Scalable Production of High-Purity Thiosulfonates via Green Aerobic Oxidation Technology
Scalable Production of High-Purity Thiosulfonates via Green Aerobic Oxidation Technology
The landscape of fine chemical synthesis is undergoing a paradigm shift towards greener, more atom-economical processes, a trend vividly exemplified by the technological breakthroughs detailed in Chinese Patent CN114213298A. This pivotal intellectual property introduces a highly efficient method for preparing thiosulfonate compounds through the direct oxidation of thiophenols, utilizing molecular oxygen from the air as the terminal oxidant. Unlike traditional methodologies that rely on stoichiometric amounts of hazardous reagents or expensive transition metals, this novel approach leverages N-chlorosuccinimide (NCS) as a reaction promoter under remarkably mild conditions. For R&D directors and process chemists seeking robust pathways for API intermediates, this technology represents a significant leap forward in operational simplicity and environmental compliance. The ability to conduct these transformations at room temperature with short reaction times not only enhances safety profiles but also drastically reduces energy consumption, aligning perfectly with modern sustainable manufacturing goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
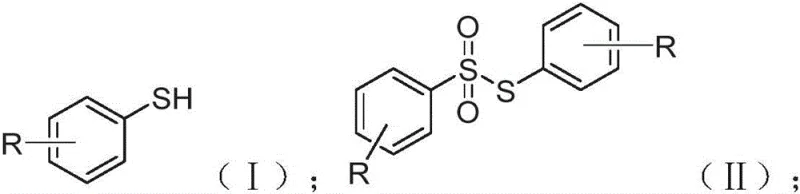
Historically, the synthesis of thiosulfonic acid esters and their derivatives has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Conventional routes often necessitate the use of benzene sulfonic acids or sodium benzene sulfinates coupled with thiophenols or disulfides in the presence of aggressive activating agents such as cyanuric chloride, iodine, or bromine. More critically, many established protocols depend heavily on transition metal catalysts, particularly copper complexes with specialized ligands, to drive the sulfonylation reaction. These metal-dependent systems introduce severe complications regarding product purity, as trace metal residues must be rigorously removed to meet pharmaceutical standards, adding costly purification steps like chelation or extensive chromatography. Furthermore, the use of halogenating reagents like bromine generates substantial hazardous waste, creating disposal challenges and increasing the overall environmental footprint of the manufacturing process. These factors collectively result in higher production costs, longer lead times, and reduced atom economy, making traditional methods less attractive for cost-sensitive commercial applications.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN114213298A offers a streamlined, metal-free alternative that fundamentally simplifies the synthetic workflow. By employing N-chlorosuccinimide (NCS) as a promoter and utilizing ambient air as the oxidant, this method bypasses the need for toxic metals and harsh halogenating agents entirely. The reaction proceeds efficiently in common solvents like acetonitrile at room temperature, typically reaching completion within 30 to 60 minutes. This drastic reduction in reaction severity not only preserves the integrity of sensitive functional groups on the aromatic ring but also minimizes the formation of complex byproduct mixtures. The operational simplicity is further enhanced by a straightforward workup procedure involving extraction and distillation, which facilitates the isolation of high-purity products with yields ranging from 60% to 90%. This approach effectively decouples high performance from high environmental cost, providing a viable pathway for the industrial production of thiosulfonates.
Mechanistic Insights into NCS-Promoted Aerobic Oxidation
The mechanistic elegance of this transformation lies in the synergistic interaction between the NCS promoter and molecular oxygen, which facilitates the direct conversion of the thiol group into the thiosulfonate moiety without external metal catalysis. The process likely initiates with the activation of the thiophenol by NCS, generating a reactive sulfur-chlorine intermediate or a sulfenyl chloride species in situ. This activated intermediate then undergoes oxidation by atmospheric oxygen, leading to the formation of the sulfonyl group while simultaneously coupling with another equivalent of the thiol or its derivative to form the S-S bond characteristic of thiosulfonates. The absence of transition metals eliminates the risk of metal-ligand complexation issues and ensures that the reaction trajectory is governed purely by organic redox potentials. This mechanism is highly tolerant of various substituents on the phenyl ring, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as nitro and halogens, demonstrating broad substrate scope. Such mechanistic robustness is crucial for R&D teams aiming to diversify their chemical libraries without needing to re-optimize catalytic systems for each new substrate.
From an impurity control perspective, this metal-free oxidation pathway offers distinct advantages in managing the杂质 profile of the final API intermediate. Traditional metal-catalyzed reactions often suffer from side reactions such as homocoupling to disulfides or over-oxidation to sulfonic acids, exacerbated by the presence of metal ions that can catalyze radical degradation pathways. In the NCS-promoted aerobic system, the reaction conditions are sufficiently mild to suppress these deleterious side reactions, resulting in a cleaner crude product profile. The use of air as the oxidant ensures that the only byproduct is essentially water or benign succinimide derivatives, which are easily removed during the aqueous workup. This inherent cleanliness reduces the burden on downstream purification units, allowing for simpler crystallization protocols rather than resource-intensive chromatographic separations. For quality assurance teams, this translates to more consistent batch-to-batch reproducibility and easier validation of the cleaning procedures in multi-purpose manufacturing facilities.
How to Synthesize Thiosulfonate Compounds Efficiently
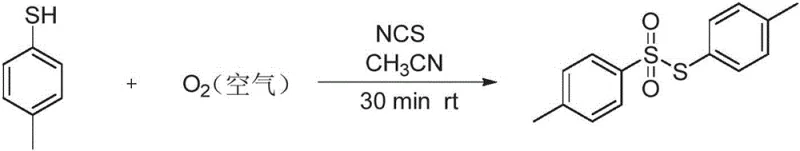
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and solvent choices to maximize efficiency and yield. The standard protocol involves dissolving the thiophenol substrate in acetonitrile, followed by the addition of NCS in a molar ratio ranging from 1:1.5 to 1:5, with 1:3 being optimal for many substrates. The mixture is then stirred under an open atmosphere or oxygen balloon at room temperature for approximately 30 minutes. Upon completion, the reaction mixture is subjected to a liquid-liquid extraction using ethyl acetate, washed to remove succinimide byproducts, and concentrated. The crude solid can often be purified simply by recrystallization from ethyl acetate, yielding the pure thiosulfonate ester as a white solid. This straightforward procedure minimizes operator exposure to hazardous chemicals and reduces the complexity of the equipment required, making it accessible for both small-scale discovery and larger production runs.
- Combine thiophenol substrate and N-chlorosuccinimide (NCS) promoter in acetonitrile solvent at a molar ratio of approximately 1: 3.
- Stir the reaction mixture under an atmosphere of air or oxygen at room temperature (15-60°C) for 30 to 60 minutes to allow direct oxidation.
- Quench the reaction, extract the organic phase with ethyl acetate, dry, and purify via recrystallization or column chromatography to obtain the pure thiosulfonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aerobic oxidation technology presents a compelling value proposition centered on cost reduction and supply security. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of commodities like copper or palladium. Furthermore, the reliance on air as the primary oxidant means that the process does not require the storage and handling of dangerous compressed gases or unstable peroxide reagents, thereby lowering insurance premiums and safety compliance costs. The simplified workup and purification steps reduce the consumption of solvents and silica gel, directly impacting the cost of goods sold (COGS). These cumulative efficiencies allow for a more competitive pricing structure for high-purity thiosulfonate intermediates, enabling downstream pharmaceutical manufacturers to optimize their own production budgets without compromising on quality standards.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts and expensive ligands from the synthetic route results in substantial raw material savings. Additionally, the mild reaction conditions eliminate the need for energy-intensive heating or cooling systems, further driving down utility costs. The simplified purification process reduces the consumption of chromatography media and solvents, leading to a leaner and more cost-effective manufacturing operation that maximizes margin potential for high-volume orders.
- Enhanced Supply Chain Reliability: By utilizing commodity chemicals like NCS and acetonitrile, which are widely available from multiple global suppliers, the risk of raw material shortages is significantly minimized. The robustness of the reaction against variations in substrate electronics ensures consistent yields across different batches, reducing the likelihood of production delays due to failed runs. This reliability allows supply chain planners to maintain lower safety stock levels while ensuring continuous availability of critical intermediates for time-sensitive drug development projects.
- Scalability and Environmental Compliance: The metal-free nature of this process simplifies waste stream management, as there are no heavy metal contaminants requiring specialized treatment before discharge. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, reducing the regulatory burden on manufacturing sites. The short reaction time and ambient temperature conditions make the process highly scalable from gram to ton quantities without the need for complex engineering controls, facilitating rapid capacity expansion to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiosulfonate synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing manufacturing workflows.
Q: What are the primary advantages of this aerobic oxidation method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive and toxic transition metal catalysts (like copper) and hazardous brominating reagents. It utilizes air as a green oxidant, significantly reducing environmental impact and downstream purification costs associated with heavy metal removal.
Q: What is the typical yield and purity profile for thiosulfonates produced via this protocol?
A: According to patent data, the process achieves yields ranging from 60% to 90% with high conversion rates. The mild reaction conditions (room temperature) minimize side reactions, facilitating the production of high-purity intermediates suitable for sensitive pharmaceutical applications.
Q: Is this synthesis method scalable for industrial manufacturing?
A: Yes, the protocol is explicitly designed for industrial popularization. It features short reaction times (15-60 min), simple operation (one-pot), and uses cheap, readily available reagents like NCS and acetonitrile, making it highly amenable to large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiosulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthetic routes in the modern pharmaceutical supply chain. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the NCS-promoted aerobic oxidation are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of thiosulfonate intermediate delivered meets the highest global standards. We are committed to leveraging our technical expertise to optimize these green chemistry protocols, delivering cost-effective solutions that support your drug development timelines.
We invite you to collaborate with us to explore how this advanced synthesis method can enhance your project's economics and sustainability profile. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
