Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Commercial API Production
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic methodologies for nitrogen-containing heterocycles, particularly quinazolinones, due to their pervasive presence in bioactive molecules ranging from anticancer agents to anticonvulsants. As illustrated in the structural diversity of known bioactive compounds such as Afloqualone and Luotonin F, the quinazolinone scaffold serves as a critical pharmacophore in modern drug discovery. The strategic introduction of a trifluoromethyl group into this scaffold further enhances metabolic stability, lipophilicity, and bioavailability, addressing key ADME challenges in drug development. Patent CN111675662B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds that leverages inexpensive iron catalysis to overcome the limitations of traditional synthetic routes. This innovation represents a significant leap forward for process chemists aiming to optimize the manufacturing of complex heterocyclic intermediates.
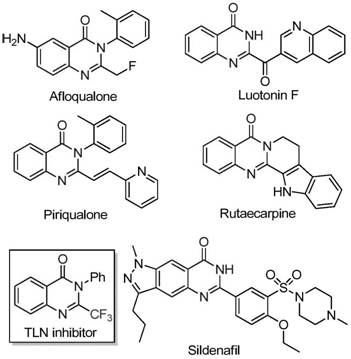
This novel approach not only streamlines the synthetic pathway but also aligns with green chemistry principles by utilizing earth-abundant metals instead of scarce precious metals. For R&D directors and procurement managers alike, understanding the mechanistic nuances and commercial implications of this patent is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates. The ability to access these fluorinated scaffolds efficiently opens new avenues for the development of next-generation therapeutics with improved pharmacokinetic profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has relied heavily on the cyclization of synthons such as anthranilamide, anthranilic acid, or isatoic anhydride with trifluoroacetic anhydride or ethyl trifluoroacetate. While these methods are established in academic literature, they suffer from significant drawbacks when translated to industrial settings. The reaction conditions are often severe, requiring harsh reagents that pose safety hazards and complicate waste management. Furthermore, the substrate scope is frequently narrow, limiting the ability to introduce diverse functional groups necessary for structure-activity relationship (SAR) studies. Perhaps most critically for procurement teams, the reliance on specialized trifluoromethyl synthons drives up raw material costs, and the overall yields are often modest, leading to inefficient atom economy and increased production expenses.
The Novel Approach
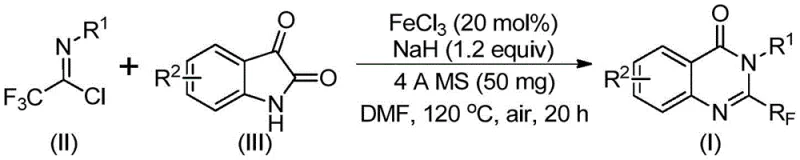
In stark contrast, the methodology described in patent CN111675662B utilizes readily available trifluoroethylimidoyl chloride and isatin derivatives as starting materials. This shift in synthon strategy is transformative; isatins are commodity chemicals with stable supply chains, and the imidoyl chlorides can be easily prepared from aromatic amines. The reaction proceeds via a tandem sequence involving alkali-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization. This cascade process effectively constructs the quinazolinone core in a single pot, minimizing isolation steps and solvent usage. The result is a highly efficient protocol that tolerates a wide array of substituents, including halogens, alkyls, and nitro groups, thereby providing process chemists with the flexibility to design and synthesize a broad spectrum of analogues without redesigning the entire synthetic route.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the unique reactivity of the iron catalyst in facilitating the decarbonylative cyclization. The reaction initiates with the nucleophilic attack of the isatin nitrogen on the imidoyl chloride, promoted by sodium hydride, to form a trifluoroacetamidine intermediate. Subsequently, the ferric chloride catalyst activates the system for a crucial decarbonylation step, where the carbonyl group of the isatin moiety is extruded. This is followed by an intramolecular cyclization to close the pyrimidine ring, yielding the final 2-trifluoromethyl quinazolinone structure. The use of 4A molecular sieves plays a pivotal role in sequestering moisture, which is critical for maintaining the activity of the base and preventing hydrolysis of the sensitive imidoyl chloride species. This mechanistic pathway avoids the formation of stable byproducts often seen in thermal cyclizations, leading to cleaner reaction profiles.
From an impurity control perspective, this mechanism offers distinct advantages. The mild nature of the iron catalysis compared to strong Lewis acids or transition metals reduces the risk of over-reaction or decomposition of sensitive functional groups on the aromatic rings. For instance, the patent demonstrates successful synthesis with nitro and bromo substituents, which might be susceptible to reduction or side reactions under more vigorous conditions. The high selectivity ensures that the crude product contains fewer structurally related impurities, significantly easing the burden on downstream purification processes like column chromatography or recrystallization. This level of control is paramount for meeting the stringent purity specifications required for pharmaceutical intermediates intended for clinical use.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The operational simplicity of this protocol makes it highly attractive for both laboratory scale-up and commercial manufacturing. The procedure involves mixing the catalyst, base, molecular sieves, and substrates in a polar aprotic solvent such as DMF. The reaction is typically heated in a two-stage temperature profile, starting at a lower temperature to facilitate the initial coupling and then ramping up to drive the cyclization to completion. Detailed standardized synthesis steps for implementing this protocol in your facility are provided below.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in DMF solvent.
- Stir the reaction mixture at 40°C for 10 hours to initiate the coupling, then increase temperature to 120°C for 20 hours under air atmosphere.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed route offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic reduction of raw material costs. By replacing expensive trifluoroacetic anhydride and precious metal catalysts with commodity isatins and ferric chloride, the direct material cost of goods sold (COGS) is significantly lowered. Ferric chloride is one of the most inexpensive Lewis acids available globally, ensuring that catalyst costs remain negligible even at multi-ton scales. Furthermore, the elimination of precious metals removes the regulatory and logistical burden associated with sourcing, tracking, and recovering metals like palladium or rhodium, which are subject to volatile market pricing and supply constraints.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the simplification of the purification train. Since the reaction generates fewer byproducts and avoids heavy metal contamination, the need for expensive scavenger resins or complex extraction protocols is minimized. This streamlined downstream processing translates directly into reduced labor hours, lower solvent consumption, and decreased waste disposal costs. Additionally, the high atom economy of the decarbonylative cyclization ensures that a greater proportion of the starting mass is converted into the valuable product, maximizing the return on investment for every kilogram of raw material purchased.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Isatin and its derivatives are produced by numerous chemical manufacturers worldwide, reducing the risk of single-source dependency. Similarly, the aromatic amines required to generate the imidoyl chlorides are commodity chemicals with robust global supply networks. This diversification of the supply base ensures continuity of supply even during market disruptions. The ability to synthesize the key imidoyl chloride intermediate in situ or purchase it off-the-shelf provides flexibility in inventory management, allowing manufacturers to respond rapidly to fluctuating demand without long lead times for specialized reagents.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process is exceptionally well-suited for industrial expansion. The reaction operates under air atmosphere, eliminating the need for costly inert gas blanketing systems (nitrogen or argon) which are often required for sensitive organometallic reactions. This simplifies reactor design and operation, facilitating a smoother transition from pilot plant to full-scale production. Moreover, the use of iron, a non-toxic and environmentally benign metal, aligns with increasingly strict environmental regulations regarding heavy metal discharge. This 'green' credential not only reduces compliance costs but also enhances the sustainability profile of the final pharmaceutical product, a factor of growing importance to end-users and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the process capabilities and limitations for potential adopters.
Q: What are the advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: Iron(III) chloride is significantly more cost-effective and environmentally benign compared to precious metal catalysts like palladium or rhodium. It eliminates the need for rigorous heavy metal scavenging steps often required in GMP manufacturing, thereby reducing overall production costs and simplifying the purification workflow while maintaining high catalytic efficiency.
Q: Can this synthetic route tolerate diverse functional groups on the isatin substrate?
A: Yes, the methodology demonstrates excellent functional group tolerance. The patent data confirms successful synthesis with substrates containing electron-donating groups (methyl, methoxy) and electron-withdrawing groups (fluoro, bromo, chloro, nitro) at various positions (ortho, meta, para), making it highly versatile for generating diverse libraries of drug candidates.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Absolutely. The process utilizes readily available starting materials like isatin and imidoyl chlorides, operates under air atmosphere without stringent inert gas requirements, and uses common solvents like DMF. These factors, combined with the robustness of the iron catalyst, make the route highly amenable to scale-up from gram-level laboratory synthesis to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists has thoroughly analyzed the technology disclosed in patent CN111675662B and is fully equipped to leverage this iron-catalyzed route for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. Our state-of-the-art facilities are designed to handle reactive chemistries safely, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to optimize your supply chain for 2-trifluoromethyl quinazolinone derivatives. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you reduce lead time for high-purity pharmaceutical intermediates and secure a competitive advantage in the global market.
