Advanced Palladium-Catalyzed Carbonylation for High-Purity 2-Trifluoromethyl Imidazole Intermediates
Advanced Palladium-Catalyzed Carbonylation for High-Purity 2-Trifluoromethyl Imidazole Intermediates
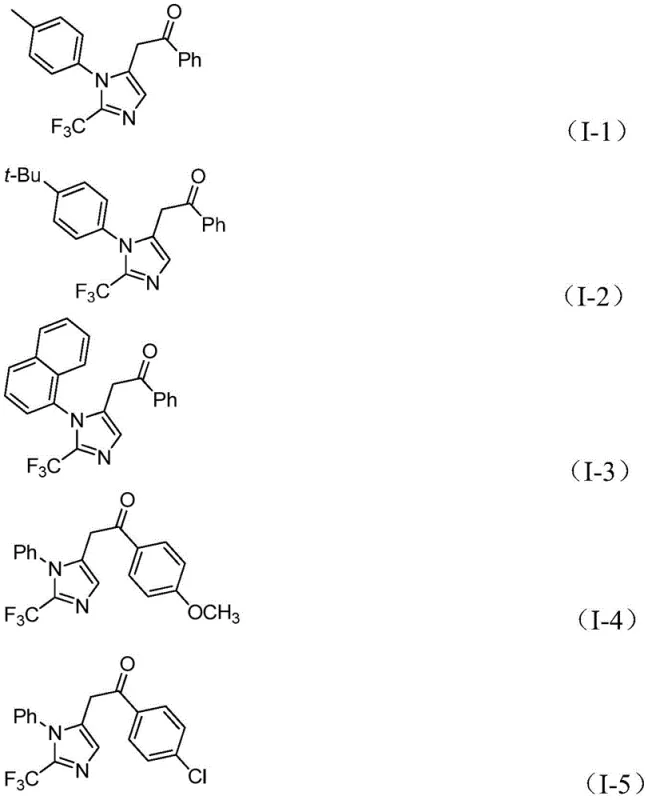
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are critical for enhancing metabolic stability and bioavailability. A significant breakthrough in this domain is documented in Chinese Patent CN111423381A, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted imidazole compounds. This technology represents a paradigm shift from traditional synthetic routes by employing a transition metal palladium-catalyzed carbonylation tandem reaction. The process utilizes readily available starting materials including trifluoroethylimide chloride, propargylamine, and diaryliodonium salts to construct the imidazole core under remarkably mild conditions. For R&D directors and procurement specialists, this patent offers a compelling value proposition: a streamlined pathway to high-value intermediates that avoids the safety hazards associated with diazo compounds while maintaining exceptional substrate tolerance. The ability to synthesize diversified 1,5-disubstituted imidazoles with a trifluoromethyl group at the 2-position opens new avenues for drug discovery and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on the use of specialized trifluoromethyl synthons reacting directly with suitable substrates. Literature precedents often cite the use of trifluorodiazoethane or similar hazardous reagents which pose significant safety risks due to their explosive nature and toxicity. Furthermore, conventional methods frequently require harsh reaction conditions, such as elevated temperatures or strong acidic/basic environments, which can lead to the decomposition of sensitive functional groups and result in complex impurity profiles. The reliance on unstable diazo compounds not only complicates the supply chain due to storage and transportation restrictions but also necessitates specialized equipment for safe handling, thereby inflating the overall cost of manufacturing. Additionally, many traditional routes suffer from limited substrate scope, failing to accommodate bulky or electron-deficient aryl groups, which restricts the chemical space available for medicinal chemists during lead optimization phases.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a palladium-catalyzed carbonylation strategy that operates at a mild temperature of 30°C. This method leverages trifluoroethylimide acid chloride and propargylamine as key building blocks, reacting them with diaryliodonium salts in the presence of a carbon monoxide surrogate generated in situ from formic acid and acetic anhydride. This innovation eliminates the need for handling gaseous carbon monoxide directly, significantly enhancing operational safety. The reaction mechanism involves a sophisticated tandem sequence where base-promoted intermolecular carbon-nitrogen bond formation is followed by palladium-catalyzed alkyne aminopalladation and subsequent carbonylation. This cascade process efficiently constructs the imidazole ring with high atom economy. The use of diaryliodonium salts as oxidants allows for the introduction of diverse aryl groups at the 5-position of the imidazole ring, providing unparalleled flexibility in molecular design.
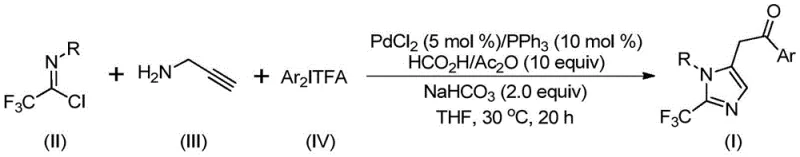
The versatility of this synthetic route is further evidenced by its compatibility with a wide array of functional groups. As demonstrated in the patent examples, substituents such as methyl, tert-butyl, chloro, bromo, trifluoromethyl, and nitro groups on the aromatic rings are well-tolerated, yielding the corresponding products in high yields ranging from 65% to 97%. This broad substrate scope is crucial for the rapid generation of compound libraries in drug discovery programs. Moreover, the reaction can be scaled up to the gram level without significant loss in efficiency, indicating strong potential for industrial application. The simplicity of the post-processing steps, involving filtration and standard column chromatography, further reduces the operational burden on production teams, making this method highly attractive for both laboratory-scale synthesis and commercial manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Reaction
A deep understanding of the reaction mechanism is essential for R&D teams aiming to optimize this process for specific applications. The proposed mechanism begins with the base-promoted formation of a trifluoroacetamidine intermediate through the reaction of trifluoroethylimide chloride with propargylamine. This intermediate subsequently undergoes isomerization to facilitate the palladium-catalyzed aminopalladation of the alkyne moiety. This step generates a vinyl-palladium species which then isomerizes to an alkyl-palladium intermediate. The critical carbonylation step occurs when carbon monoxide, released from the decomposition of the formic acid/acetic anhydride mixture, inserts into the palladium-carbon bond to form an acyl-palladium intermediate. This acyl species is then subjected to oxidative addition by the diaryliodonium salt, generating a high-valent tetravalent palladium complex. The catalytic cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted imidazole product and regenerates the active palladium catalyst. This intricate dance of organometallic transformations ensures high selectivity and minimizes side reactions.
From an impurity control perspective, the mild reaction conditions play a pivotal role in maintaining product purity. By operating at 30°C, the thermal degradation of sensitive intermediates is minimized, reducing the formation of polymeric byproducts often seen in high-temperature cyclizations. The use of sodium bicarbonate as a mild base helps to neutralize acidic byproducts without promoting hydrolysis of the sensitive imidoyl chloride starting material. Furthermore, the stoichiometric control of reagents, specifically the slight excess of trifluoroethylimide chloride relative to propargylamine, drives the initial amidine formation to completion, preventing the accumulation of unreacted amine which could lead to downstream purification challenges. The choice of tetrahydrofuran (THF) as the solvent is also critical, as it effectively dissolves all reactants and stabilizes the palladium intermediates, ensuring a homogeneous reaction environment that promotes consistent kinetics throughout the batch.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
To implement this synthesis effectively, precise adherence to the optimized reaction parameters is required. The process involves mixing palladium chloride, triphenylphosphine, sodium bicarbonate, and the formic acid/acetic anhydride mixture in an organic solvent, followed by the addition of the three key substrates. The reaction is then maintained at a controlled temperature for a specific duration to ensure full conversion. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, and a mixture of formic acid and acetic anhydride in an organic solvent such as THF.
- Add trifluoroethylimide chloride, propargylamine, and diaryliodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits beyond mere chemical efficiency. The primary advantage lies in the significant reduction of raw material costs and logistical complexity. By utilizing commercially available and inexpensive starting materials such as aromatic amines (precursors to the imidoyl chloride) and simple propargylamine, the dependency on exotic or custom-synthesized reagents is eliminated. This shift not only lowers the direct material cost but also mitigates supply risk, as these commodities are produced by multiple global suppliers, ensuring continuity of supply even during market fluctuations. Furthermore, the avoidance of hazardous diazo reagents removes the need for specialized storage facilities and expensive safety protocols, indirectly reducing overhead costs associated with regulatory compliance and insurance.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive transition metal catalysts often required in cross-coupling reactions, as the palladium loading here is relatively low (5 mol%). Additionally, the use of formic acid and acetic anhydride as a safe, liquid carbon monoxide surrogate avoids the capital expenditure associated with high-pressure gas reactors and CO monitoring systems. The mild reaction temperature of 30°C significantly reduces energy consumption compared to traditional reflux conditions, leading to lower utility costs per kilogram of product. The high reaction efficiency and yields observed across diverse substrates mean less raw material is wasted, directly improving the overall process mass intensity and reducing waste disposal costs.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain resilience by simplifying the sourcing strategy. Since the key reagents like diaryliodonium salts and trifluoroethylimide chlorides can be prepared from widely available precursors, the supply chain is less vulnerable to single-source bottlenecks. The scalability of the reaction, demonstrated from milligram to gram scales in the patent data, suggests a smooth path to kilogram and ton-scale production without the need for extensive re-optimization. This predictability allows supply chain planners to forecast lead times more accurately and maintain leaner inventory levels, knowing that production batches can be turned around quickly due to the short reaction times and simple workup procedures.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method aligns well with green chemistry principles. The use of THF as a solvent, while requiring recovery, is standard in the industry and easily managed with existing distillation infrastructure. The reaction generates minimal hazardous waste compared to routes involving heavy metal oxidants or toxic gases. The ability to tolerate various functional groups means that protection and deprotection steps can often be avoided, shortening the overall synthetic sequence and reducing the total volume of solvents and reagents consumed. This streamlined approach facilitates easier regulatory approval for new drug applications, as the impurity profile is cleaner and the process is more reproducible on a large scale.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity on critical process parameters.
Q: What are the optimal reaction conditions for this carbonylation process?
A: The patent specifies a mild temperature of 30°C using THF as the solvent, with a reaction time of 16 to 24 hours to ensure complete conversion.
Q: Which catalyst system provides the highest efficiency?
A: The preferred system utilizes palladium chloride (PdCl2) combined with triphenylphosphine (PPh3) ligand, offering a balance of cost and catalytic activity.
Q: Can this method tolerate diverse functional groups on the aryl rings?
A: Yes, the method demonstrates excellent substrate compatibility, tolerating substituents such as methyl, tert-butyl, halogens, trifluoromethyl, and nitro groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists has extensively analyzed the potential of the palladium-catalyzed carbonylation route described in CN111423381A and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are designed to handle complex organometallic reactions safely, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 2-trifluoromethyl imidazole intermediate meets the highest industry standards. Whether you require custom synthesis for early-stage research or bulk manufacturing for clinical trials, our infrastructure is ready to support your project.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, identifying opportunities to optimize the synthetic route further. We encourage you to contact our technical procurement team today to request specific COA data for related intermediates and comprehensive route feasibility assessments. Let us help you secure a reliable supply of high-quality pharmaceutical intermediates while driving down your overall manufacturing costs through intelligent process engineering.
