Advanced Rhodium-Catalyzed Cyclization for High-Purity Isoquinolinone and Isocoumarin Intermediates
Advanced Rhodium-Catalyzed Cyclization for High-Purity Isoquinolinone and Isocoumarin Intermediates
The landscape of organic synthesis for bioactive heterocycles is undergoing a significant transformation, driven by the need for more atom-economical and operationally simple processes. Patent CN110746353B introduces a groundbreaking methodology for the construction of isoquinolinone and isocoumarin scaffolds, which are ubiquitous motifs in pharmaceuticals and natural products. This technology leverages a synergistic rhodium-catalyzed system to achieve direct cyclization between benzoic acid derivatives and N-vinylamides. For R&D directors and process chemists, this represents a pivotal shift away from multi-step sequences involving transient directing groups. The ability to access these complex cores in a single operational step not only accelerates lead optimization but also lays a robust foundation for commercial manufacturing, addressing critical pain points regarding yield consistency and impurity profiles in the production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoquinolinone derivatives has relied heavily on transition metal-catalyzed C-H activation strategies that necessitate the installation of a specific directing group on the benzamide substrate. This conventional approach typically involves converting benzoic acid to benzamide, introducing a coordinating group to guide the metal catalyst, performing the cyclization, and finally executing an additional synthetic step to remove the directing group. This multi-step sequence inherently suffers from low overall atom economy and increased waste generation. Furthermore, the requirement for specific directing groups limits the substrate scope, often excluding sensitive functional groups that might be incompatible with the installation or removal conditions. From a supply chain perspective, these extra steps translate to longer lead times, higher consumption of reagents and solvents, and increased complexity in purification, all of which drive up the cost of goods sold (COGS) for the final active pharmaceutical ingredient (API) intermediate.
The Novel Approach
In stark contrast, the methodology disclosed in CN110746353B utilizes a direct oxidative annulation strategy that bypasses the need for pre-functionalized benzamides. By employing readily available benzoic acids and N-vinylformamides or N-vinylacetamides, the reaction constructs the heterocyclic core in a single pot. The process operates under relatively mild conditions, utilizing a rhodium catalyst in conjunction with a metal oxidant and a base. This streamlined approach eliminates the cumbersome directing group installation and removal steps, thereby drastically reducing the number of unit operations required. The versatility of this method is evident in its tolerance for a broad spectrum of substituents on the aromatic ring, allowing for the rapid generation of diverse chemical libraries. This operational simplicity is a game-changer for process development, enabling faster scale-up and more reliable supply chains for complex heterocyclic building blocks.
Mechanistic Insights into Rhodium-Catalyzed Oxidative Annulation
The core of this technological advancement lies in the sophisticated interplay between the rhodium catalyst, the metal oxidant, and the base. The reaction initiates with the coordination of the rhodium(III) species to the carboxylate oxygen of the benzoic acid substrate, facilitating a concerted metalation-deprotonation (CMD) process that activates the ortho C-H bond. This generates a stable rhodacycle intermediate, which is the key determinant of regioselectivity. Subsequently, the N-vinylamide inserts into the Rh-C bond, followed by migratory insertion and reductive elimination to form the new C-C and C-N bonds simultaneously. The presence of the metal oxidant, such as silver or copper salts, is crucial for regenerating the active Rh(III) species from the reduced Rh(I) state, thus closing the catalytic cycle. This redox-neutral or oxidative manifold ensures high turnover numbers and sustained catalytic activity throughout the reaction duration, which is essential for maintaining consistent quality in large-scale batches.
From an impurity control perspective, the mechanism offers distinct advantages over radical-based or harsh acidic cyclizations. The well-defined coordination sphere of the rhodium catalyst minimizes non-specific side reactions, leading to cleaner crude reaction profiles. The use of N-vinylamides as coupling partners is particularly strategic; the vinyl group acts as an internal oxidant and carbon source, streamlining the stoichiometry. Moreover, the choice of solvent, such as acetonitrile or benzonitrile, plays a vital role in stabilizing the cationic rhodium intermediates and ensuring homogeneous reaction conditions. Understanding these mechanistic nuances allows process chemists to fine-tune parameters like temperature (40°C to 100°C) and catalyst loading (2.5 to 10 mol%) to optimize yield and minimize trace metal residues, ensuring the final product meets stringent pharmaceutical purity specifications without requiring excessive downstream processing.
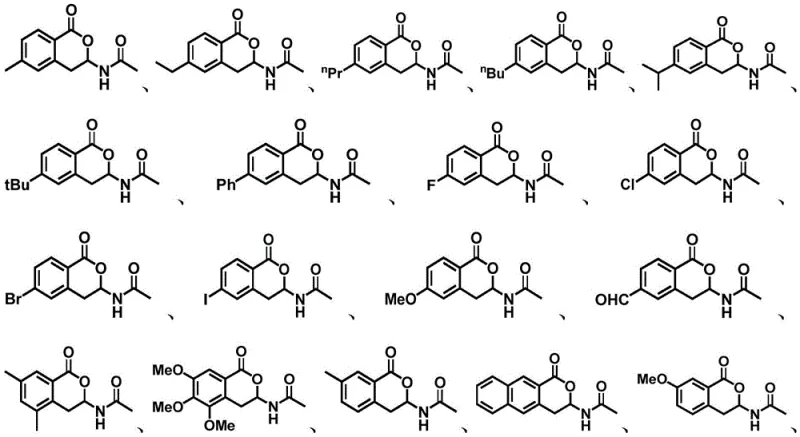
How to Synthesize Isoquinolinone and Isocoumarin Derivatives Efficiently
Implementing this rhodium-catalyzed protocol requires careful attention to reagent quality and reaction parameters to ensure reproducibility and safety. The procedure generally involves charging a reaction vessel with the benzoic acid derivative, the N-vinylamide coupling partner, the rhodium catalyst precursor (such as [Cp*RhCl2]2), a metal oxidant like silver acetate, and a mild base. The mixture is then suspended in a polar aprotic solvent and heated under stirring. The robustness of the method allows it to be performed under air, eliminating the need for rigorous inert atmosphere techniques which can be a bottleneck in large-scale manufacturing. Post-reaction workup is straightforward, typically involving filtration to remove insoluble metal salts, followed by solvent evaporation and standard extraction protocols. The crude product can be purified via flash column chromatography or recrystallization, depending on the specific substitution pattern and desired purity level. This operational ease makes it highly attractive for both kilogram-scale pilot runs and multi-ton commercial production.
- Mix benzoic acid derivative, N-vinylformamide or N-vinylacetamide, rhodium catalyst (e.g., [Cp*RhCl2]2), metal oxidant, and base in a solvent like acetonitrile.
- Heat the reaction mixture to a temperature between 40°C and 100°C and stir for 18 to 36 hours under air or inert atmosphere.
- Filter the mixture, remove solvent, extract with ethyl acetate, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical elegance. The primary driver for cost reduction lies in the drastic simplification of the synthetic route. By collapsing a multi-step sequence into a single transformation, manufacturers can significantly reduce labor costs, energy consumption, and solvent usage. The elimination of the directing group installation and removal steps removes two entire unit operations from the production schedule, effectively shortening the manufacturing cycle time. Furthermore, the starting materials—substituted benzoic acids and N-vinylamides—are commodity chemicals that are commercially available in bulk quantities at competitive prices. This reliance on abundant feedstocks mitigates supply risk and protects against price volatility associated with specialized, custom-synthesized intermediates. The overall process efficiency translates directly into a lower cost per kilogram for the final isoquinolinone or isocoumarin intermediate, enhancing the margin potential for downstream API production.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the reduction in step count and reagent consumption. Traditional routes often require expensive coupling reagents and protecting group chemistry, which generate substantial waste and increase raw material costs. In contrast, this rhodium-catalyzed method utilizes catalytic amounts of metal and stoichiometric oxidants that are relatively inexpensive. The high atom economy means that a larger proportion of the input mass ends up in the final product, reducing waste disposal costs. Additionally, the simplified workflow reduces the requirement for specialized equipment and extensive operator time, leading to substantial operational expenditure (OPEX) savings. These factors combine to create a highly cost-effective manufacturing process that remains competitive even when scaling to industrial volumes.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of generic, widely available starting materials. Unlike proprietary intermediates that may be sourced from a single supplier, benzoic acid derivatives are produced by numerous chemical manufacturers globally. This diversity in the supply base ensures continuity of supply and provides leverage in negotiations. Moreover, the robustness of the reaction conditions—tolerating air and moisture to some extent—reduces the risk of batch failures due to environmental factors. The ability to produce high-quality intermediates with consistent purity profiles minimizes the need for rework or rejection, ensuring that delivery schedules are met reliably. This predictability is crucial for pharmaceutical companies managing tight production timelines for clinical trials and commercial launches.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces challenges related to heat transfer and mixing, but this exothermic cyclization is manageable within standard reactor configurations. The use of common solvents like acetonitrile facilitates solvent recovery and recycling, aligning with green chemistry principles. The reduction in waste generation, particularly the avoidance of heavy metal waste from stoichiometric reagents used in older methods, simplifies environmental compliance and waste treatment. The process is designed to be scalable from gram to ton quantities without fundamental changes to the chemistry, allowing for seamless technology transfer from R&D to production facilities. This scalability ensures that the supply chain can respond flexibly to market demand fluctuations without compromising on quality or regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed synthesis platform. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear picture of the technology's capabilities and limitations. Understanding these details helps stakeholders make informed decisions about integrating this methodology into their existing manufacturing portfolios. The answers reflect the consensus on best practices for handling rhodium catalysts and optimizing reaction outcomes for diverse substrate classes.
Q: What is the primary advantage of this rhodium-catalyzed method over traditional synthesis?
A: Unlike traditional methods that require pre-functionalization of benzamide with a directing group and a subsequent removal step, this patent describes a direct one-step cyclization using commercially available benzoic acids and N-vinylamides, significantly simplifying the workflow.
Q: What types of substituents are tolerated on the benzoic acid substrate?
A: The process demonstrates high substrate flexibility, tolerating a wide range of substituents including halogens (F, Cl, Br, I), alkyl groups (methyl, ethyl, tert-butyl), alkoxy groups, and even bulky aromatic systems like naphthyl groups.
Q: Is the rhodium catalyst loading economically viable for scale-up?
A: Yes, the patent specifies catalyst loadings between 2.5 mol% and 10 mol%, which is considered efficient for precious metal catalysis in fine chemical synthesis, especially given the high yields and operational simplicity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN110746353B for the production of high-value heterocyclic intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. We are committed to delivering high-purity isoquinolinone and isocoumarin derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex organometallic catalysis allows us to offer cost-effective solutions that do not compromise on quality or regulatory compliance.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this efficient synthetic route for their pipeline projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this technology can optimize your supply chain. We encourage you to contact our technical procurement team to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs. Let us help you accelerate your development timeline and reduce your manufacturing costs with our proven expertise in fine chemical intermediates.
