Advanced Palladium-Catalyzed Synthesis of 3-Arylquinoline Derivatives for Commercial Pharmaceutical Manufacturing
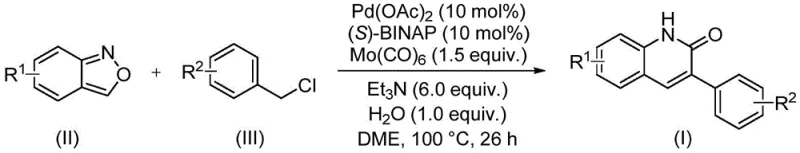
Patent CN113045489B introduces a groundbreaking methodology for synthesizing 3-arylquinoline-2(1H) ketone derivatives through a novel palladium-catalyzed aminocarbonylation process that represents a significant advancement in heterocyclic compound synthesis for pharmaceutical applications. This innovative approach utilizes benzisoxazole as both nitrogen and formyl source within a single reaction vessel, eliminating the need for multiple reagents and complex sequential steps that characterize conventional synthetic routes. The technology addresses longstanding challenges in quinolinone production by providing exceptional functional group tolerance across diverse substrate combinations while maintaining operational simplicity under mild reaction conditions. With its demonstrated ability to produce high-value intermediates at yields up to 97% across fifteen different substrate variations, this methodology offers pharmaceutical manufacturers unprecedented flexibility in developing new therapeutic agents while significantly reducing production complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for quinolinone derivatives, including Vilsmeier-Haack, Knorr, and Friedlander reactions, often require harsh conditions such as strong acids or high temperatures that limit functional group compatibility and generate significant waste streams complicating purification processes. These methods typically suffer from narrow substrate scope due to sensitivity toward various functional groups, necessitating extensive protection/deprotection strategies that increase both cost and production time while reducing overall yield efficiency. The multi-step nature of conventional approaches creates substantial barriers to process scalability as each additional step introduces potential failure points that compromise product consistency at commercial scale. Furthermore, traditional syntheses frequently rely on expensive transition metal catalysts requiring complex removal procedures to meet pharmaceutical purity standards, creating additional processing challenges that impact both cost structure and environmental sustainability metrics in manufacturing operations.
The Novel Approach
The patented methodology (CN113045489B) overcomes these limitations through an elegant one-pot palladium-catalyzed aminocarbonylation reaction that utilizes readily available benzisoxazole as dual functionality source within a carefully optimized catalytic system. Operating under mild conditions at precisely 100°C in DME solvent, this process eliminates the need for pre-formed amine and aldehyde components while maintaining excellent functional group compatibility across diverse substrates including those containing halogens, cyano groups, and various alkyl/aryl substituents. The strategic use of benzisoxazole as both nitrogen and formyl source streamlines the synthetic pathway by reducing reagent count from six or more components in conventional methods to just four essential reagents plus catalysts in this innovative approach. This methodology achieves consistently high yields (74%-97% across fifteen examples) with minimal byproduct formation through precise control of catalyst composition ratios, significantly improving atom economy while reducing environmental impact compared to traditional synthetic routes commonly employed in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The reaction mechanism begins with oxidative addition of benzyl chloride to the palladium(0) species generated in situ from palladium acetate and (S)-BINAP ligand under inert atmosphere conditions. This forms a key arylpalladium intermediate that subsequently coordinates with benzisoxazole, triggering ring-opening to release the nitrogen functionality while simultaneously providing the formyl group through a unique rearrangement pathway that avoids external carbon monoxide sources typically required in carbonylation reactions. The carbonyl molybdenum source facilitates CO insertion into the arylpalladium bond at precisely controlled temperature conditions, creating an acylpalladium species that undergoes intramolecular cyclization to form the quinolinone core structure through regioselective ring closure. This cascade process is critically enabled by the dual functionality of benzisoxazole as both nitrogen donor and formyl precursor, eliminating the need for separate amine sources while maintaining excellent regioselectivity throughout the transformation without requiring additional directing groups or protecting strategies.
Impurity control in this process is achieved through precise optimization of reaction parameters including temperature control at exactly 100°C and strict adherence to specified molar ratios of palladium catalyst (0.1 mol%), (S)-BINAP ligand (0.1 mol%), triethylamine (6.0 equiv.), and water (1.0 equiv.). The chiral environment created by the (S)-BINAP ligand minimizes undesired side reactions such as dimerization or over-reduction pathways while promoting selective cyclization to the desired quinolinone structure through steric control mechanisms inherent in the ligand design. Water plays a crucial role as both proton shuttle and mild base during key transformation steps without requiring additional acid/base catalysts that could introduce new impurity pathways or complicate purification procedures. This optimized system consistently delivers high-purity products as evidenced by NMR data across multiple examples without requiring specialized purification techniques beyond standard column chromatography protocols commonly available in pharmaceutical manufacturing facilities.
How to Synthesize 3-Arylquinolinone Derivatives Efficiently
This patented methodology offers pharmaceutical manufacturers a streamlined pathway to produce high-value quinolinone intermediates with exceptional efficiency and purity through carefully optimized reaction parameters that ensure consistent product quality across different production scales. The process begins with precise selection of appropriately substituted benzisoxazole precursors based on target molecular requirements followed by accurate measurement of catalyst components according to established molar ratios that have been validated across multiple substrate variations during patent development work. Reaction setup requires only standard laboratory equipment with temperature control capabilities making it readily adaptable to existing manufacturing facilities without significant capital investment or specialized infrastructure requirements.
- Prepare reaction mixture with precise molar ratios: benzisoxazole (1 equiv), benzyl chloride (2.5 equiv), palladium acetate (0.1 mol%), (S)-BINAP (0.1 mol%), triethylamine (6.0 equiv), water (1.0 equiv), and Mo(CO)6 (1.5 equiv) in DME solvent.
- Heat reaction mixture to precisely 100°C under inert atmosphere with continuous stirring for exactly 26 hours to ensure complete conversion while minimizing side reactions.
- Perform standard workup procedure: filter reaction mixture through celite, mix with silica gel, and purify product using column chromatography to obtain high-purity quinolinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic route delivers substantial value to procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate manufacturing through simplified material requirements and enhanced process robustness that directly impact operational efficiency metrics across multiple dimensions of supply chain performance. The methodology significantly reduces raw material complexity while maintaining access to diverse structural variants needed for drug development programs providing procurement teams with greater flexibility in supplier selection strategies without compromising on quality requirements or regulatory compliance standards essential for pharmaceutical applications.
- Cost Reduction in Manufacturing: The simplified one-pot process eliminates expensive reagents and complex purification steps required by conventional methods resulting in substantial cost savings through reduced material consumption lower processing costs and minimized waste disposal expenses associated with multi-step syntheses commonly employed in quinolinone production.
- Enhanced Supply Chain Reliability: Utilization of readily available starting materials with established global supply chains ensures consistent access to critical raw materials regardless of regional disruptions while operational simplicity allows rapid technology transfer between manufacturing sites providing procurement teams with multiple sourcing options to mitigate supply chain risks effectively.
- Scalability and Environmental Compliance: Demonstrated scalability from laboratory scale through pilot plant validation enables seamless transition from development phase to commercial production without requiring significant process modifications while reduced number of processing steps lowers environmental impact aligning with green chemistry principles increasingly demanded by regulatory agencies worldwide.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations derived directly from patent documentation reflecting specific advantages operational parameters established through rigorous experimental validation during development phase of this innovative synthetic methodology.
Q: How does this methodology improve upon traditional quinolinone synthesis routes?
A: The patented process eliminates multiple synthetic steps required by conventional methods like Vilsmeier-Haack or Friedlander reactions by utilizing benzisoxazole as dual nitrogen and formyl source, resulting in simplified workflow with higher atom economy and reduced waste generation while maintaining excellent functional group tolerance.
Q: What are the key advantages for pharmaceutical manufacturers regarding impurity control?
A: The optimized catalyst system with precise molar ratios creates a controlled reaction environment that minimizes side products while maintaining excellent functional group tolerance across diverse substrates, consistently delivering high-purity products suitable for pharmaceutical applications without requiring additional purification steps.
Q: How does this process support commercial scale-up requirements?
A: The methodology demonstrates exceptional scalability from laboratory to commercial production without requiring significant process modifications, operating under mild conditions with standard equipment while maintaining high yields across diverse substrate combinations through robust reaction parameters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolinone Derivative Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex pharmaceutical intermediates while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical capabilities for comprehensive impurity profiling required by global regulatory authorities. This patented methodology represents just one example of our commitment to developing innovative solutions that address critical challenges in pharmaceutical manufacturing where we have successfully implemented similar catalytic processes across multiple therapeutic areas demonstrating consistent ability to deliver high-quality intermediates meeting the most demanding regulatory requirements worldwide.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who can provide detailed COA data route feasibility assessments tailored specifically to your production requirements ensuring optimal implementation strategy for your manufacturing operations.
