Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharma
Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharma
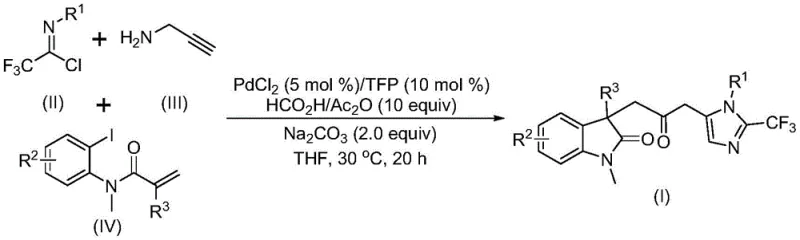
The pharmaceutical industry continuously seeks efficient routes to complex heterocyclic scaffolds that serve as critical building blocks for new drug candidates. A significant breakthrough in this domain is detailed in patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. This technology addresses the longstanding challenges associated with constructing indolinone and imidazole fused systems, which are prevalent in molecules exhibiting broad-spectrum biological activities. By leveraging a transition metal palladium-catalyzed carbonylation cascade, this invention enables the one-pot assembly of diversified double heterocyclic compounds containing trifluoromethyl and carbonyl functionalities. For R&D directors and procurement specialists, this represents a pivotal shift towards safer, more atom-economical processes that bypass the need for toxic carbon monoxide gas while maintaining high reaction efficiency and substrate compatibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of biheterocyclic frameworks has relied on three primary strategies, each fraught with significant operational and safety drawbacks that hinder commercial viability. The first approach involves the direct coupling of two pre-formed heterocycle substrates, which often suffers from low atom economy and requires harsh conditions to overcome the kinetic stability of the heterocyclic rings. The second method utilizes substrates bearing dual nucleophiles reacting with activated methyl-substituted heterocycles via oxidative cyclization; however, this pathway frequently necessitates stoichiometric oxidants and generates substantial chemical waste. The third conventional route employs transition metal-catalyzed tandem cyclizations, which, while efficient, historically struggled with the specific incorporation of carbonyl bridges due to the difficulty of controlling carbonylation steps without using dangerous high-pressure carbon monoxide gas. These limitations collectively result in elevated production costs, complex purification protocols, and significant safety risks that supply chain managers strive to avoid in modern API manufacturing.
The Novel Approach
In stark contrast to legacy techniques, the patented methodology introduces a streamlined, palladium-catalyzed multicomponent reaction that operates under remarkably mild conditions. By utilizing cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide, the process achieves the simultaneous formation of multiple chemical bonds in a single operational step. Crucially, the system replaces toxic external carbon monoxide sources with an in situ generation mechanism using a formic acid and acetic anhydride mixture, drastically improving workplace safety and reducing regulatory burdens. This innovative strategy not only simplifies the synthetic workflow but also expands the accessible chemical space, allowing for the rapid generation of libraries with diverse substituents at the R1, R2, and R3 positions. The ability to execute this transformation at 30°C in common solvents like THF underscores its potential as a robust platform for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic elegance of this transformation lies in its intricate cascade sequence, which orchestrates multiple catalytic cycles to forge the complex biheterocyclic architecture. The reaction is initiated by the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, followed by an intramolecular Heck reaction that generates a divalent alkyl-palladium intermediate. This key intermediate subsequently undergoes a carbonylation event driven by carbon monoxide released from the formic acid and acetic anhydride additive mixture, yielding a reactive acyl-palladium species. Concurrently, a base-promoted intermolecular carbon-nitrogen bond formation occurs between the trifluoroethylimidoyl chloride and propargylamine, producing a trifluoroacetamidine compound that undergoes isomerization. The convergence of these pathways culminates when the acyl-palladium intermediate activates the trifluoroacetamidine species, catalyzing an intramolecular cyclization that delivers the final carbonyl-bridged biheterocyclic product with high fidelity.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses by minimizing the isolation of unstable intermediates that often degrade into hard-to-remove byproducts. The use of a specific ligand system, such as trifurylphosphine, alongside palladium chloride, ensures high catalyst turnover and selectivity, effectively suppressing side reactions like homocoupling or premature hydrolysis. Furthermore, the mild reaction temperature of 30°C prevents thermal decomposition of sensitive functional groups, ensuring that the final crude mixture contains a cleaner profile of the desired product. This inherent selectivity reduces the burden on downstream purification units, allowing for simpler workup procedures involving filtration and standard silica gel chromatography. For quality assurance teams, this translates to a more predictable impurity profile and higher overall yields, which are critical metrics for validating a reliable pharmaceutical intermediate supplier.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
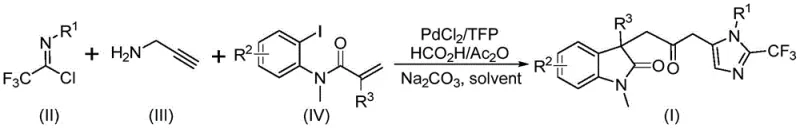
Implementing this synthesis route requires precise attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The protocol dictates the use of a palladium catalyst system comprising palladium chloride and trifurylphosphine, combined with sodium carbonate as a base and a formic acid/acetic anhydride mixture as the carbonyl source. Substrates including trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative are dissolved in an aprotic organic solvent, with tetrahydrofuran being the preferred medium for optimal conversion. The reaction proceeds at a controlled temperature of 30°C for a duration of 12 to 20 hours, after which the mixture is filtered and subjected to column chromatography. Detailed standardized synthesis steps for specific derivatives are outlined in the guide below.
- Mix palladium chloride, trifurylphosphine, sodium carbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and acrylamide substrates to the reaction vessel under stirring.
- React the mixture at 30°C for 12-20 hours, then filter and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers transformative benefits that directly impact the bottom line and operational resilience. By shifting away from hazardous high-pressure gases and expensive specialized equipment, the process significantly lowers the capital expenditure required for reactor setup and maintenance. The reliance on commercially available, commodity-grade starting materials ensures a stable supply chain that is less susceptible to the volatility often seen with exotic reagents. Moreover, the simplified one-pot nature of the reaction reduces labor costs and solvent consumption, contributing to substantial cost savings in API manufacturing without compromising on the structural complexity of the final molecule.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas removes the need for expensive high-pressure autoclaves and specialized gas handling infrastructure, leading to significant capital and operational expenditure reductions. Additionally, the use of inexpensive catalysts like palladium chloride and readily available ligands minimizes raw material costs compared to proprietary catalytic systems. The high atom economy of the multicomponent reaction means less waste generation, which further lowers disposal costs and improves the overall green chemistry profile of the production line.
- Enhanced Supply Chain Reliability: Since all key starting materials, including propargylamine and acrylamide derivatives, are commodity chemicals available from multiple global vendors, the risk of supply disruption is drastically minimized. The robustness of the reaction conditions, operating at mild temperatures and atmospheric pressure, allows for flexible manufacturing scheduling and reduces the likelihood of batch failures due to equipment malfunction. This reliability ensures consistent delivery timelines for downstream customers, reinforcing the manufacturer's reputation as a dependable partner in the pharmaceutical value chain.
- Scalability and Environmental Compliance: The process has been successfully demonstrated at the gram scale with clear pathways for expansion to kilogram and ton-level production, supported by simple workup procedures that do not require complex quenching steps. The avoidance of toxic gases and the use of standard organic solvents facilitate easier compliance with environmental regulations regarding emissions and waste treatment. This scalability, combined with a safer operational footprint, makes the technology ideally suited for the commercial scale-up of complex heterocycles required for late-stage drug development.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on safety, scope, and scalability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this synthesis method require toxic carbon monoxide gas?
A: No, the method utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in situ, eliminating the need for handling hazardous high-pressure CO gas cylinders.
Q: What is the substrate compatibility of this palladium-catalyzed reaction?
A: The reaction exhibits excellent functional group tolerance, accommodating various substituents such as halogens, alkyl groups, alkoxy groups, nitro, and trifluoromethyl groups on the aromatic rings.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the protocol has been validated at the gram scale with mild reaction conditions (30°C) and simple post-treatment, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic methodologies to accelerate drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop innovation to industrial reality. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for high-purity biheterocyclic compounds.
