Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Commercial API Production
Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Commercial API Production
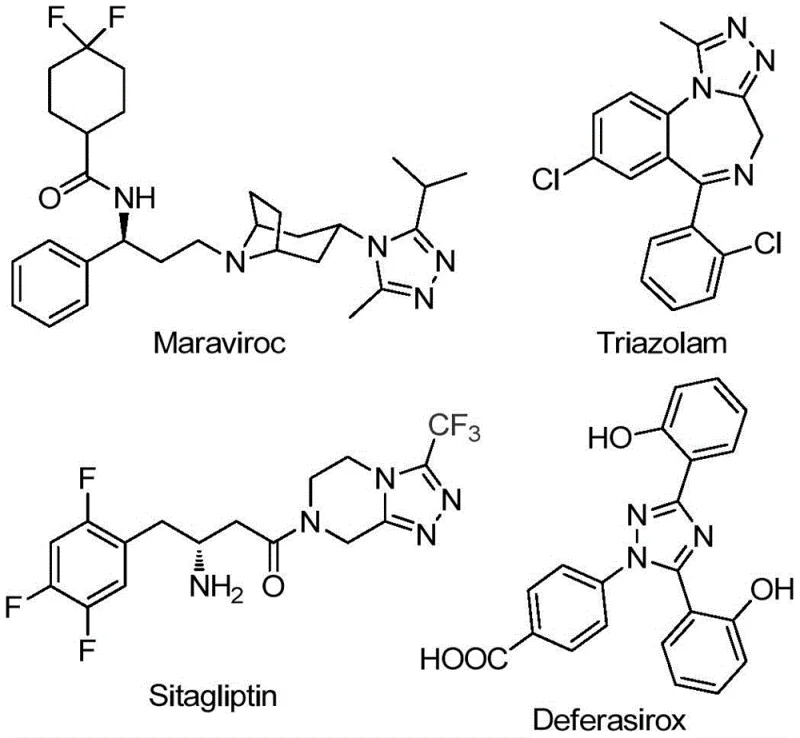
The pharmaceutical and agrochemical industries continuously seek robust synthetic methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which enhance metabolic stability and bioavailability. Patent CN111978265B introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole derivatives, addressing critical bottlenecks in current manufacturing processes. This technology leverages a cost-effective iron-catalyzed cyclization strategy that eliminates the need for stringent anhydrous or oxygen-free environments, thereby streamlining the production workflow for complex molecular scaffolds found in blockbuster drugs like Maraviroc and Sitagliptin. By utilizing readily available starting materials such as trifluoroethylimidoyl chloride and hydrazides, this innovation offers a scalable pathway for generating high-purity intermediates essential for modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been plagued by significant operational challenges that hinder efficient commercial manufacturing. Traditional literature methods often rely on the condensation of 3,5-ditrifluoromethyl-1,3,4-oxadiazoles with primary amines or the cyclization of trifluoromethyl hydrazides with amidines, processes that frequently demand harsh reaction conditions and multi-step sequences. These conventional routes typically suffer from narrow substrate scopes, limiting the structural diversity accessible to medicinal chemists, and often result in disappointing reaction yields that compromise overall process economics. Furthermore, previous tandem cyclization methods developed for similar scaffolds have shown poor reactivity towards alkyl hydrazones, creating a substantial gap in the ability to synthesize 3-alkyl fluoro-1,2,4-triazoles efficiently. Such limitations necessitate complex purification protocols and increase the environmental footprint due to excessive solvent and reagent consumption.
The Novel Approach
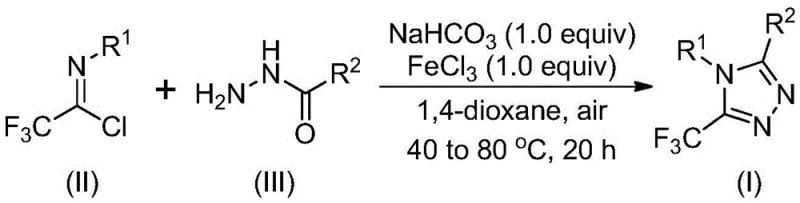
In stark contrast to these legacy techniques, the novel methodology disclosed in the patent utilizes a simple yet highly effective ferric chloride-promoted system that operates under remarkably mild conditions. The process initiates with the mixing of sodium bicarbonate, trifluoroethylimidoyl chloride, and hydrazides in an organic solvent, followed by a two-stage temperature protocol that ensures high conversion rates without requiring inert atmosphere gloveboxes. This approach not only broadens the substrate tolerance to include various substituted aryl and alkyl groups but also significantly simplifies the post-reaction workup to basic filtration and silica gel treatment. The ability to perform this transformation in common aprotic solvents like 1,4-dioxane at moderate temperatures represents a paradigm shift towards greener and more economical chemical manufacturing for these valuable heterocyclic cores.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation is believed to proceed through a base-promoted intermolecular carbon-nitrogen bond formation, initially generating a trifluoroacetamidine derivative intermediate. Subsequent addition of the metal Lewis acid, specifically ferric chloride, facilitates an intramolecular dehydration condensation that drives the cyclization to form the final 5-trifluoromethyl substituted 1,2,4-triazole ring system. This dual-activation strategy ensures that the reaction proceeds smoothly even with sterically hindered or electronically diverse substrates, providing a robust mechanism for constructing the triazole core with high fidelity. The use of iron as a catalyst is particularly advantageous from a regulatory perspective, as it avoids the toxicity concerns associated with precious heavy metals like palladium or rhodium, thereby simplifying the residual metal testing required for pharmaceutical grade intermediates.
From an impurity control standpoint, the mild reaction conditions and the specific selectivity of the ferric chloride promoter minimize the formation of side products that often plague high-temperature cyclizations. The protocol allows for precise control over the reaction progression by adjusting the temperature stages, first at 30-50°C for bond formation and then at 70-90°C for ring closure, which helps in managing exothermic events and preventing decomposition of sensitive functional groups. This level of control is critical for maintaining a clean impurity profile, ensuring that the resulting triazole derivatives meet the stringent purity specifications demanded by global regulatory bodies for active pharmaceutical ingredients. The broad functional group tolerance further implies that complex molecules can be synthesized without extensive protecting group strategies, reducing the overall step count and material loss.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. Operators begin by combining the key reactants with sodium bicarbonate in a suitable solvent, stirring at a lower temperature range to establish the initial adduct before introducing the catalyst for the final cyclization step. This standardized approach minimizes operator error and ensures consistent batch-to-batch quality, which is paramount for supply chain stability. For detailed standard operating procedures and specific stoichiometric ratios optimized for different substrates, please refer to the comprehensive guide below.
- Mix sodium bicarbonate, trifluoroethylimidoyl chloride, and hydrazide in an organic solvent like 1,4-dioxane.
- Stir the mixture at 30-50°C for 8-16 hours to facilitate initial bond formation.
- Add ferric chloride catalyst and heat to 70-90°C for 6-10 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical efficiency. The reliance on commodity chemicals such as sodium bicarbonate and ferric chloride, which are abundant and inexpensive, drastically reduces the raw material cost burden compared to methods requiring exotic catalysts or specialized reagents. Moreover, the elimination of strict anhydrous and oxygen-free requirements means that production can be conducted in standard glass-lined reactors without the need for expensive nitrogen purging systems or specialized drying infrastructure, leading to significant capital expenditure savings and reduced utility costs. This operational flexibility allows manufacturers to utilize existing facility capacity more effectively, enhancing overall throughput without major retrofitting investments.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of earth-abundant iron catalysts instead of precious metals, which removes the need for costly metal scavenging steps and reduces the risk of supply disruptions associated with rare earth elements. By simplifying the reaction setup to tolerate ambient air and moisture, the method lowers energy consumption related to solvent drying and inert gas generation, directly impacting the cost of goods sold. Additionally, the high yields reported across a wide range of substrates mean less raw material is wasted, optimizing the atom economy and reducing the volume of waste streams that require disposal. These factors collectively contribute to a leaner manufacturing model that improves margin potential for high-volume API intermediates.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term production contracts, and this method mitigates risk by utilizing starting materials that are widely available in the global chemical market. The robustness of the reaction conditions ensures that production schedules are less likely to be disrupted by minor variations in environmental conditions or reagent quality, providing a more predictable output for downstream customers. Furthermore, the scalability demonstrated in the patent suggests that transitioning from kilogram to ton-scale production can be achieved with minimal process re-engineering, securing a continuous supply flow for critical pharmaceutical programs. This reliability is essential for maintaining just-in-time inventory levels and avoiding stockouts that could halt downstream drug formulation lines.
- Scalability and Environmental Compliance: As regulatory pressure mounts for greener chemical processes, this iron-catalyzed route aligns perfectly with sustainability goals by avoiding toxic heavy metals and reducing solvent intensity through higher efficiency. The simplified workup procedure involving filtration and standard chromatography reduces the generation of hazardous waste, lowering disposal costs and easing the burden on environmental health and safety teams. The ability to scale this reaction easily means that manufacturers can respond rapidly to surges in demand for specific triazole derivatives without compromising on environmental standards or safety protocols. This adaptability makes the technology future-proof against tightening environmental regulations while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios. The answers reflect the specific advantages and operational parameters defined in the intellectual property documentation.
Q: What are the key advantages of this FeCl3-catalyzed method over traditional routes?
A: Unlike traditional methods requiring harsh conditions or narrow substrate scopes, this protocol operates under air-tolerant conditions using cheap, commercially available reagents like ferric chloride and sodium bicarbonate, significantly simplifying the operational complexity.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability to the gram level with simple post-processing involving filtration and column chromatography, indicating strong potential for commercial scale-up without specialized anhydrous or oxygen-free equipment.
Q: What types of substituents are tolerated in this triazole synthesis?
A: The method exhibits broad functional group tolerance, successfully accommodating various substituted aryl groups (methyl, methoxy, halogen, trifluoromethyl) and alkyl chains, allowing for the design of diverse 3,4-disubstituted triazole derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this FeCl3-catalyzed synthesis for producing high-value pharmaceutical intermediates with superior cost-efficiency. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless and compliant. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole derivatives delivered meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with our technical team to explore how this innovative route can optimize your specific supply chain requirements and reduce overall manufacturing costs. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can accelerate your development timelines and secure your raw material supply for the long term.
