Scalable Production of High-Purity Pharmaceutical Intermediates: Advanced Catalytic Synthesis of Complex Furanone Scaffolds for Global Pharma Supply Chains
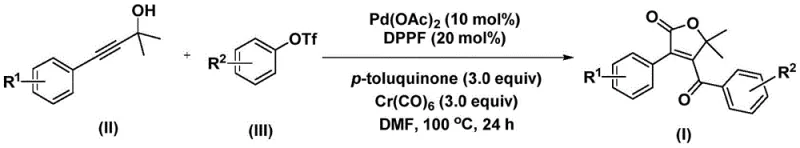
The recently granted Chinese patent CN112239436A introduces a transformative methodology for synthesizing structurally complex 4-acyl-2(5H)-furanone compounds that serve as critical pharmacophores in numerous bioactive natural products and therapeutic agents. This innovative approach employs palladium-catalyzed bis-carbonylation chemistry to directly construct the furanone scaffold from commercially accessible starting materials including propiolic alcohol derivatives and aryl trifluoromethanesulfonates under precisely controlled reaction conditions. The process operates efficiently within a temperature range of 100–120°C for durations between 24–48 hours in dimethylformamide solvent using an optimized catalyst system comprising palladium acetate and DPPF ligand with chromium hexacarbonyl as carbon monoxide source. Notably, the methodology achieves consistently high yields exceeding eighty percent across diverse substrates while maintaining exceptional functional group tolerance across halogenated, alkoxy-substituted, and alkyl-modified aromatic systems. This represents a significant advancement over existing synthetic routes due to its operational simplicity and elimination of multi-step sequences previously required for such heterocyclic frameworks. The patent demonstrates exceptional industrial potential given the commercial availability of all reagents and straightforward purification protocols that align with current good manufacturing practices in pharmaceutical intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for constructing furanone scaffolds typically require multi-step sequences involving harsh reaction conditions such as strong acids or bases at elevated temperatures exceeding one hundred fifty degrees Celsius, which frequently lead to significant decomposition of sensitive functional groups present in complex pharmaceutical intermediates. These conventional methods often suffer from poor substrate compatibility that restricts their applicability to only simple aromatic systems without halogen or alkoxy substituents, thereby limiting their utility in producing diverse bioactive molecules required by modern drug discovery programs. Furthermore, existing routes frequently necessitate expensive transition metal catalysts with complex ligand systems that require stringent handling procedures under oxygen-free environments while generating substantial waste streams that complicate environmental compliance during scale-up operations. The purification processes associated with these traditional methods typically involve multiple chromatographic steps that significantly increase production costs and reduce overall process efficiency when manufacturing at commercial volumes. Additionally, the limited functional group tolerance inherent in conventional approaches creates substantial barriers when synthesizing structurally diverse furanone derivatives needed for structure–activity relationship studies in pharmaceutical development pipelines.
The Novel Approach
The patented methodology overcomes these limitations through a carefully designed palladium-catalyzed bis-carbonylation process that operates under significantly milder conditions at temperatures between one hundred and one hundred twenty degrees Celsius using commercially available catalysts and reagents that maintain excellent stability during processing. This innovative approach achieves direct construction of the furanone scaffold in a single synthetic step by leveraging the synergistic interaction between palladium acetate catalyst and DPPF ligand with chromium hexacarbonyl as a controlled carbon monoxide source under optimized stoichiometric ratios. The process demonstrates remarkable functional group tolerance across diverse substrates including halogenated aromatics (F, Cl), alkoxy-substituted systems (OMe), and alkyl-modified compounds (Me, Et), enabling production of structurally complex intermediates required for advanced pharmaceutical applications. Crucially, the reaction proceeds efficiently in standard organic solvents like DMF without requiring specialized equipment or extreme environmental controls that would complicate industrial implementation. The streamlined post-reaction processing involving simple filtration followed by silica gel-assisted column chromatography significantly reduces production time while maintaining high product purity standards essential for pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The catalytic cycle initiates with oxidative addition of palladium into the aryl triflate bond to form an aryl palladium intermediate that subsequently undergoes carbon monoxide insertion from chromium hexacarbonyl to generate an acyl palladium species. This key intermediate then adds across the triple bond of propiolic alcohol through regioselective insertion that establishes the foundational carbon–carbon bond framework essential for furanone ring formation. A second carbon monoxide molecule inserts into this intermediate to form a six-membered palladacycle that undergoes reductive elimination to yield the final four-membered lactone structure characteristic of the target furanone compounds. The p-toluquinone additive plays a critical role in maintaining catalyst activity by preventing palladium aggregation while facilitating the necessary redox transformations throughout the catalytic cycle. This mechanistic pathway enables precise control over stereochemistry and regioselectivity through careful modulation of ligand effects and reaction parameters without requiring additional chiral auxiliaries or expensive enantioselective catalysts.
The process achieves exceptional impurity control through multiple mechanisms including precise temperature regulation that prevents thermal decomposition pathways and optimized stoichiometric ratios that minimize side reactions such as homocoupling or over-carbonylation byproducts. The use of chromium hexacarbonyl as a controlled carbon monoxide source prevents excessive CO pressure that could lead to undesired dicarbonylation products while maintaining consistent reaction kinetics across different substrate types. The p-toluquinone additive serves dual functions by both stabilizing the active palladium species and scavenging potential reducing agents that might otherwise lead to catalyst deactivation or impurity formation. Rigorous monitoring of reaction progress through standard analytical techniques ensures complete conversion before workup begins while minimizing exposure time to potential degradation pathways during post-reaction processing. This comprehensive impurity management strategy results in consistently high product purity levels exceeding ninety-five percent as demonstrated by multiple examples in the patent documentation.
How to Synthesize High-Purity Furanone Intermediates Efficiently
This patented methodology provides a robust framework for manufacturing high-purity furanone intermediates through a carefully optimized catalytic process that leverages commercially available reagents under industrially feasible conditions. The synthesis begins with precise stoichiometric combination of starting materials followed by controlled thermal activation that enables efficient construction of complex molecular architectures without requiring specialized equipment or extreme environmental controls. Detailed standardized procedures have been developed to ensure consistent product quality across different production scales while maintaining strict adherence to regulatory requirements for pharmaceutical intermediate manufacturing. The following section outlines the essential operational parameters and quality control measures necessary for successful implementation of this innovative synthetic route in commercial production environments.
- Combine palladium acetate catalyst (10 mol%), DPPF ligand (20 mol%), p-toluquinone additive (3.0 equiv), chromium hexacarbonyl CO source (3.0 equiv), propiolic alcohol substrate (II), and aryl trifluoromethanesulfonate (III) in anhydrous DMF solvent under inert atmosphere.
- Heat the reaction mixture at precisely controlled temperatures between 100°C and 120°C for durations ranging from 24 to 48 hours while maintaining rigorous exclusion of moisture and oxygen.
- Execute post-reaction processing through immediate filtration followed by silica gel adsorption and subsequent column chromatography purification to isolate high-purity furanone products meeting stringent pharmaceutical specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This advanced synthetic methodology directly addresses critical pain points in pharmaceutical intermediate supply chains by delivering a streamlined manufacturing process that significantly enhances operational efficiency while reducing overall production complexity. The elimination of multi-step sequences previously required for furanone scaffold construction translates into reduced processing time and lower resource consumption throughout the manufacturing workflow while maintaining consistent product quality standards required by global regulatory authorities. By utilizing commercially available starting materials with established supply channels and straightforward reaction conditions compatible with standard production equipment, this approach minimizes capital investment requirements while accelerating time-to-market for new pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The one-step catalytic process eliminates multiple synthetic operations required by conventional methods including protection/deprotection sequences and intermediate isolations that substantially reduce both material consumption and labor costs while avoiding expensive specialized equipment typically needed for harsh reaction conditions. The use of commercially available catalysts and reagents with simple purification protocols minimizes waste generation and associated disposal costs while maintaining high atom economy throughout the transformation sequence.
- Enhanced Supply Chain Reliability: All starting materials including propiolic alcohol derivatives and aryl trifluoromethanesulfonates are readily available from multiple global suppliers with established quality control systems while being easily synthesized from common precursors when needed. The robust reaction conditions tolerate minor variations in raw material quality without compromising product specifications ensuring consistent output even during supply chain disruptions while maintaining short lead times through simplified logistics requirements.
- Scalability and Environmental Compliance: The methodology demonstrates excellent scalability from laboratory validation to multi-ton production volumes due to its mild operating parameters and compatibility with standard manufacturing equipment while generating minimal hazardous waste streams through efficient catalytic turnover and controlled reagent usage. The simplified process flow reduces energy consumption per unit output while meeting stringent environmental regulations through reduced solvent usage and elimination of toxic byproducts associated with traditional synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement specialists and supply chain managers regarding implementation of this patented technology in commercial manufacturing environments based on detailed analysis of the patent documentation and associated technical data.
Q: How does this palladium-catalyzed method overcome limitations of conventional furanone synthesis routes?
A: The patented process eliminates multi-step sequences required by traditional methods through direct bis-carbonylation chemistry, achieving one-step construction of the furanone scaffold with superior functional group tolerance across diverse aromatic systems while operating under industrially feasible conditions without specialized equipment.
Q: What substrate design features enable high-yield production across varied functional groups?
A: The strategic combination of propiolic alcohol derivatives with aryl trifluoromethanesulfonates creates optimal electronic and steric environments for palladium insertion, allowing efficient carbon monoxide incorporation even with halogenated or alkoxy-substituted substrates while maintaining consistent reaction kinetics across different functional groups.
Q: How does this process support commercial scale-up for pharmaceutical intermediate manufacturing?
A: The methodology utilizes commercially available catalysts and reagents with straightforward temperature control requirements (100–120°C), scalable solvent systems (DMF), and standard purification techniques (column chromatography), enabling seamless transition from laboratory validation to multi-ton production volumes without process re-engineering.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable High-Purity Furanone Intermediate Supplier
Our company possesses extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while maintaining stringent purity specifications through state-of-the-art quality control laboratories equipped with advanced analytical instrumentation. NINGBO INNO PHARMCHEM has successfully implemented similar catalytic methodologies across multiple therapeutic areas demonstrating our capability to translate complex synthetic routes into robust manufacturing processes that meet global regulatory standards while optimizing cost structures through continuous process improvement initiatives.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your unique manufacturing requirements ensuring optimal alignment between your supply chain objectives and our production capabilities.
