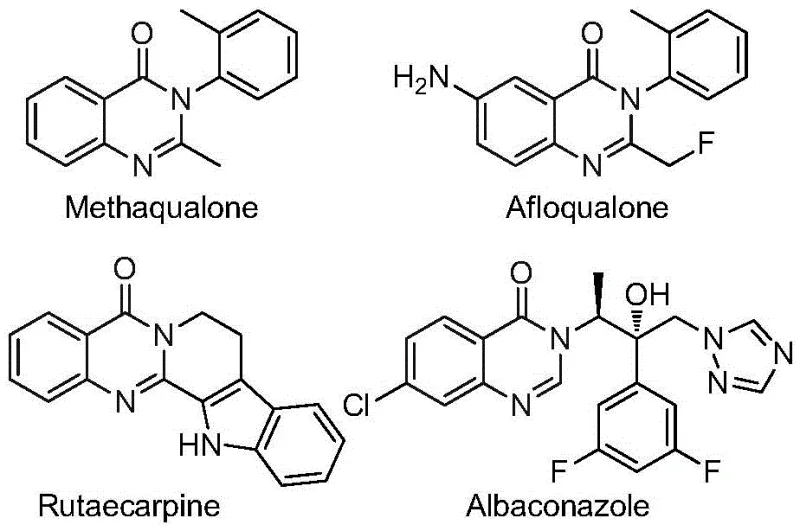
Revolutionizing Quinazolinone Synthesis: Scalable Pd-Catalyzed Process for High-Purity Pharmaceutical Intermediates
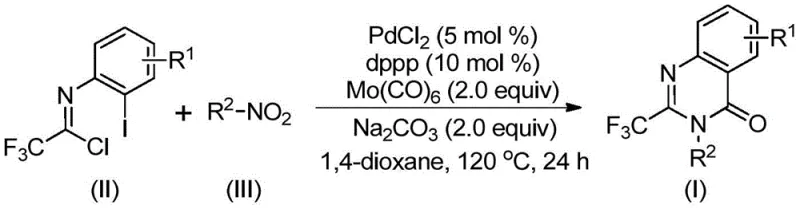
Patent CN112480015B represents a significant advancement in the synthesis of 2-trifluoromethyl substituted quinazolinones, a critical class of heterocyclic compounds with extensive applications in pharmaceutical development. This innovative multi-component one-pot methodology addresses longstanding challenges in quinazolinone chemistry by providing a streamlined route that eliminates the need for high-pressure carbon monoxide environments and pre-functionalized substrates required in conventional approaches. The process leverages readily available starting materials including trifluoroethylimidoyl chloride and diverse nitro compounds, enabling efficient construction of the quinazolinone scaffold under mild reaction conditions at 120°C. This patent demonstrates remarkable substrate tolerance across various functional groups while maintaining high reaction efficiency, making it particularly valuable for pharmaceutical manufacturers seeking scalable routes to complex heterocyclic intermediates. The methodology's compatibility with standard laboratory equipment and straightforward purification protocol further enhances its industrial applicability for commercial-scale production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to quinazolinone derivatives have been plagued by significant operational challenges that hinder their industrial implementation. The ruthenium or platinum-catalyzed reductive N-heterocyclization of nitro-substituted benzamides requires specialized high-pressure carbon monoxide equipment, creating substantial capital investment barriers and safety concerns for manufacturing facilities. Iron-catalyzed condensation methods suffer from limited substrate scope and often necessitate pre-activated starting materials that increase raw material costs and complicate supply chain management. Palladium-catalyzed cyclization approaches using bromo or iodo precursors typically require stoichiometric amounts of expensive molybdenum hexacarbonyl and generate significant metal waste streams that demand complex purification protocols. These conventional methods frequently produce inconsistent yields due to sensitivity to moisture and oxygen, requiring stringent reaction condition controls that increase operational complexity and reduce manufacturing throughput. Furthermore, the narrow functional group tolerance of existing methodologies restricts their applicability to specific structural variants, limiting their utility in diverse pharmaceutical development programs that require rapid access to structurally varied quinazolinone scaffolds.
The Novel Approach
The patented methodology overcomes these limitations through an elegant multi-component one-pot cascade reaction that operates under ambient pressure conditions without requiring specialized equipment. By utilizing palladium chloride with dppp ligand in combination with molybdenum hexacarbonyl as a carbon monoxide source, the process achieves efficient carbonylation at moderate temperatures of 120°C in standard glassware reactors. The strategic use of trifluoroethylimidoyl chloride as a key building block enables direct construction of the quinazolinone core without pre-functionalization steps, significantly simplifying the synthetic sequence. This approach demonstrates exceptional substrate flexibility, accommodating a wide range of substituents on both the imidoyl chloride and nitro compound components while maintaining consistent reaction efficiency across diverse structural variants. The methodology's compatibility with common organic solvents like 1,4-dioxane and straightforward purification through filtration and column chromatography makes it readily adaptable to existing manufacturing infrastructure without requiring substantial capital investment or process re-engineering.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The reaction mechanism proceeds through a sophisticated cascade sequence that begins with molybdenum hexacarbonyl-mediated reduction of the nitro compound to the corresponding amine intermediate under thermal conditions. This amine then undergoes base-promoted nucleophilic substitution with trifluoroethylimidoyl chloride to form a trifluoroacetamidine derivative through carbon-nitrogen bond formation. The palladium catalyst subsequently inserts into the carbon-iodine bond of the imidoyl chloride component, generating a key arylpalladium(II) intermediate that undergoes oxidative addition with carbon monoxide released from molybdenum hexacarbonyl decomposition. This forms an acylpalladium species that participates in intramolecular cyclization through nucleophilic attack by the amine nitrogen, creating a seven-membered palladacycle intermediate. The final reductive elimination step releases the desired 2-trifluoromethyl quinazolinone product while regenerating the active palladium(0) species to continue the catalytic cycle. This mechanistic pathway avoids the formation of common side products through precise control of reaction intermediates and careful optimization of catalyst loading at 5 mol% PdCl2.
The process demonstrates exceptional control over impurity formation through multiple complementary mechanisms that ensure high product purity essential for pharmaceutical applications. The carefully optimized molar ratio of palladium catalyst to ligand (0.05:0.1) prevents catalyst aggregation and maintains consistent catalytic activity throughout the reaction, minimizing metal-mediated side reactions that could generate impurities. The use of sodium carbonate as a mild base promotes selective transformation while avoiding over-reaction or decomposition pathways that commonly occur with stronger bases. The one-pot design eliminates intermediate isolation steps that typically introduce contamination risks in multi-step syntheses, while the moderate reaction temperature of 120°C prevents thermal degradation of sensitive functional groups present in diverse substrate combinations. This comprehensive impurity control strategy results in consistently high product quality with minimal need for extensive purification beyond standard filtration and chromatography protocols.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
This patented methodology provides a robust framework for manufacturing high-purity 2-trifluoromethyl quinazolinones through a carefully optimized multi-component one-pot process that integrates multiple synthetic transformations into a single operational sequence. The approach leverages commercially available starting materials and standard laboratory equipment to deliver consistent results across diverse structural variants while maintaining excellent functional group tolerance. By eliminating high-pressure requirements and pre-functionalization steps common in conventional methods, this process significantly reduces operational complexity and enhances manufacturing reliability for pharmaceutical intermediates production. The following standardized synthesis protocol has been developed based on extensive process optimization studies to ensure reproducible results at commercial scale.
- Prepare the reaction mixture with trifluoroethylimidoyl chloride, nitro compound, palladium catalyst, dppp ligand, molybdenum hexacarbonyl, and sodium carbonate in dioxane solvent
- Heat the mixture to 120°C under inert atmosphere and maintain for 16-30 hours with continuous stirring
- Perform post-reaction processing including filtration, silica gel mixing, and column chromatography purification to obtain high-purity product
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value to procurement and supply chain operations through multiple strategic advantages that address critical pain points in pharmaceutical intermediate manufacturing. By utilizing readily available starting materials and eliminating specialized equipment requirements, the process significantly reduces supply chain complexity while enhancing manufacturing flexibility across diverse production environments. The methodology's compatibility with standard infrastructure enables seamless integration into existing manufacturing workflows without requiring capital-intensive modifications, providing immediate operational benefits for procurement teams seeking reliable sources for complex heterocyclic intermediates.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide systems required in conventional approaches substantially reduces capital expenditure while avoiding specialized safety protocols that increase operational costs. The use of cost-effective starting materials including commercially available nitro compounds and easily synthesized trifluoroethylimidoyl chloride derivatives creates significant raw material savings compared to pre-functionalized substrates used in traditional methods. The streamlined one-pot design minimizes solvent usage and reduces processing time by eliminating intermediate isolation steps, leading to substantial efficiency gains in manufacturing operations without requiring additional equipment investment.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials with established global supply chains ensures consistent raw material availability regardless of regional sourcing constraints. The process's tolerance for minor variations in raw material quality reduces supplier qualification burdens while maintaining consistent product quality through robust reaction design. The methodology's compatibility with standard manufacturing equipment enables rapid technology transfer between production sites, providing flexibility to meet changing demand patterns without requiring specialized facility modifications or extended validation timelines.
- Scalability and Environmental Compliance: The reaction's operation under ambient pressure conditions with moderate temperature requirements facilitates straightforward scale-up from laboratory to commercial production without encountering typical engineering challenges associated with high-pressure processes. The simplified waste stream profile resulting from reduced metal catalyst loading and elimination of specialized reagents lowers environmental impact while simplifying waste treatment protocols. The well-defined reaction pathway with minimal byproduct formation enables efficient purification through standard techniques, reducing solvent consumption and waste generation compared to conventional multi-step approaches.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding the implementation of this patented synthesis methodology for pharmaceutical intermediate manufacturing. These responses are based on detailed analysis of the patent specifications and extensive process development experience in complex heterocyclic synthesis.
Q: How does this method improve upon conventional quinazolinone synthesis approaches?
A: The patented method eliminates the need for high-pressure CO conditions required in traditional ruthenium/platinum-catalyzed processes, while avoiding the pre-activation steps necessary in iron-catalyzed methods. The one-pot design significantly simplifies operation compared to multi-step conventional approaches.
Q: What are the key advantages of using trifluoroethylimidoyl chloride as a starting material?
A: Trifluoroethylimidoyl chloride is readily available and cost-effective compared to pre-functionalized substrates used in conventional methods. Its compatibility with diverse nitro compounds enables broad substrate scope without requiring specialized precursors, enhancing supply chain flexibility.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The reaction mechanism avoids transition metal residues through optimized catalyst loading (5 mol% PdCl₂), while the mild purification protocol (filtration followed by column chromatography) effectively removes impurities. The well-defined reaction pathway minimizes byproduct formation, ensuring stringent purity specifications required for API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
Our patented methodology represents a significant advancement in quinazolinone chemistry that directly addresses the critical challenges faced by pharmaceutical manufacturers seeking reliable sources for complex heterocyclic intermediates. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring seamless technology transfer from laboratory development to full-scale manufacturing operations. Our rigorous QC labs implement stringent purity specifications through advanced analytical methodologies that guarantee consistent product quality meeting global regulatory requirements for pharmaceutical applications.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments for your development programs. Take advantage of our Customized Cost-Saving Analysis service to evaluate how this innovative synthesis approach can optimize your supply chain while maintaining the highest quality standards required for pharmaceutical manufacturing.
