Advanced Heterogeneous Catalysis for Scalable Benzothiazole Derivative Production
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for synthesizing critical heterocyclic scaffolds. Patent CN112142686B introduces a groundbreaking preparation method for benzothiazole derivatives utilizing a heterogeneous sustainable catalytic system. This technology leverages a Cu-MOF (Metal-Organic Framework) derived material, specifically HKUST-1-400, to facilitate the dehydrogenative coupling of o-aminothiophenols and benzyl alcohols. Unlike traditional methods that often rely on harsh oxidants or non-recyclable homogeneous catalysts, this approach offers a green alternative where water is the sole byproduct. The ability to separate the catalyst post-reaction and reuse it without significant loss of catalytic efficiency represents a major leap forward in process chemistry. For R&D teams and procurement strategists, this patent outlines a route that not only simplifies purification but also aligns with modern green chemistry principles, ensuring a reliable supply of high-purity pharmaceutical intermediates.
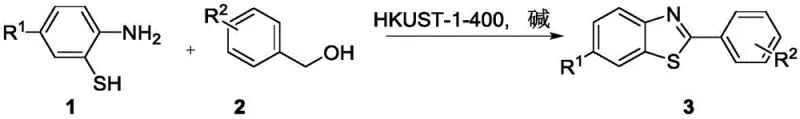
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzothiazole derivatives has relied heavily on condensation reactions involving carboxylic acids or their derivatives, which often require high temperatures and generate substantial amounts of waste. Alternatively, oxidative cyclization methods using stoichiometric oxidants or homogeneous transition metal catalysts have been employed. While effective on a small scale, these conventional methods present significant challenges for commercial manufacturing. Homogeneous catalysts, such as soluble copper or palladium salts, are notoriously difficult to remove from the final product, often necessitating expensive and time-consuming purification steps like column chromatography or specialized scavenging resins to meet strict residual metal specifications for API intermediates. Furthermore, the use of stoichiometric oxidants leads to poor atom economy and creates hazardous waste streams that increase disposal costs and environmental liability. These factors collectively drive up the cost of goods sold and complicate the supply chain for high-volume production.
The Novel Approach
The novel approach detailed in patent CN112142686B fundamentally shifts the paradigm by employing a heterogeneous Cu-MOF derived catalyst, HKUST-1-400. This material is prepared by carbonizing HKUST-1 at controlled temperatures, resulting in a robust solid catalyst that facilitates the direct dehydrogenative coupling of o-aminothiophenols and benzyl alcohols. The key innovation lies in the heterogeneous nature of the catalyst, which allows for simple filtration to separate it from the reaction mixture. This eliminates the need for complex metal removal processes, drastically reducing processing time and solvent consumption. Moreover, the reaction proceeds with high atom economy, generating only water as a byproduct, which significantly simplifies waste management. The catalyst's stability allows it to be recycled for multiple runs while maintaining high catalytic activity, offering a sustainable and cost-effective solution for the large-scale manufacturing of benzothiazole derivatives.
Mechanistic Insights into Cu-MOF Catalyzed Dehydrogenative Coupling
The mechanistic pathway of this transformation involves the activation of the benzyl alcohol by the copper species embedded within the carbonized MOF matrix. The HKUST-1-400 catalyst acts as a Lewis acid and redox mediator, facilitating the dehydrogenation of the alcohol to the corresponding aldehyde in situ. This transient aldehyde then undergoes condensation with the amine group of the o-aminothiophenol to form a Schiff base intermediate. Subsequently, the thiol group attacks the imine carbon, followed by oxidative aromatization to yield the benzothiazole ring. The unique porous structure of the MOF-derived material provides a high surface area and accessible active sites, enhancing the reaction kinetics. Crucially, the carbonization process stabilizes the copper species, preventing leaching into the solution and ensuring the heterogeneous nature of the catalysis is preserved throughout the reaction cycle. This stability is confirmed by XRD analysis showing unchanged crystal structures before and after reaction.
Impurity control in this process is inherently superior due to the specificity of the catalytic surface and the mild reaction conditions. Traditional methods often suffer from over-oxidation or side reactions involving the solvent or oxidant. In contrast, the HKUST-1-400 catalyzed system operates under relatively mild thermal conditions (90-120°C) and avoids strong external oxidants. The selectivity of the catalyst ensures that the dehydrogenation is coupled efficiently with the cyclization, minimizing the formation of byproducts such as disulfides or over-oxidized sulfones. The ability to tune the catalyst preparation temperature (e.g., 400°C vs 600°C) allows for optimization of the active site density, further refining the impurity profile. For pharmaceutical applications, this means a cleaner crude product that requires less intensive downstream purification, directly translating to higher overall yields and reduced production costs for high-purity pharmaceutical intermediates.
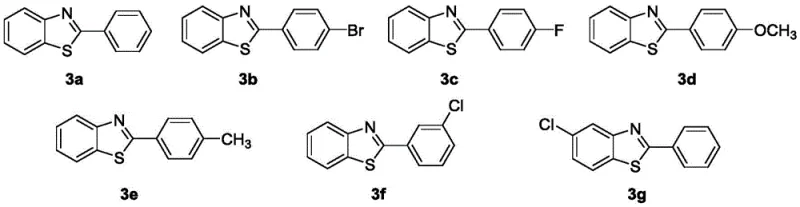
How to Synthesize 2-Phenylbenzo[d]thiazole Efficiently
The synthesis of 2-phenylbenzo[d]thiazole and its derivatives via this patented method is designed for operational simplicity and scalability. The process begins with the dissolution of the o-aminothiophenol derivative in a suitable solvent such as toluene or 1,4-dioxane at room temperature. To this solution, the HKUST-1-400 catalyst, a base such as potassium tert-butoxide, and the benzyl alcohol derivative are added sequentially. The reaction mixture is then heated to a temperature range of 90-120°C and maintained for a period of 1 to 3 days, depending on the specific substrate reactivity. Monitoring via TLC ensures optimal conversion. Upon completion, the solid catalyst is removed by simple filtration, and the solvent is evaporated under reduced pressure. The resulting residue is purified by column chromatography to afford the target benzothiazole derivative. This streamlined workflow minimizes unit operations and maximizes throughput.
- Dissolve o-aminothiophenol derivative in solvent (toluene or 1,4-dioxane) at room temperature.
- Add HKUST-1-400 catalyst, base (e.g., potassium tert-butoxide), and benzyl alcohol derivative.
- Heat the mixture to 90-120°C for 1-3 days, then filter to recover the catalyst and purify the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this heterogeneous catalytic technology offers profound strategic advantages beyond mere technical feasibility. The primary benefit lies in the drastic simplification of the downstream processing workflow. By replacing homogeneous catalysts with a filterable solid, the costly and time-intensive steps associated with heavy metal scavenging and validation are effectively eliminated. This reduction in processing complexity directly correlates to lower manufacturing costs and shorter lead times. Furthermore, the recyclability of the HKUST-1-400 catalyst means that the effective cost of the catalyst per kilogram of product is significantly reduced over multiple batches. This sustainability feature not only lowers raw material expenses but also enhances the environmental profile of the supply chain, a critical factor for multinational corporations adhering to strict ESG (Environmental, Social, and Governance) mandates.
- Cost Reduction in Manufacturing: The elimination of expensive homogeneous metal catalysts and the associated removal agents results in substantial cost savings. Since the catalyst can be recovered and reused multiple times without significant loss of activity, the amortized cost of the catalytic system is minimal. Additionally, the generation of water as the only byproduct reduces the burden on waste treatment facilities, lowering disposal fees and environmental compliance costs. The simplified workup procedure also reduces solvent consumption and energy usage during purification, contributing to a leaner and more cost-efficient manufacturing process for complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the HKUST-1-400 catalyst ensures consistent batch-to-batch quality, which is vital for maintaining a stable supply of critical intermediates. The method's tolerance for a wide range of substrates, including those with electron-withdrawing or electron-donating groups, allows for flexible sourcing of starting materials. This flexibility mitigates the risk of supply disruptions caused by the scarcity of specific reagents. Moreover, the scalable nature of the heterogeneous reaction means that production can be ramped up from laboratory to commercial scale with minimal re-optimization, ensuring that supply commitments can be met reliably even during periods of high demand.
- Scalability and Environmental Compliance: Scaling up homogeneous reactions often presents significant safety and engineering challenges, particularly regarding heat transfer and mixing. In contrast, heterogeneous catalysis in a fixed-bed or slurry reactor is inherently easier to scale. The green chemistry attributes of this process, specifically the high atom economy and lack of toxic byproducts, align perfectly with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden and potential liabilities associated with chemical manufacturing. Consequently, this technology enables the commercial scale-up of complex benzothiazole derivatives while maintaining a minimal environmental footprint, securing long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. Understanding these details is crucial for R&D directors evaluating process feasibility and procurement teams assessing supplier capabilities. The answers are derived directly from the experimental data and beneficial effects reported in patent CN112142686B, providing a factual basis for decision-making. These insights highlight the practical advantages of the HKUST-1-400 catalytic system in real-world manufacturing scenarios.
Q: What are the advantages of using HKUST-1-400 over homogeneous catalysts?
A: HKUST-1-400 is a heterogeneous catalyst that can be easily separated by filtration and reused multiple times without significant loss of activity, eliminating the need for complex metal removal steps required by homogeneous catalysts.
Q: What is the primary byproduct of this dehydrogenative coupling reaction?
A: The reaction produces water as the only byproduct, making it an atom-economical and environmentally friendly process compared to traditional methods that generate stoichiometric waste.
Q: Can this method tolerate various substituents on the aromatic rings?
A: Yes, the protocol demonstrates excellent tolerance for various substituents including halogens, alkyl groups, and alkoxy groups on both the o-aminothiophenol and benzyl alcohol substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phenylbenzo[d]thiazole Supplier
The technological advancements described in patent CN112142686B underscore the immense potential of heterogeneous catalysis in modern organic synthesis. At NINGBO INNO PHARMCHEM, we recognize the value of such innovations and have integrated similar advanced catalytic technologies into our CDMO capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of benzothiazole derivatives meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality and sustainability makes us an ideal partner for your complex synthesis needs.
We invite you to leverage our expertise to optimize your supply chain and reduce manufacturing costs. By collaborating with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific project requirements. We encourage you to reach out to request specific COA data and route feasibility assessments for your target molecules. Whether you require clinical trial materials or commercial quantities, NINGBO INNO PHARMCHEM is dedicated to delivering high-quality solutions that drive your business forward. Contact us today to discuss how our advanced catalytic capabilities can support your next breakthrough.
