Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
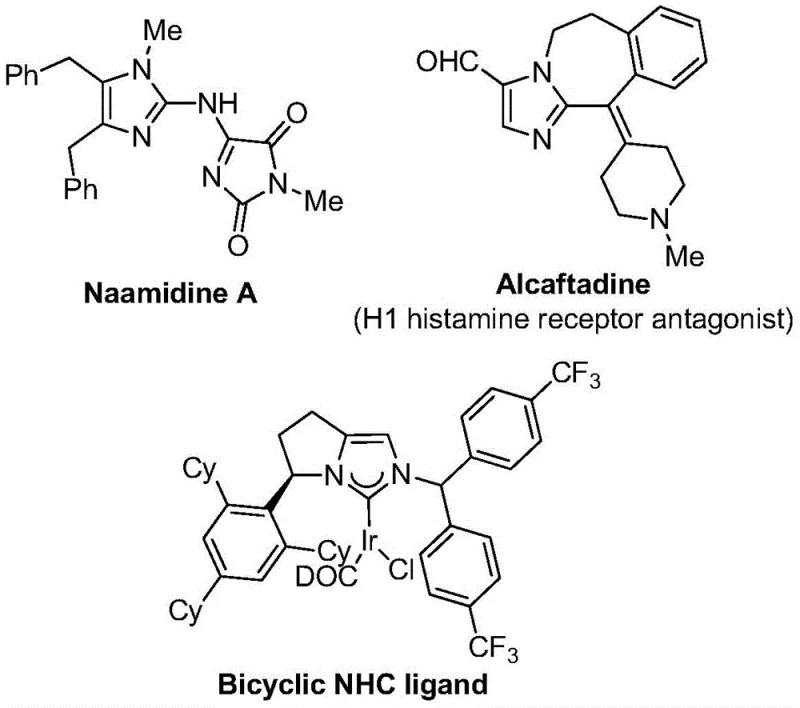
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds represents a cornerstone of modern medicinal chemistry, profoundly enhancing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As highlighted in patent CN111423381B, a novel and highly efficient preparation method for 2-trifluoromethyl substituted imidazole compounds has been developed, addressing critical bottlenecks in the synthesis of these valuable pharmacophores. This technology leverages a sophisticated palladium-catalyzed carbonylation strategy that transforms readily available starting materials—specifically trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts—into complex imidazole derivatives under remarkably mild conditions. The significance of this advancement cannot be overstated for the pharmaceutical industry, where the demand for fluorinated building blocks continues to surge. By utilizing a carbon monoxide surrogate system generated in situ from formic acid and acetic anhydride, this process eliminates the need for hazardous high-pressure CO gas, thereby streamlining the operational workflow and enhancing safety profiles for manufacturing facilities. Furthermore, the versatility of this synthetic route allows for the rapid generation of diverse molecular libraries, facilitating the discovery of next-generation therapeutics targeting various biological pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted nitrogen-containing heterocycles has been plagued by significant synthetic challenges that hinder efficient commercial production. Traditional methodologies often rely on the direct reaction of trifluoromethyl synthons, such as trifluorodiazoethane or specialized trifluoroethylimidoyl halides, with suitable substrates under rigorous conditions. These conventional routes frequently suffer from poor atom economy, limited substrate scope, and the requirement for harsh reaction parameters, including elevated temperatures and strong bases, which can degrade sensitive functional groups. Moreover, many existing protocols necessitate the use of stoichiometric amounts of expensive reagents or toxic heavy metal promoters, leading to complicated downstream purification processes and substantial waste generation. The reliance on gaseous carbon monoxide for carbonylation steps introduces severe safety hazards and logistical complexities, requiring specialized high-pressure reactors that are not universally available in standard pilot plants. Consequently, the scalability of these legacy methods is often compromised, resulting in inconsistent batch quality and inflated production costs that ultimately impact the commercial viability of potential drug candidates containing these motifs.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the invention disclosed in patent CN111423381B introduces a transformative catalytic manifold that redefines the efficiency of imidazole synthesis. This innovative approach employs a transition metal palladium catalyst system, specifically utilizing palladium chloride and triphenylphosphine, to orchestrate a multi-component coupling reaction at a gentle 30°C. The core brilliance of this method lies in its use of diaryl iodonium salts as oxidants and a formic acid/acetic anhydride mixture as a safe, liquid carbon monoxide surrogate. This strategic substitution not only mitigates the risks associated with handling toxic CO gas but also ensures a steady, controlled release of the carbonyl source directly within the reaction medium. The process demonstrates exceptional functional group tolerance, seamlessly accommodating a wide array of substituents including alkyl, halogen, nitro, and trifluoromethyl groups on the aromatic rings. As illustrated in the general reaction scheme, the convergence of trifluoroethylimidoyl chloride, propargylamine, and the iodonium salt proceeds with high regioselectivity to yield the desired 2-trifluoromethyl imidazole core. This streamlined one-pot procedure drastically reduces the number of isolation steps, minimizes solvent consumption, and delivers high yields, establishing a new benchmark for the sustainable manufacturing of fluorinated heterocycles.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
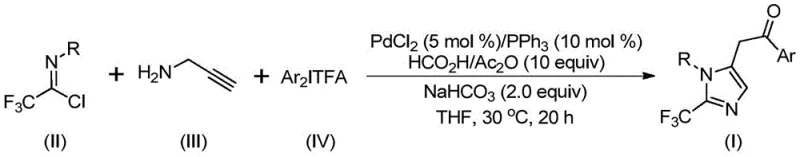
A deep understanding of the catalytic cycle is essential for optimizing this process for industrial application and ensuring consistent product quality. The reaction initiates with the formation of a trifluoroacetamidine intermediate through an intermolecular carbon-nitrogen bond formation promoted by the alkaline additive, sodium bicarbonate. Following an initial isomerization, the palladium catalyst engages with the alkyne moiety of the propargylamine derivative via palladation, generating a pivotal alkenyl palladium intermediate. This species subsequently undergoes isomerization to form a more stable alkyl palladium complex, setting the stage for the critical carbonylation event. The carbon monoxide, released in situ from the decomposition of the formic acid/acetic anhydride adduct, inserts into the palladium-carbon bond to create an acyl palladium intermediate. This step is the defining feature of the transformation, effectively building the ketone functionality adjacent to the heterocycle. The cycle culminates with the oxidative addition of the diaryl iodonium salt to the palladium center, forming a transient tetravalent palladium species, which then undergoes reductive elimination to release the final 2-trifluoromethyl-substituted imidazole product and regenerate the active palladium catalyst. This intricate dance of organometallic steps ensures high turnover numbers and minimizes the formation of side products, which is crucial for maintaining a clean impurity profile in pharmaceutical intermediates.
Controlling the impurity profile is paramount for any synthetic route intended for GMP manufacturing, and this mechanism offers inherent advantages in that regard. The mild reaction temperature of 30°C significantly suppresses thermal degradation pathways and non-selective radical reactions that often plague high-temperature syntheses. Furthermore, the use of specific ligands like triphenylphosphine helps stabilize the palladium center, preventing the aggregation of palladium black which can lead to catalyst deactivation and metal contamination in the final product. The choice of sodium bicarbonate as a mild base avoids the aggressive deprotonation conditions that could lead to polymerization of the alkyne starting material or hydrolysis of the sensitive imidoyl chloride. By carefully tuning the molar ratios of the reactants, particularly maintaining an excess of the trifluoroethylimidoyl chloride relative to the propargylamine, the process drives the equilibrium towards the desired product while minimizing the accumulation of unreacted amines that are difficult to remove. The post-treatment procedure, involving simple filtration followed by silica gel column chromatography, is robust enough to remove trace palladium residues and organic byproducts, ensuring the final API intermediate meets stringent purity specifications required by regulatory bodies.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
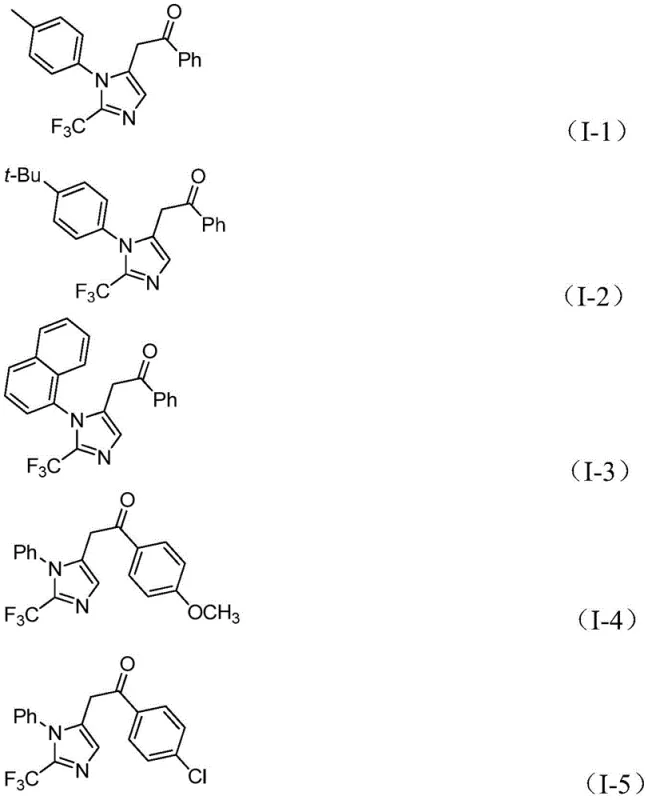
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory scale-up and industrial production. The protocol involves charging a reaction vessel with the palladium catalyst system, the carbonyl surrogate components, and the three key organic building blocks in a suitable aprotic solvent such as tetrahydrofuran. The detailed standardized synthesis steps below outline the precise addition order and workup procedures necessary to achieve optimal yields and purity.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16-24 hours, then filter and purify via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The shift from hazardous gaseous reagents to stable liquid surrogates fundamentally alters the risk profile of the manufacturing process, reducing insurance costs and eliminating the need for specialized high-pressure infrastructure. This translates directly into lower capital expenditure requirements for production facilities and faster deployment times for new product lines. Additionally, the reliance on commodity chemicals like formic acid, acetic anhydride, and palladium chloride ensures a resilient supply chain that is less susceptible to the volatility often seen with exotic reagents. The ability to run the reaction at near-ambient temperatures also results in significant energy savings, as there is no need for extensive heating or cooling loops, contributing to a lower overall carbon footprint for the manufacturing site. These factors combined create a robust value proposition for companies seeking to secure a reliable supply of high-quality fluorinated intermediates.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide equipment and the use of inexpensive, commercially available starting materials drastically simplify the production process. By avoiding the need for specialized autoclaves and complex gas handling systems, manufacturers can achieve substantial cost savings in both equipment maintenance and operational overhead. Furthermore, the high atom economy of the reaction minimizes raw material waste, ensuring that a greater proportion of input costs are converted into valuable product rather than disposal fees. The simplified workup procedure, which avoids complex extraction sequences, further reduces labor costs and solvent consumption, driving down the total cost of goods sold for these critical intermediates.
- Enhanced Supply Chain Reliability: The use of stable, shelf-stable reagents such as diaryl iodonium salts and trifluoroethylimidoyl chloride mitigates the risk of supply disruptions caused by the degradation of sensitive precursors. Unlike methods requiring cryogenic conditions or immediate use of unstable diazo compounds, this protocol allows for the stocking of key ingredients, enabling just-in-time manufacturing strategies that improve inventory turnover. The broad substrate scope means that a single manufacturing platform can be adapted to produce a wide variety of analogues without retooling, providing flexibility to respond rapidly to changing market demands or clinical trial requirements. This adaptability ensures a continuous supply of materials even when specific structural modifications are needed late in the development pipeline.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic gas emissions make this process inherently safer and more environmentally friendly, aligning with increasingly strict global environmental regulations. Scaling from gram to kilogram or ton quantities does not require a fundamental change in the reaction engineering, as the heat transfer requirements are minimal due to the low operating temperature. This linear scalability reduces the time and resources needed for process validation and tech transfer between sites. Additionally, the reduced generation of hazardous waste simplifies effluent treatment processes, lowering the environmental compliance burden and associated costs for the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and reliability for decision-makers.
Q: What are the key advantages of this palladium-catalyzed method over traditional synthesis?
A: This method operates at mild temperatures (30°C) using safe carbonyl surrogates instead of toxic CO gas, significantly improving operational safety and substrate compatibility compared to harsh traditional cyclization methods.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the protocol uses commercially available reagents like palladium chloride and formic acid, avoids high-pressure equipment, and demonstrates excellent yields up to 97%, making it highly scalable for industrial manufacturing.
Q: What is the substrate scope for the aryl groups in this reaction?
A: The reaction exhibits broad functional group tolerance, successfully accommodating electron-donating groups like methoxy and tert-butyl, as well as electron-withdrawing groups such as chloro, bromo, nitro, and trifluoromethyl on both the imidoyl chloride and iodonium salt components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has thoroughly analyzed the potential of this palladium-catalyzed carbonylation route and is fully prepared to leverage it for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl imidazole intermediate we deliver meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to unlock the full potential of this technology for your pipeline. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target timelines. We encourage you to reach out today to discuss your project specifics,索取 specific COA data for our reference standards, and obtain comprehensive route feasibility assessments that will demonstrate how we can optimize your supply chain for maximum efficiency and cost-effectiveness.
