Advanced Indole-Based Phosphine Ligands: Scalable Synthesis and Catalytic Applications
The landscape of transition metal catalysis is continually evolving, driven by the demand for more robust and efficient ligand systems that can withstand rigorous industrial conditions while maintaining high selectivity. A significant advancement in this domain is detailed in patent CN107445989B, which introduces a novel class of phosphine ligands featuring a 3-(disubstituted phosphino)-2-(2-(disubstituted phosphino) substituted phenyl)-1-alkyl-indole skeleton. These compounds represent a strategic evolution in ligand design, addressing the persistent challenges of catalyst deactivation and limited substrate scope often encountered in complex cross-coupling reactions. By integrating a rigid indole core with bulky phosphine substituents, this technology offers a unique steric and electronic environment that stabilizes active palladium species, thereby enhancing turnover numbers and enabling reactions under milder conditions. For R&D directors and process chemists seeking reliable pharmaceutical intermediate supplier partnerships, understanding the structural nuances and synthetic accessibility of these ligands is paramount for optimizing downstream synthetic routes.
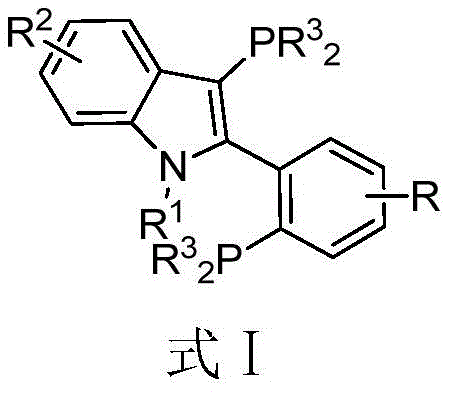
The development of effective phosphine ligands has long been a bottleneck in the synthesis of biaryls and heteroaryl compounds, which are ubiquitous motifs in active pharmaceutical ingredients (APIs) and agrochemicals. Conventional methods often rely on ligands that are either prohibitively expensive, difficult to synthesize, or susceptible to oxidation, leading to inconsistent batch-to-batch performance. Traditional systems, such as those based on simple triarylphosphines or early-generation biaryl phosphines, frequently struggle with the activation of challenging substrates like aryl chlorides or the formation of carbon-heteroatom bonds without requiring excessive catalyst loading. Furthermore, the sensitivity of many existing ligands to air and moisture necessitates stringent inert atmosphere protocols, increasing operational complexity and cost in large-scale manufacturing environments. These limitations underscore the critical need for next-generation ligands that combine high catalytic activity with practical handling properties.
The novel approach presented in the patent data overcomes these historical limitations through a rational design strategy centered on the indole scaffold. Unlike flexible alkyl chains found in older ligand generations, the indole backbone provides a rigid framework that pre-organizes the phosphine donors for optimal coordination with the metal center. This structural rigidity not only enhances the thermal stability of the resulting catalyst complex but also allows for fine-tuning of the electronic properties via substitution on the indole nitrogen or the phenyl ring. The patent explicitly highlights that these ligands can be synthesized via a straightforward sequence involving Fischer indole synthesis, bromination, and phosphorylation, utilizing commercially available starting materials. This accessibility translates directly into cost reduction in pharmaceutical intermediate manufacturing, as it eliminates the need for exotic precursors or multi-step protecting group strategies often required for other privileged ligand classes.
Mechanistic Insights into Indole-Based Bidentate Phosphine Coordination
The catalytic efficacy of the 3-(disubstituted phosphino)-2-(2-(disubstituted phosphino) substituted phenyl)-1-alkyl-indole ligands stems from their ability to form stable, chelated complexes with transition metals such as palladium. In the catalytic cycle, the bidentate nature of the ligand—where two phosphine groups coordinate to the metal center—creates a well-defined coordination sphere that protects the active site from decomposition pathways like aggregation into palladium black. The steric bulk provided by substituents such as cyclohexyl or tert-butyl groups on the phosphorus atoms prevents the approach of unwanted nucleophiles that could lead to side reactions, while simultaneously promoting the oxidative addition step, which is often the rate-determining step in cross-coupling reactions involving sterically hindered aryl halides. This balance of steric protection and electronic donation is crucial for maintaining high turnover frequencies over extended reaction times.
Furthermore, the electronic tunability of the indole core allows chemists to modulate the electron density at the metal center, which is essential for adapting the catalyst to specific reaction manifolds. For instance, electron-rich phosphine substituents increase the electron density on the palladium, facilitating the oxidative addition of electron-deficient aryl bromides. Conversely, the presence of electron-withdrawing groups on the indole ring can stabilize the reductive elimination step, accelerating the release of the coupled product. The patent data indicates that these ligands are particularly effective in carbon-sulfur bond formation, a transformation that is notoriously difficult due to the strong coordination of sulfur species to the metal catalyst which often leads to poisoning. The robust architecture of these indole ligands resists such poisoning, ensuring consistent performance even in the presence of thiol substrates.
How to Synthesize 3-(Disubstituted Phosphino)-2-(2-(Disubstituted Phosphino) Substituted Phenyl)-1-Alkyl-Indole Efficiently
The synthetic pathway outlined in the patent offers a scalable and reproducible method for accessing these high-value ligands. The process begins with the condensation of 2'-bromoacetophenone and an N-alkyl phenylhydrazine derivative under acidic conditions to form the indole core, followed by regioselective bromination and finally, double lithiation-phosphorylation. This sequence avoids the use of precious metal catalysts in the ligand synthesis itself, relying instead on classical organic transformations that are well-understood in process chemistry. The detailed standardized synthesis steps, including specific molar ratios and temperature controls required to maximize yield and purity, are provided in the guide below.
- Perform Fischer indole synthesis by reacting 2'-bromoacetophenone with N-alkyl phenylhydrazine using phosphoric acid and polyphosphoric acid at 80-120 °C.
- Execute selective bromination of the indole intermediate using N-bromosuccinimide (NBS) in dimethylformamide at room temperature.
- Conduct double lithiation at -78 °C followed by quenching with disubstituted phosphine chloride to install the phosphine groups.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this ligand technology offers significant strategic advantages rooted in process simplicity and raw material availability. The synthesis relies on commodity chemicals such as bromoacetophenone and phenylhydrazines, which are produced globally at massive scales, ensuring a stable and resilient supply chain that is less susceptible to geopolitical disruptions or raw material shortages. The elimination of complex chiral resolutions or rare earth metal catalysts in the ligand preparation further simplifies the sourcing strategy, allowing procurement managers to negotiate better terms with multiple qualified vendors. This diversification of the supply base is critical for maintaining business continuity in the fast-paced pharmaceutical sector.
- Cost Reduction in Manufacturing: The streamlined synthetic route significantly lowers the cost of goods sold (COGS) by minimizing the number of unit operations and avoiding expensive reagents. The use of polyphosphoric acid and common solvents like tetrahydrofuran and dimethylformamide means that waste streams are easier to manage and treat compared to processes generating heavy metal waste. Additionally, the high isolated yields reported in the patent examples suggest that material throughput is maximized, reducing the overall consumption of starting materials per kilogram of final ligand produced. This efficiency directly translates to substantial cost savings for downstream users who incorporate these ligands into their own API synthesis campaigns.
- Enhanced Supply Chain Reliability: The robustness of the ligand structure against air and moisture is a major logistical benefit. Unlike many sensitive phosphine ligands that require cold chain shipping and specialized glovebox handling, these indole-based ligands can be stored and transported under ambient conditions without significant degradation. This reduces the risk of spoilage during transit and lowers the infrastructure costs associated with warehousing. For supply chain heads, this reliability ensures that production schedules are not disrupted by the arrival of compromised reagents, thereby improving overall plant utilization rates and on-time delivery performance to customers.
- Scalability and Environmental Compliance: The reaction conditions described, such as heating at 80-120 °C and stirring at room temperature, are easily transferable from laboratory glassware to industrial reactors. The absence of cryogenic requirements for the initial steps (except for the lithiation which is manageable at -78 °C with standard industrial chillers) facilitates rapid scale-up from pilot plant to commercial production. Moreover, the process generates fewer hazardous byproducts compared to alternative ligand syntheses, aligning with increasingly stringent environmental regulations and corporate sustainability goals. This ease of scale-up ensures that commercial scale-up of complex pharmaceutical intermediates utilizing these ligands can be achieved with minimal technical risk.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ligand technology in industrial processes. These answers are derived directly from the experimental data and claims within patent CN107445989B, providing a factual basis for decision-making. Understanding these specifics helps R&D and procurement teams evaluate the feasibility of integrating these ligands into their existing workflows.
Q: What are the stability characteristics of these indole phosphine ligands?
A: According to patent CN107445989B, these ligands exhibit exceptional stability towards air and moisture, facilitating easier storage and handling compared to traditional sensitive phosphines.
Q: What is the primary catalytic application described in the patent?
A: The ligands are specifically optimized for palladium-catalyzed cross-coupling reactions, demonstrating high efficiency (up to 90% yield) in forming carbon-sulfur bonds in aryl bromides.
Q: Can this synthesis route be scaled for industrial production?
A: Yes, the process utilizes readily available raw materials like 2'-bromoacetophenone and employs standard unit operations such as heating, extraction, and column chromatography, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Phosphine Ligand Supplier
As the global demand for efficient catalytic solutions grows, NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our advanced capabilities in organophosphorus chemistry. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand delivered meets the highest standards of quality and consistency required for GMP pharmaceutical synthesis.
We invite you to collaborate with our technical team to explore how these novel indole phosphine ligands can optimize your specific cross-coupling applications. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this superior ligand system. Please contact our technical procurement team today to索取 specific COA data and route feasibility assessments tailored to your project requirements, and let us help you accelerate your path to market with confidence.
