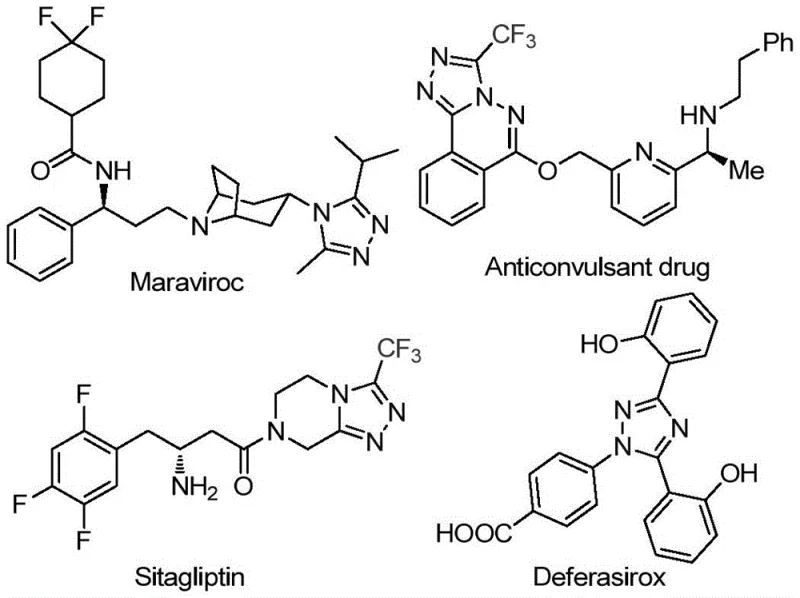
Revolutionizing Triazole Synthesis: Iodine-Catalyzed Method for Scalable Pharmaceutical Intermediate Production with Enhanced Purity
The Chinese patent CN113105402B discloses a groundbreaking method for synthesizing 3,4,5-trisubstituted 1,2,4-triazole compounds through an innovative iodine-catalyzed approach that eliminates the need for heavy metal catalysts while maintaining high reaction efficiency and product purity. This novel synthetic route represents a significant advancement in heterocyclic chemistry with direct implications for pharmaceutical manufacturing where triazole-containing compounds serve as critical building blocks for numerous therapeutic agents including antiviral drugs and metabolic disorder treatments. The process leverages readily available starting materials under mild reaction conditions that do not require anhydrous or oxygen-free environments, thereby simplifying operational complexity while enhancing scalability potential for commercial production. By addressing longstanding challenges in triazole synthesis such as metal catalyst contamination and narrow substrate scope, this patent offers a robust solution that aligns with modern pharmaceutical industry requirements for sustainable and cost-effective manufacturing processes that maintain stringent quality standards without compromising on yield or purity parameters.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing 3,4,5-trisubstituted 1,2,4-triazole compounds have historically relied on transition metal-catalyzed reactions that require strict anhydrous and oxygen-free conditions to prevent catalyst deactivation and ensure acceptable yields. These approaches often involve expensive palladium or copper catalysts that necessitate complex removal procedures to meet pharmaceutical purity standards, significantly increasing production costs and complicating scale-up processes. The narrow substrate scope of conventional methods limits their applicability when introducing specific functional groups such as trifluoromethyl moieties that are crucial for enhancing the physicochemical properties of pharmaceutical compounds including bioavailability and metabolic stability. Furthermore, the multi-step nature of traditional syntheses often results in lower overall yields due to intermediate purification requirements and sensitivity to reaction conditions that demand specialized equipment and highly controlled environments not readily available in standard manufacturing facilities.
The Novel Approach
The patented iodine-catalyzed method represents a paradigm shift in triazole synthesis by eliminating transition metal catalysts entirely while maintaining excellent reaction efficiency under significantly milder conditions that do not require specialized environmental controls. This innovative approach utilizes inexpensive and readily available starting materials including aryl ethyl ketones and trifluoroethylimide hydrazides that react through a tandem cyclization mechanism facilitated by iodine in dimethyl sulfoxide solvent at moderate temperatures between 90-130°C. The process demonstrates remarkable functional group tolerance that enables the incorporation of diverse substituents at the 3 and 4 positions while simultaneously introducing both trifluoromethyl and acyl groups essential for pharmaceutical applications. Crucially, the method's simplicity allows for straightforward scale-up from laboratory to commercial production without requiring significant process modifications or specialized equipment investments that would otherwise hinder rapid implementation in existing manufacturing facilities.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism begins with iodine-mediated Kornblum oxidation of aryl ethyl ketones in dimethyl sulfoxide solvent at elevated temperatures (90-110°C), generating aryl diketone intermediates through a well-established iodination/oxidation sequence that proceeds without requiring anhydrous conditions typically necessary for transition metal catalysis. This critical first step creates the necessary electrophilic centers that subsequently react with trifluoroethylimide hydrazides under the influence of sodium dihydrogen phosphate and pyridine at higher temperatures (110-130°C), forming hydrazone intermediates through dehydration condensation reactions that benefit from the dual activation provided by both iodine and the phosphate buffer system. The resulting hydrazone species then undergo intramolecular cyclization through nucleophilic attack on the carbonyl carbon by the terminal hydrazine nitrogen atom, followed by aromatization to yield the final triazole ring structure with simultaneous incorporation of the trifluoromethyl group at position 5 and acyl substituent at position 4.
Impurity control is achieved through the carefully balanced reaction conditions that favor selective formation of the desired triazole product while minimizing side reactions; the use of iodine as both oxidant and catalyst creates a self-regulating system where excess iodine is consumed in the initial oxidation step before participating in the cyclization phase. The phosphate buffer system maintains optimal pH conditions throughout the reaction sequence to prevent unwanted hydrolysis or decomposition of sensitive intermediates while pyridine serves as both base and coordinating ligand that stabilizes reactive species without introducing additional metal contaminants. This elegant mechanistic design ensures high product purity without requiring extensive purification steps beyond standard column chromatography, making it particularly suitable for pharmaceutical applications where stringent impurity profiles must be maintained to meet regulatory requirements for drug substances.
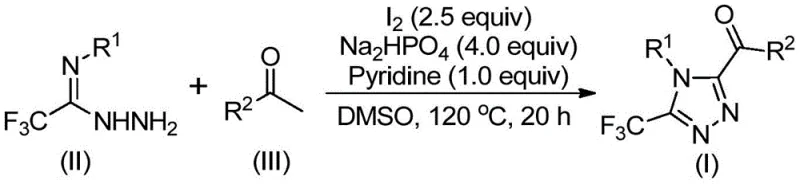
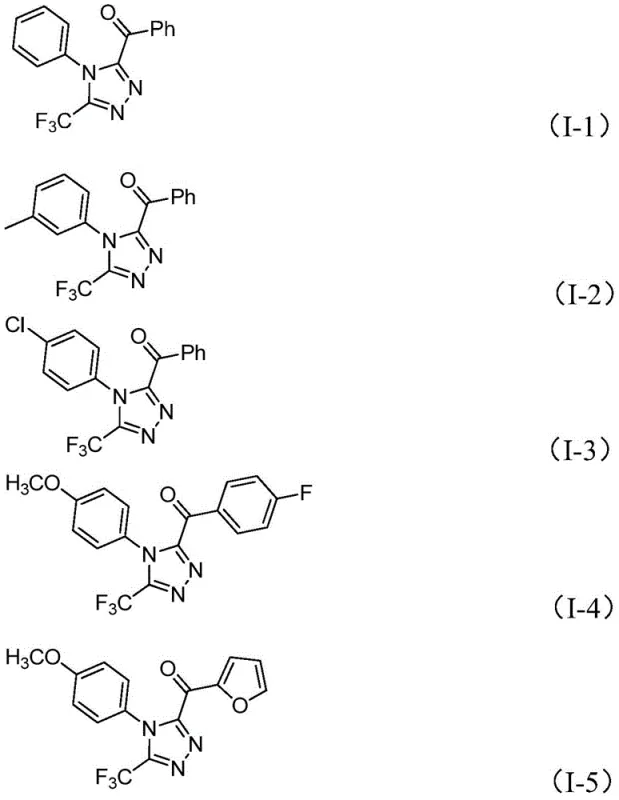
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
This innovative synthesis route represents a significant advancement over conventional methods by eliminating transition metal catalysts while maintaining excellent yield and purity profiles across diverse substrate combinations. The process begins with iodination/Kornblum oxidation of aryl ethyl ketones followed by tandem cyclization with trifluoroethylimide hydrazides under mild conditions that do not require specialized environmental controls or expensive catalyst removal procedures. Detailed standardized synthesis steps are provided below to facilitate seamless implementation in pharmaceutical manufacturing environments where consistent product quality and process reliability are paramount considerations for regulatory compliance and commercial success.
- Combine aryl ethyl ketone with iodine in DMSO solvent at 90-110°C for 4-6 hours to form aryl diketone intermediates through iodination/Kornblum oxidation without requiring anhydrous conditions.
- Add trifluoroethylimide hydrazide along with sodium dihydrogen phosphate and pyridine at elevated temperature (110-130°C) for extended reaction time (12-20 hours) to facilitate tandem cyclization.
- Complete synthesis through standard post-processing including filtration and column chromatography purification to obtain high-purity triazole products suitable for pharmaceutical applications.
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthetic approach addresses critical pain points in pharmaceutical intermediate supply chains by offering a streamlined manufacturing process that eliminates dependency on specialized catalysts and complex environmental controls required by conventional methods. The elimination of heavy metal catalysts removes a major source of supply chain vulnerability while simultaneously reducing production complexity and associated costs throughout the manufacturing lifecycle from laboratory development through commercial scale-up to routine production.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts represents substantial cost savings throughout production; avoiding costly metal contamination issues removes requirements for specialized purification equipment and extensive quality control testing needed to ensure compliance with strict metal residue limits in pharmaceutical products.
- Enhanced Supply Chain Reliability: Utilizing readily available starting materials from multiple global suppliers significantly reduces single-source dependency risks while simplified reaction conditions enable consistent production across diverse manufacturing facilities without requiring specialized infrastructure investments typically needed for metal-catalyzed reactions.
- Scalability and Environmental Compliance: The straightforward process design allows seamless scale-up from laboratory to commercial production volumes while maintaining excellent product quality; elimination of toxic heavy metals from manufacturing significantly reduces environmental impact compared to conventional methods generating hazardous metal-containing byproducts requiring specialized disposal protocols.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding implementation of this novel triazole synthesis method in pharmaceutical manufacturing environments; answers are based on detailed technical analysis of patented process parameters validated through extensive experimental data presented in original patent documentation.
Q: How does this iodine-catalyzed method improve upon traditional triazole synthesis approaches?
A: This novel method eliminates transition metal catalysts entirely while maintaining high functional group tolerance; it operates under standard atmospheric conditions without requiring anhydrous environments or specialized equipment typically needed for conventional approaches.
Q: What are the key advantages of this synthesis method for pharmaceutical manufacturing?
A: The process delivers significant cost reduction by removing expensive metal catalyst removal steps while ensuring consistent high purity; its simplified operation enables seamless scale-up from laboratory to commercial production without compromising quality parameters.
Q: How does this process address supply chain reliability concerns?
A: By utilizing widely available starting materials from multiple global suppliers and avoiding specialized catalyst dependencies, this method ensures consistent production capacity while maintaining stringent quality standards required for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required for pharmaceutical applications through our state-of-the-art QC labs equipped with advanced analytical instrumentation capable of detecting impurities at trace levels. As a leading CDMO partner specializing in complex heterocyclic chemistry including triazole-based intermediates, we have successfully implemented this patented iodine-catalyzed synthesis method across multiple client projects with excellent results in terms of yield consistency and product quality that meet or exceed regulatory requirements for pharmaceutical manufacturing.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis approach can optimize your specific manufacturing requirements; please contact us to obtain detailed COA data and route feasibility assessments tailored to your production needs.