Scalable Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles: A Breakthrough in Metal-Free Heterocyclic Chemistry
Introduction to Advanced Triazole Synthesis Technology
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, particularly the 1,2,4-triazole scaffold, which serves as a critical pharmacophore in numerous bioactive molecules. As highlighted in patent CN113105402A, published in July 2021, a novel preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds has been developed that addresses many of the limitations associated with traditional synthetic routes. This technology is particularly relevant for the production of high-value intermediates used in drugs such as Maraviroc, Sitagliptin, and Deferasirox, where the introduction of a trifluoromethyl group can drastically enhance metabolic stability and lipophilicity.
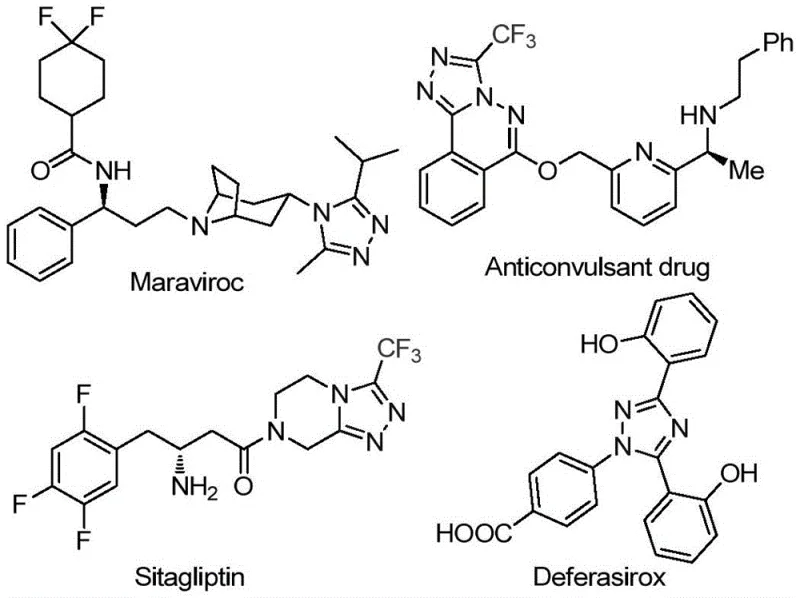
The significance of this patent lies in its ability to generate complex heterocyclic frameworks bearing both trifluoromethyl and acyl groups, a structural motif that is notoriously difficult to access via conventional means. By leveraging a non-metal-promoted synthesis strategy, this approach not only improves the physicochemical properties of the resulting compounds but also aligns with modern green chemistry principles by eliminating the need for precious metal catalysts. For R&D directors and procurement managers alike, this represents a pivotal shift towards more sustainable and cost-effective manufacturing processes for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles has relied heavily on transition metal catalysis or harsh reaction conditions that pose significant challenges for scale-up and regulatory compliance. Traditional methods often require strict anhydrous and oxygen-free environments, necessitating specialized equipment and increasing operational costs substantially. Furthermore, the reliance on heavy metal catalysts introduces a critical bottleneck in the downstream processing, as removing trace metal residues to meet stringent pharmaceutical purity specifications often requires complex and expensive purification steps. These factors collectively contribute to longer lead times and higher overall production costs, making conventional routes less attractive for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN113105402A offers a streamlined alternative that utilizes cheap and easily obtainable starting materials such as arylethanone and trifluoroethylimine hydrazide. This innovative route operates under relatively mild conditions in dimethyl sulfoxide (DMSO) and, crucially, does not require the exclusion of air or moisture, thereby simplifying the reactor setup and operational protocol. The use of elemental iodine as a promoter instead of a stoichiometric heavy metal catalyst not only reduces the raw material costs but also eliminates the risk of heavy metal contamination in the final product. This simplicity in operation, combined with the wide tolerance for various functional groups on the aryl rings, makes this method highly versatile for generating diverse libraries of triazole derivatives for drug discovery and development.
Mechanistic Insights into Iodine-Promoted Cyclization
The core of this synthetic breakthrough involves a tandem sequence initiated by the iodine-promoted oxidation of arylethanone, likely proceeding through an alpha-iodination followed by a Kornblum oxidation to generate an aryl diketone intermediate in situ. This reactive species then undergoes a dehydration condensation with trifluoroethylimine hydrazide to form a hydrazone intermediate, which subsequently cyclizes under the combined promotion of iodine and base to yield the final 3,4,5-trisubstituted 1,2,4-triazole compound. The elegance of this mechanism lies in its one-pot nature, where multiple bond-forming events occur sequentially without the need for isolating unstable intermediates, thus maximizing atom economy and minimizing waste generation.
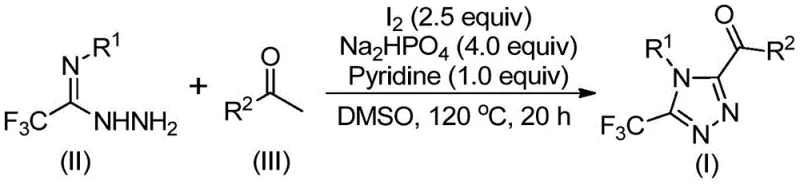
From an impurity control perspective, the use of sodium dihydrogen phosphate and pyridine as additives plays a vital role in buffering the reaction medium and facilitating the cyclization step while suppressing side reactions. The specific molar ratio of sodium dihydrogen phosphate to pyridine to elemental iodine (optimized at 4:1:2.5) ensures that the reaction proceeds with high conversion rates, minimizing the formation of by-products that could complicate purification. For quality control teams, understanding this mechanistic pathway is essential for establishing robust in-process controls that ensure consistent batch-to-batch reproducibility and high purity of the final API intermediate.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for executing this transformation with high efficiency and reliability. The process begins with the activation of the arylethanone substrate in DMSO, followed by the sequential addition of the hydrazide component and promoters to drive the cyclization to completion. Detailed standard operating procedures regarding temperature ramping, addition rates, and workup protocols are critical for achieving the reported yields and maintaining safety standards during scale-up. The following guide summarizes the key operational steps derived from the patent data to assist process chemists in implementing this technology.
- Add arylethanone and iodine into dimethyl sulfoxide (DMSO) and heat to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Introduce additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimine hydrazide into the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis route offers tangible benefits that extend beyond mere chemical novelty. The primary advantage stems from the utilization of commodity chemicals like arylethanone and elemental iodine, which are widely available in the global market at competitive prices, ensuring a stable and resilient supply chain. Unlike processes dependent on scarce precious metals or custom-synthesized ligands, this method mitigates the risk of raw material shortages and price volatility, providing a secure foundation for long-term production planning and cost management strategies.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the avoidance of rigorous anhydrous conditions lead to a substantial reduction in both material and operational expenditures. By removing the need for specialized drying agents, inert gas purging, and complex metal scavenging steps during purification, the overall cost of goods sold (COGS) is significantly lowered. This economic efficiency allows manufacturers to offer high-purity pharmaceutical intermediates at more competitive price points, enhancing market competitiveness without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this chemical process against atmospheric moisture and oxygen translates directly into improved supply chain reliability. Facilities do not need to invest in specialized gloveboxes or highly sophisticated inert atmosphere reactors to run this chemistry, allowing for greater flexibility in manufacturing site selection and capacity utilization. This operational simplicity reduces the likelihood of batch failures due to environmental excursions, ensuring consistent delivery schedules and strengthening partnerships with downstream API manufacturers.
- Scalability and Environmental Compliance: The method is explicitly designed to be easily expanded from gram-scale laboratory experiments to multi-ton commercial production, addressing a common pain point in process development. Furthermore, the absence of toxic heavy metals simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations and corporate sustainability goals. This eco-friendly profile not only reduces the environmental footprint of the manufacturing process but also minimizes the regulatory burden associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction scope, safety, and scalability for potential partners and stakeholders.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN113105402A utilizes elemental iodine as a promoter instead of toxic or expensive heavy metal catalysts, significantly simplifying purification and reducing environmental impact.
Q: What are the typical reaction conditions for this triazole formation?
A: The reaction proceeds in dimethyl sulfoxide (DMSO) without the need for strict anhydrous or oxygen-free conditions. It involves an initial heating stage at 90-110°C followed by a second stage at 110-130°C.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method is easily expanded to the gram level and potentially industrial scale due to the use of cheap, commercially available raw materials and simple post-treatment procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced heterocyclic chemistry in accelerating drug discovery and development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory methodologies like the one described in CN113105402A can be successfully translated into reliable industrial processes. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole intermediate performs consistently in your downstream synthesis.
We invite you to collaborate with us to leverage this cost-effective and scalable technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain objectives and drive innovation in your pharmaceutical programs.
