please contact us to request specific COA data for related intermediates and comprehensive route feasibility assessments, allowing you to make informed decisions about integrating this cutting-edge technology into your supply chain.